University of California, San Diego
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
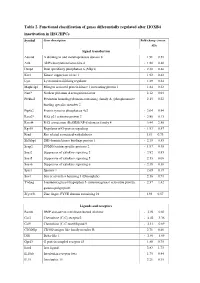
Table 2. Functional Classification of Genes Differentially Regulated After HOXB4 Inactivation in HSC/Hpcs
Table 2. Functional classification of genes differentially regulated after HOXB4 inactivation in HSC/HPCs Symbol Gene description Fold-change (mean ± SD) Signal transduction Adam8 A disintegrin and metalloprotease domain 8 1.91 ± 0.51 Arl4 ADP-ribosylation factor-like 4 - 1.80 ± 0.40 Dusp6 Dual specificity phosphatase 6 (Mkp3) - 2.30 ± 0.46 Ksr1 Kinase suppressor of ras 1 1.92 ± 0.42 Lyst Lysosomal trafficking regulator 1.89 ± 0.34 Mapk1ip1 Mitogen activated protein kinase 1 interacting protein 1 1.84 ± 0.22 Narf* Nuclear prelamin A recognition factor 2.12 ± 0.04 Plekha2 Pleckstrin homology domain-containing. family A. (phosphoinosite 2.15 ± 0.22 binding specific) member 2 Ptp4a2 Protein tyrosine phosphatase 4a2 - 2.04 ± 0.94 Rasa2* RAS p21 activator protein 2 - 2.80 ± 0.13 Rassf4 RAS association (RalGDS/AF-6) domain family 4 3.44 ± 2.56 Rgs18 Regulator of G-protein signaling - 1.93 ± 0.57 Rrad Ras-related associated with diabetes 1.81 ± 0.73 Sh3kbp1 SH3 domain kinase bindings protein 1 - 2.19 ± 0.53 Senp2 SUMO/sentrin specific protease 2 - 1.97 ± 0.49 Socs2 Suppressor of cytokine signaling 2 - 2.82 ± 0.85 Socs5 Suppressor of cytokine signaling 5 2.13 ± 0.08 Socs6 Suppressor of cytokine signaling 6 - 2.18 ± 0.38 Spry1 Sprouty 1 - 2.69 ± 0.19 Sos1 Son of sevenless homolog 1 (Drosophila) 2.16 ± 0.71 Ywhag 3-monooxygenase/tryptophan 5- monooxygenase activation protein. - 2.37 ± 1.42 gamma polypeptide Zfyve21 Zinc finger. FYVE domain containing 21 1.93 ± 0.57 Ligands and receptors Bambi BMP and activin membrane-bound inhibitor - 2.94 ± 0.62 -

Cross-Talk Between HER2 and MED1 Regulates Tamoxifen Resistance of Human Breast Cancer Cells
Published OnlineFirst September 10, 2012; DOI: 10.1158/0008-5472.CAN-12-1305 Cancer Tumor and Stem Cell Biology Research Cross-talk between HER2 and MED1 Regulates Tamoxifen Resistance of Human Breast Cancer Cells Jiajun Cui1, Katherine Germer1, Tianying Wu2, Jiang Wang3, Jia Luo5, Shao-chun Wang1, Qianben Wang4, and Xiaoting Zhang1 Abstract Despite the fact that most breast cancer patients have estrogen receptor (ER) a-positive tumors, up to 50% of the patients are or soon develop resistance to endocrine therapy. It is recognized that HER2 activation is one of the major mechanisms contributing to endocrine resistance. In this study, we report that the ER coactivator MED1 is a novel cross-talk point for the HER2 and ERa pathways. Tissue microarray analysis of human breast cancers revealed that MED1 expression positively correlates most strongly with HER2 status of the tumors. MED1 was highly phosphorylated, in a HER2-dependent manner, at the site known to be critical for its activation. Importantly, RNAi-mediated attenuation of MED1 sensitized HER2-overexpressing cells to tamoxifen treatment. MED1 and its phosphorylated form, but not the corepressors N-CoR and SMRT, were recruited to the ERa target gene promoter by tamoxifen in HER2-overexpressing cells. Significantly, MED1 attenuation or mutation of MED1 phosphorylation sites was sufficient to restore the promoter recruitment of N-CoR and SMRT. Notably, we found that MED1 is required for the expression of not only traditional E2-ERa target genes but also the newly described EGF-ERa target genes. Our results additionally indicated that MED1 is recruited to the HER2 gene and required for its expression. -

