Transcriptional and Epigenetic Control of Brown and Beige Adipose Cell Fate and Function
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Chapter 7: Monogenic Forms of Diabetes
CHAPTER 7 MONOGENIC FORMS OF DIABETES Mark A. Sperling, MD, and Abhimanyu Garg, MD Dr. Mark A. Sperling is Emeritus Professor and Chair, University of Pittsburgh, Department of Pediatrics, Children’s Hospital of Pittsburgh of UPMC, Pittsburgh, PA. Dr. Abhimanyu Garg is Professor of Internal Medicine and Chief of the Division of Nutrition and Metabolic Diseases at University of Texas Southwestern Medical Center, Dallas, TX. SUMMARY Types 1 and 2 diabetes have multiple and complex genetic influences that interact with environmental triggers, such as viral infections or nutritional excesses, to result in their respective phenotypes: young, lean, and insulin-dependence for type 1 diabetes patients or older, overweight, and often manageable by lifestyle interventions and oral medications for type 2 diabetes patients. A small subset of patients, comprising ~2%–3% of all those diagnosed with diabetes, may have characteristics of either type 1 or type 2 diabetes but have single gene defects that interfere with insulin production, secretion, or action, resulting in clinical diabetes. These types of diabetes are known as MODY, originally defined as maturity-onset diabetes of youth, and severe early-onset forms, such as neonatal diabetes mellitus (NDM). Defects in genes involved in adipocyte development, differentiation, and death pathways cause lipodystrophy syndromes, which are also associated with insulin resistance and diabetes. Although these syndromes are considered rare, more awareness of these disorders and increased availability of genetic testing in clinical and research laboratories, as well as growing use of next generation, whole genome, or exome sequencing for clinically challenging phenotypes, are resulting in increased recognition. A correct diagnosis of MODY, NDM, or lipodystrophy syndromes has profound implications for treatment, genetic counseling, and prognosis. -

THE ROLE of HOMEODOMAIN TRANSCRIPTION FACTOR IRX5 in CARDIAC CONTRACTILITY and HYPERTROPHIC RESPONSE by © COPYRIGHT by KYOUNG H
THE ROLE OF HOMEODOMAIN TRANSCRIPTION FACTOR IRX5 IN CARDIAC CONTRACTILITY AND HYPERTROPHIC RESPONSE By KYOUNG HAN KIM A THESIS SUBMITTED IN CONFORMITY WITH THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY GRADUATE DEPARTMENT OF PHYSIOLOGY UNIVERSITY OF TORONTO © COPYRIGHT BY KYOUNG HAN KIM (2011) THE ROLE OF HOMEODOMAIN TRANSCRIPTION FACTOR IRX5 IN CARDIAC CONTRACTILITY AND HYPERTROPHIC RESPONSE KYOUNG HAN KIM DOCTOR OF PHILOSOPHY GRADUATE DEPARTMENT OF PHYSIOLOGY UNIVERSITY OF TORONTO 2011 ABSTRACT Irx5 is a homeodomain transcription factor that negatively regulates cardiac fast transient + outward K currents (Ito,f) via the KV4.2 gene and is thereby a major determinant of the transmural repolarization gradient. While Ito,f is invariably reduced in heart disease and changes in Ito,f can modulate both cardiac contractility and hypertrophy, less is known about a functional role of Irx5, and its relationship with Ito,f, in the normal and diseased heart. Here I show that Irx5 plays crucial roles in the regulation of cardiac contractility and proper adaptive hypertrophy. Specifically, Irx5-deficient (Irx5-/-) hearts had reduced cardiac contractility and lacked the normal regional difference in excitation-contraction with decreased action potential duration, Ca2+ transients and myocyte shortening in sub-endocardial, but not sub-epicardial, myocytes. In addition, Irx5-/- mice showed less cardiac hypertrophy, but increased