Cisplatin and Phenanthriplatin Modulate Long-Noncoding
www.nature.com/scientificreports OPEN Cisplatin and phenanthriplatin modulate long‑noncoding RNA expression in A549 and IMR90 cells revealing regulation of microRNAs, Wnt/β‑catenin and TGF‑β signaling Jerry D. Monroe1,2, Satya A. Moolani2,3, Elvin N. Irihamye2,4, Katheryn E. Lett1, Michael D. Hebert1, Yann Gibert1* & Michael E. Smith2* The monofunctional platinum(II) complex, phenanthriplatin, acts by blocking transcription, but its regulatory efects on long‑noncoding RNAs (lncRNAs) have not been elucidated relative to traditional platinum‑based chemotherapeutics, e.g., cisplatin. Here, we treated A549 non‑small cell lung cancer and IMR90 lung fbroblast cells for 24 h with either cisplatin, phenanthriplatin or a solvent control, and then performed microarray analysis to identify regulated lncRNAs. RNA22 v2 microRNA software was subsequently used to identify microRNAs (miRNAs) that might be suppressed by the most regulated lncRNAs. We found that miR‑25‑5p, ‑30a‑3p, ‑138‑5p, ‑149‑3p, ‑185‑5p, ‑378j, ‑608, ‑650, ‑708‑5p, ‑1253, ‑1254, ‑4458, and ‑4516, were predicted to target the cisplatin upregulated lncRNAs, IMMP2L‑1, CBR3‑1 and ATAD2B‑5, and the phenanthriplatin downregulated lncRNAs, AGO2‑1, COX7A1‑2 and SLC26A3‑1. Then, we used qRT‑PCR to measure the expression of miR‑25‑5p, ‑378j, ‑4516 (A549) and miR‑149‑3p, ‑608, and ‑4458 (IMR90) to identify distinct signaling efects associated with cisplatin and phenanthriplatin. The signaling pathways associated with these miRNAs suggests that phenanthriplatin may modulate Wnt/β‑catenin and TGF‑β signaling through the MAPK/ ERK and PTEN/AKT pathways diferently than cisplatin. Further, as some of these miRNAs may be subject to dissimilar lncRNA targeting in A549 and IMR90 cells, the monofunctional complex may not cause toxicity in normal lung compared to cancer cells by acting through distinct lncRNA and miRNA networks. -

Key Roles for MED1 Lxxll Motifs in Pubertal Mammary Gland Development and Luminal-Cell Differentiation
Key roles for MED1 LxxLL motifs in pubertal mammary gland development and luminal-cell differentiation Pingping Jianga,b,1, Qiuping Hua,1, Mitsuhiro Itoc,1,3, Sara Meyera, Susan Waltza, Sohaib Khana, Robert G. Roederc,2, and Xiaoting Zhanga,2 aDepartment of Cancer and Cell Biology, College of Medicine, University of Cincinnati, 3125 Eden Avenue, OH 45267; bCollege of Life Sciences, Zhejiang University, 388 Yuhangtang Road, Hangzhou 310058, China; and cLaboratory of Biochemistry and Molecular Biology, The Rockefeller University, 1230 York Avenue, New York, NY 10065 Contributed by Robert G. Roeder, February 16, 2010 (sent for review January 18, 2010) Mediator recently has emerged as a central player in the direct (7–9). In general, individual Mediator subunits interact transduction of signals from transcription factors to the general specifically with their corresponding transcription factors and de- transcriptional machinery. In the case of nuclear receptors, in vitro letions of these Mediator subunits often affect expression primar- studies have shown that the transcriptional coactivator function of ily of target genes and pathways controlled by their corresponding the Mediator involves direct ligand-dependent interactions of the transcription factor(s) (9). In the case of nuclear receptors, the MED1 subunit, through its two classical LxxLL motifs, with the re- Mediator interactions are ligand- and AF-2-dependent and ceptor AF2 domain. However, despite the strong in vitro evidence, mediated through the LxxLL motifs in the MED1 (a.k.a. there currently is little information regarding in vivo functions of TRAP220/PBP/DRIP205) subunit (10–13). Importantly, in rela- the LxxLL motifs either in MED1 or in other coactivators. -

In Acute Myeloid Leukemia
7283 Original Article Comprehensively analyze the expression and prognostic role for ten-eleven translocations (TETs) in acute myeloid leukemia Yan Huang1#, Jie Wei1#, Xunjun Huang1, Weijie Zhou1, Yuling Xu2, Dong-Hong Deng2, Peng Cheng2 1Department of Hematology and Rheumatology, People’s Hospital of Baise, Baise, China; 2Department of Hematology, the First Affiliated Hospital of Guangxi Medical University, Nanning, China Contributions: (I) Conception and design: J Wei, Y Huang; (II) Administrative support: X Huang, Y Xu; (III) Provision of study materials or patients: W Zhou; (IV) Collection and assembly of data: J Wei, Y Huang; (V) Data analysis and interpretation: DH Deng, P Cheng; (VI) Manuscript writing: All authors; (VII) Final approval of manuscript: All authors. #These authors contributed equally to this work. Correspondence to: Yuling Xu. Department of Hematology, The First Affiliated Hospital of Guangxi Medical University, Nanning, China. Email: [email protected]; Dong-Hong Deng. Department of Hematology, The First Affiliated Hospital of Guangxi Medical University, Nanning, China. Email: [email protected]; Peng Cheng. Department of Hematology, The First Affiliated Hospital of Guangxi Medical University, Nanning 530000, China. Email: [email protected]. Background: The ten-eleven translocation (TET) family oxidize 5-methylcytosines (5mCs) and promote the locus-specific reversal of DNA. The role of TETs in acute myeloid leukemia (AML) is mostly unknown. Methods: TETs mRNA expression levels were analyzed via Gene Expression Profiling Interactive Analysis (GEPIA). The association TETs expression levels and methylation with prognosis by UALCAN GenomicScape, and METHsurv. We analyzed TETs’ aberration types, located mutations, and structures via cBioPortal. GeneMANIA performed the functional network. Gene ontology (GO) enrichment was analyzed via LinkedOmics. -

Supplementary Figures and Table Legends
Supplementary Information Supplementary Figure Legends Figure S1. CARM1 is required for estrogen-induced gene transcriptional activation. (A) Box plot showing the expression of CARM1 (FPKM) in a cohort of clinical breast cancer (n=1,102) and normal (n=113) samples from TCGA (The Cancer Genome Atlas). (B, C) Kaplan Meier survival analyses for DMFS (distal metastasis free survival) (n=1,747) and OS (overall survival) (n=1,402) of breast cancer patients using CARM1 as input. (D, E) RNA-seq as described in Figure 1A for specific genes were shown as indicated. (F) Correlation of CARM1’s effects on whole transcriptome in response to estrogen between two RNA-seq biological repeats (n=11,816). (G) Correlation of CARM1 effects on whole transcriptome in response to estrogen based on RNA- seq between two independent siRNAs targeting CARM1 (siCARM1 (1) and siCARM1 (2)) (n=12,234). (H, I) MCF7 cells as described in (G) were subjected to immunoblotting (IB) (H) and RT-qPCR (I) analysis to examine the levels of indicated protein and mRNA, respectively. Actin was served as a loading control. (J) MCF7 cells as described in Figure 1E were subjected to RT-qPCR analysis to examine the expression of genes as indicated. (K) MCF7 cells as described in Figure 1E were subjected to immunoblotting (IB) analysis to examine the protein levels of CARM1. Actin was served as a loading control. (L) Genomic DNA was extracted from CARM1 knockout cells, followed by PCR using specific primer sets spanning gRNA targeting region (boxed in blue). The resultant PCR products were subjected to Sanger sequencing as shown. -