interstitial fibrosis and greater contractility impairment following pressure overload. A defect in hypertrophic responses in Irx5-/- myocardium was confirmed in cultured neonatal mouse ventricular myocytes, exposed to norepinephrine while being restored with Irx5 replacement. Interestingly, studies using mice ii -/- virtually lacking Ito,f (i.e. KV4.2-deficient) showed that reduced contractility in Irx5 mice was completely restored by loss of KV4.2, whereas hypertrophic responses to pressure-overload in hearts remained impaired when both Irx5 and Ito,f were absent. -
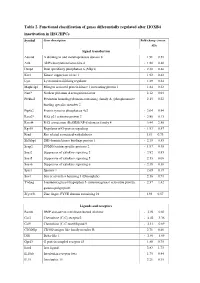
Table 2. Functional Classification of Genes Differentially Regulated After HOXB4 Inactivation in HSC/Hpcs
Table 2. Functional classification of genes differentially regulated after HOXB4 inactivation in HSC/HPCs Symbol Gene description Fold-change (mean ± SD) Signal transduction Adam8 A disintegrin and metalloprotease domain 8 1.91 ± 0.51 Arl4 ADP-ribosylation factor-like 4 - 1.80 ± 0.40 Dusp6 Dual specificity phosphatase 6 (Mkp3) - 2.30 ± 0.46 Ksr1 Kinase suppressor of ras 1 1.92 ± 0.42 Lyst Lysosomal trafficking regulator 1.89 ± 0.34 Mapk1ip1 Mitogen activated protein kinase 1 interacting protein 1 1.84 ± 0.22 Narf* Nuclear prelamin A recognition factor 2.12 ± 0.04 Plekha2 Pleckstrin homology domain-containing. family A. (phosphoinosite 2.15 ± 0.22 binding specific) member 2 Ptp4a2 Protein tyrosine phosphatase 4a2 - 2.04 ± 0.94 Rasa2* RAS p21 activator protein 2 - 2.80 ± 0.13 Rassf4 RAS association (RalGDS/AF-6) domain family 4 3.44 ± 2.56 Rgs18 Regulator of G-protein signaling - 1.93 ± 0.57 Rrad Ras-related associated with diabetes 1.81 ± 0.73 Sh3kbp1 SH3 domain kinase bindings protein 1 - 2.19 ± 0.53 Senp2 SUMO/sentrin specific protease 2 - 1.97 ± 0.49 Socs2 Suppressor of cytokine signaling 2 - 2.82 ± 0.85 Socs5 Suppressor of cytokine signaling 5 2.13 ± 0.08 Socs6 Suppressor of cytokine signaling 6 - 2.18 ± 0.38 Spry1 Sprouty 1 - 2.69 ± 0.19 Sos1 Son of sevenless homolog 1 (Drosophila) 2.16 ± 0.71 Ywhag 3-monooxygenase/tryptophan 5- monooxygenase activation protein. - 2.37 ± 1.42 gamma polypeptide Zfyve21 Zinc finger. FYVE domain containing 21 1.93 ± 0.57 Ligands and receptors Bambi BMP and activin membrane-bound inhibitor - 2.94 ± 0.62 -

Functional Roles of Bromodomain Proteins in Cancer
cancers Review Functional Roles of Bromodomain Proteins in Cancer Samuel P. Boyson 1,2, Cong Gao 3, Kathleen Quinn 2,3, Joseph Boyd 3, Hana Paculova 3 , Seth Frietze 3,4,* and Karen C. Glass 1,2,4,* 1 Department of Pharmaceutical Sciences, Albany College of Pharmacy and Health Sciences, Colchester, VT 05446, USA; [email protected] 2 Department of Pharmacology, Larner College of Medicine, University of Vermont, Burlington, VT 05405, USA; [email protected] 3 Department of Biomedical and Health Sciences, University of Vermont, Burlington, VT 05405, USA; [email protected] (C.G.); [email protected] (J.B.); [email protected] (H.P.) 4 University of Vermont Cancer Center, Burlington, VT 05405, USA * Correspondence: [email protected] (S.F.); [email protected] (K.C.G.) Simple Summary: This review provides an in depth analysis of the role of bromodomain-containing proteins in cancer development. As readers of acetylated lysine on nucleosomal histones, bromod- omain proteins are poised to activate gene expression, and often promote cancer progression. We examined changes in gene expression patterns that are observed in bromodomain-containing proteins and associated with specific cancer types. We also mapped the protein–protein interaction network for the human bromodomain-containing proteins, discuss the