Stromule Extension Along Microtubules Coordinated with Actin
RESEARCH ARTICLE Stromule extension along microtubules coordinated with actin-mediated anchoring guides perinuclear chloroplast movement during innate immunity Amutha Sampath Kumar1†, Eunsook Park2,3†‡, Alexander Nedo1,4, Ali Alqarni1,4,5, Li Ren5, Kyle Hoban1,4, Shannon Modla1, John H McDonald4, Chandra Kambhamettu5,6, Savithramma P Dinesh-Kumar2,3*, Jeffrey Lewis Caplan1,4,5* 1Delaware Biotechnology Institute, University of Delaware, Newark, United States; 2Department of Plant Biology, College of Biological Sciences, University of California, Davis, Davis, United States; 3The Genome Center, College of Biological Sciences, University of California, Davis, Davis, United States; 4Department of Biological Sciences, College of Arts and Sciences, University of Delaware, Newark, United States; 5Department of Plant and Soil Sciences, College of Agriculture and Natural Resources, University of Delaware, Newark, United States; 6Department of Computer and Information Sciences, College of Engineering, University of *For correspondence: Delaware, Newark, United States [email protected] (SPD-K); [email protected] (JLC) † These authors contributed Abstract Dynamic tubular extensions from chloroplasts called stromules have recently been equally to this work shown to connect with nuclei and function during innate immunity. We demonstrate that stromules Present address: ‡Department extend along microtubules (MTs) and MT organization directly affects stromule dynamics since of Plant Sciences, College of stabilization of MTs chemically or genetically -

CRISPR-Enhanced Human Adipocyte “Browning” As Cell Therapy for Metabolic Disease
bioRxiv preprint doi: https://doi.org/10.1101/2020.10.13.337923; this version posted October 13, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. CRISPR-enhanced human adipocyte “browning” as cell therapy for metabolic disease Emmanouela Tsagkaraki1,6*, Sarah Nicoloro1*, Tiffany De Souza1, Javier Solivan-Rivera1, Anand Desai1, Yuefei Shen1, Mark Kelly1, Adilson Guilherme1, Felipe Henriques1, Raed Ibraheim2, Nadia Amrani2, Kevin Luk4, Stacy Maitland4, Randall H. Friedline1, Lauren Tauer1, Xiaodi Hu1, Jason K. Kim1,3, Scot A. Wolfe4,5, Erik J. Sontheimer2,5, Silvia Corvera1**, Michael P. Czech1**. 1Program in Molecular Medicine, University of Massachusetts Medical School, Worcester, MA 01605 2RNA Therapeutics Institute, University of Massachusetts Medical School, Worcester MA 01605 3Division of Endocrinology, Metabolism and Diabetes, Department of Medicine, University of Massachusetts Medical School Worcester, MA 01605 4Department of Molecular,Cell and Cancer Biology, University of Massachusetts Medical School, Worcester MA 01605 5Li Weibo Institute for Rare Diseases Research, University of Massachusetts Medical School, Worcester, MA 01605 6University of Crete School of Medicine, Crete 71003 *These authors contributed equally **Co-corresponding authors Michael P. Czech 373 Plantation Street Program in Molecular Medicine University of Massachusetts Medical School Worcester, MA 01605 [email protected] Tel: 1 508 856 2254 Fax: 1 508 856 1617 Silvia Corvera 373 Plantation Street Program in Molecular Medicine University of Massachusetts Medical School Worcester, MA 01605 [email protected] Tel: 1 508 856 6869 Fax: 1 508 856 1617 bioRxiv preprint doi: https://doi.org/10.1101/2020.10.13.337923; this version posted October 13, 2020. -