cellular roles of these epigenetic regu- lators as part of nine different functional groups, and identify bromodomain-specific mechanisms in cancer development. Lastly, we summarize emerging strategies to target bromodomain proteins in cancer therapy, including those that may be essential for overcoming resistance. Overall, this review provides a timely discussion of the different mechanisms of bromodomain-containing pro- Citation: Boyson, S.P.; Gao, C.; teins in cancer, and an updated assessment of their utility as a therapeutic target for a variety of Quinn, K.; Boyd, J.; Paculova, H.; cancer subtypes. -

Cross-Talk Between HER2 and MED1 Regulates Tamoxifen Resistance of Human Breast Cancer Cells
Published OnlineFirst September 10, 2012; DOI: 10.1158/0008-5472.CAN-12-1305 Cancer Tumor and Stem Cell Biology Research Cross-talk between HER2 and MED1 Regulates Tamoxifen Resistance of Human Breast Cancer Cells Jiajun Cui1, Katherine Germer1, Tianying Wu2, Jiang Wang3, Jia Luo5, Shao-chun Wang1, Qianben Wang4, and Xiaoting Zhang1 Abstract Despite the fact that most breast cancer patients have estrogen receptor (ER) a-positive tumors, up to 50% of the patients are or soon develop resistance to endocrine therapy. It is recognized that HER2 activation is one of the major mechanisms contributing to endocrine resistance. In this study, we report that the ER coactivator MED1 is a novel cross-talk point for the HER2 and ERa pathways. Tissue microarray analysis of human breast cancers revealed that MED1 expression positively correlates most strongly with HER2 status of the tumors. MED1 was highly phosphorylated, in a HER2-dependent manner, at the site known to be critical for its activation. Importantly, RNAi-mediated attenuation of MED1 sensitized HER2-overexpressing cells to tamoxifen treatment. MED1 and its phosphorylated form, but not the corepressors N-CoR and SMRT, were recruited to the ERa target gene promoter by tamoxifen in HER2-overexpressing cells. Significantly, MED1 attenuation or mutation of MED1 phosphorylation sites was sufficient to restore the promoter recruitment of N-CoR and SMRT. Notably, we found that MED1 is required for the expression of not only traditional E2-ERa target genes but also the newly described EGF-ERa target genes. Our results additionally indicated that MED1 is recruited to the HER2 gene and required for its expression. -

The Retinoblastoma Tumor-Suppressor Gene, the Exception That Proves the Rule
Oncogene (2006) 25, 5233–5243 & 2006 Nature Publishing Group All rights reserved 0950-9232/06 $30.00 www.nature.com/onc REVIEW The retinoblastoma tumor-suppressor gene, the exception that proves the rule DW Goodrich Department of Pharmacology & Therapeutics, Roswell Park Cancer Institute, Buffalo, NY, USA The retinoblastoma tumor-suppressor gene (Rb1)is transmission of one mutationally inactivated Rb1 allele centrally important in cancer research. Mutational and loss of the remaining wild-type allele in somatic inactivation of Rb1 causes the pediatric cancer retino- retinal cells. Hence hereditary retinoblastoma typically blastoma, while deregulation ofthe pathway in which it has an earlier onset and a greater number of tumor foci functions is common in most types of human cancer. The than sporadic retinoblastoma where both Rb1 alleles Rb1-encoded protein (pRb) is well known as a general cell must be inactivated in somatic retinal cells. To this day, cycle regulator, and this activity is critical for pRb- Rb1 remains an exception among cancer-associated mediated tumor suppression. The main focus of this genes in that its mutation is apparently both necessary review, however, is