Human Induced Pluripotent Stem Cell–Derived Podocytes Mature Into Vascularized Glomeruli Upon Experimental Transplantation
BASIC RESEARCH www.jasn.org Human Induced Pluripotent Stem Cell–Derived Podocytes Mature into Vascularized Glomeruli upon Experimental Transplantation † Sazia Sharmin,* Atsuhiro Taguchi,* Yusuke Kaku,* Yasuhiro Yoshimura,* Tomoko Ohmori,* ‡ † ‡ Tetsushi Sakuma, Masashi Mukoyama, Takashi Yamamoto, Hidetake Kurihara,§ and | Ryuichi Nishinakamura* *Department of Kidney Development, Institute of Molecular Embryology and Genetics, and †Department of Nephrology, Faculty of Life Sciences, Kumamoto University, Kumamoto, Japan; ‡Department of Mathematical and Life Sciences, Graduate School of Science, Hiroshima University, Hiroshima, Japan; §Division of Anatomy, Juntendo University School of Medicine, Tokyo, Japan; and |Japan Science and Technology Agency, CREST, Kumamoto, Japan ABSTRACT Glomerular podocytes express proteins, such as nephrin, that constitute the slit diaphragm, thereby contributing to the filtration process in the kidney. Glomerular development has been analyzed mainly in mice, whereas analysis of human kidney development has been minimal because of limited access to embryonic kidneys. We previously reported the induction of three-dimensional primordial glomeruli from human induced pluripotent stem (iPS) cells. Here, using transcription activator–like effector nuclease-mediated homologous recombination, we generated human iPS cell lines that express green fluorescent protein (GFP) in the NPHS1 locus, which encodes nephrin, and we show that GFP expression facilitated accurate visualization of nephrin-positive podocyte formation in -

Supplemental Digital Content (Sdc) Sdc, Materials
SUPPLEMENTAL DIGITAL CONTENT (SDC) SDC, MATERIALS AND METHODS Animals This study used 9-12 week old male C57BL/6 mice (Jackson Laboratory, Bar Harbor, ME). This study conformed to the National Institutes of Health guidelines and was conducted under animal protocols approved by the University of Virginia’s Institutional Animal Care and Use Committee. Murine DCD Lung Procedure Mice were anesthetized by isoflurane inhalation and euthanized by cervical dislocation followed by a 60-minute period of “no-touch” warm ischemia. Mice then underwent extended median sternotomy and midline cervical exposure followed by intubation for the initiation of mechanical ventilation at 120 strokes/minute with room air. The left atrium was vented via an atriotomy followed by infusion of the lungs with 3 mL 4°C Perfadex® solution (Vitrolife Inc., Denver, CO) supplemented with THAM Solution (Vitrolife, Kungsbacka, Sweden), estimating weight-based volume recommendations for pulmonary artery perfusion (140mL/kg) (1). The chest was then packed with ice and the trachea occluded by silk-suture tie at tidal volume (7µL/g body weight) prior to cold static preservation (CSP) for 60 minutes at 4°C. Mice were then randomized into three experimental groups: 1) CSP alone with no EVLP, 2) EVLP with Steen solution and 3) EVLP with Steen solution supplemented with the highly selective A2AR agonist, ATL1223 (30nM, Lewis and Clark Pharmaceuticals, Charlottesville, VA). Mice treated with ATL1223 during EVLP also received ATL1223 treatment (30nM) during the Perfadex flush prior to CSP whereas the EVLP group received vehicle (DMSO) during the flush. CSP lungs, which did not undergo EVLP, underwent immediate functional assessment after re-intubation as described below. -

PPM1G Promotes the Progression of Hepatocellular Carcinoma Via
www.nature.com/cddis ARTICLE OPEN PPM1G promotes the progression of hepatocellular carcinoma via phosphorylation regulation of alternative splicing protein SRSF3 ✉ ✉ Dawei Chen1, Zhenguo Zhao1, Lu Chen1, Qinghua Li1, Jixue Zou 2 and Shuanghai Liu 1 © The Author(s) 2021 Emerging evidence has demonstrated that alternative splicing has a vital role in regulating protein function, but how alternative splicing factors can be regulated remains unclear. We showed that the PPM1G, a protein phosphatase, regulated the phosphorylation of SRSF3 in hepatocellular carcinoma (HCC) and contributed to the proliferation, invasion, and metastasis of HCC. PPM1G was highly expressed in HCC tissues compared to adjacent normal tissues, and higher levels of PPM1G were observed in adverse staged HCCs. The higher levels of PPM1G were highly correlated with poor prognosis, which was further validated in the TCGA cohort. The knockdown of PPM1G inhibited the cell growth and invasion of HCC cell lines. Further studies showed that the knockdown of PPM1G inhibited tumor growth in vivo. The mechanistic analysis showed that the PPM1G interacted with proteins related to alternative splicing, including SRSF3. Overexpression of PPM1G promoted the dephosphorylation of SRSF3 and changed the alternative splicing patterns of genes related to the cell cycle, the transcriptional regulation in HCC cells. In addition, we also demonstrated that the promoter of PPM1G was activated by multiple transcription factors and co-activators, including MYC/MAX and EP300, MED1, and ELF1. Our study highlighted the essential role of PPM1G in HCC and shed new light on unveiling the regulation of alternative splicing in malignant transformation. Cell Death and Disease (2021) 12:722 ; https://doi.org/10.1038/s41419-021-04013-y INTRODUCTION The AR-V7 is specifically highly expressed in patients with relapse Hepatocellular carcinoma (HCC) is one of the most aggressive and drug resistance after targeted therapy. -

Ectopic Protein Interactions Within BRD4–Chromatin Complexes Drive Oncogenic Megadomain Formation in NUT Midline Carcinoma
Ectopic protein interactions within BRD4–chromatin complexes drive oncogenic megadomain formation in NUT midline carcinoma Artyom A. Alekseyenkoa,b,1, Erica M. Walshc,1, Barry M. Zeea,b, Tibor Pakozdid, Peter Hsic, Madeleine E. Lemieuxe, Paola Dal Cinc, Tan A. Incef,g,h,i, Peter V. Kharchenkod,j, Mitzi I. Kurodaa,b,2, and Christopher A. Frenchc,2 aDivision of Genetics, Department of Medicine, Brigham and Women’s Hospital, Harvard Medical School, Boston, MA 02115; bDepartment of Genetics, Harvard Medical School, Boston, MA 02115; cDepartment of Pathology, Brigham and Women’s Hospital, Harvard Medical School, Boston, MA 02115; dDepartment of Biomedical Informatics, Harvard Medical School, Boston, MA 02115; eBioinfo, Plantagenet, ON, Canada K0B 1L0; fDepartment of Pathology, University of Miami Miller School of Medicine, Miami, FL 33136; gBraman Family Breast Cancer Institute, University of Miami Miller School of Medicine, Miami, FL 33136; hInterdisciplinary Stem Cell Institute, University of Miami Miller School of Medicine, Miami, FL 33136; iSylvester Comprehensive Cancer Center, University of Miami Miller School of Medicine, Miami, FL 33136; and jHarvard Stem Cell Institute, Cambridge, MA 02138 Contributed by Mitzi I. Kuroda, April 6, 2017 (sent for review February 7, 2017; reviewed by Sharon Y. R. Dent and Jerry L. Workman) To investigate the mechanism that drives dramatic mistargeting of and, in the case of MYC, leads to differentiation in culture (2, 3). active chromatin in NUT midline carcinoma (NMC), we have Similarly, small-molecule BET inhibitors such as JQ1, which identified protein interactions unique to the BRD4–NUT fusion disengage BRD4–NUT from chromatin, diminish megadomain- oncoprotein compared with wild-type BRD4.