on more recent evidence demonstrating and sufficient, or at least rate limiting, for the genesis of the existence ofadditional, cell type-specific pRb func- a human cancer. The simple genetics of retinoblastoma tions in cellular differentiation and survival. These has spawned the hope that a complete molecular additional functions are relevant to carcinogenesis sug- understanding of the Rb1-encoded protein (pRb) would gesting that the net effect of Rb1 loss on the behavior of lead to deeper insight into the processes of neoplastic resulting tumors is highly dependent on biological context. -

Cisplatin and Phenanthriplatin Modulate Long-Noncoding
www.nature.com/scientificreports OPEN Cisplatin and phenanthriplatin modulate long‑noncoding RNA expression in A549 and IMR90 cells revealing regulation of microRNAs, Wnt/β‑catenin and TGF‑β signaling Jerry D. Monroe1,2, Satya A. Moolani2,3, Elvin N. Irihamye2,4, Katheryn E. Lett1, Michael D. Hebert1, Yann Gibert1* & Michael E. Smith2* The monofunctional platinum(II) complex, phenanthriplatin, acts by blocking transcription, but its regulatory efects on long‑noncoding RNAs (lncRNAs) have not been elucidated relative to traditional platinum‑based chemotherapeutics, e.g., cisplatin. Here, we treated A549 non‑small cell lung cancer and IMR90 lung fbroblast cells for 24 h with either cisplatin, phenanthriplatin or a solvent control, and then performed microarray analysis to identify regulated lncRNAs. RNA22 v2 microRNA software was subsequently used to identify microRNAs (miRNAs) that might be suppressed by the most regulated lncRNAs. We found that miR‑25‑5p, ‑30a‑3p, ‑138‑5p, ‑149‑3p, ‑185‑5p, ‑378j, ‑608, ‑650, ‑708‑5p, ‑1253, ‑1254, ‑4458, and ‑4516, were predicted to target the cisplatin upregulated lncRNAs, IMMP2L‑1, CBR3‑1 and ATAD2B‑5, and the phenanthriplatin downregulated lncRNAs, AGO2‑1, COX7A1‑2 and SLC26A3‑1. Then, we used qRT‑PCR to measure the expression of miR‑25‑5p, ‑378j, ‑4516 (A549) and miR‑149‑3p, ‑608, and ‑4458 (IMR90) to identify distinct signaling efects associated with cisplatin and phenanthriplatin. The signaling pathways associated with these miRNAs suggests that phenanthriplatin may modulate Wnt/β‑catenin and TGF‑β signaling through the MAPK/ ERK and PTEN/AKT pathways diferently than cisplatin. Further, as some of these miRNAs may be subject to dissimilar lncRNA targeting in A549 and IMR90 cells, the monofunctional complex may not cause toxicity in normal lung compared to cancer cells by acting through distinct lncRNA and miRNA networks. -

Key Roles for MED1 Lxxll Motifs in Pubertal Mammary Gland Development and Luminal-Cell Differentiation
Key roles for MED1 LxxLL motifs in pubertal mammary gland development and luminal-cell differentiation Pingping Jianga,b,1, Qiuping Hua,1, Mitsuhiro Itoc,1,3, Sara Meyera, Susan Waltza, Sohaib Khana, Robert G. Roederc,2, and Xiaoting Zhanga,2 aDepartment of Cancer and Cell Biology, College of Medicine, University of Cincinnati, 3125 Eden Avenue, OH 45267; bCollege of Life Sciences, Zhejiang University, 388 Yuhangtang Road, Hangzhou 310058, China; and cLaboratory of Biochemistry and Molecular Biology, The Rockefeller University, 1230 York Avenue, New York, NY 10065 Contributed by Robert G. Roeder, February 16, 2010 (sent for review January 18, 2010) Mediator recently has emerged as a central player in the direct (7–9). In general, individual Mediator subunits interact transduction of signals from transcription factors to the general specifically with their corresponding transcription factors and de- transcriptional machinery. In the case of nuclear receptors, in vitro letions of these Mediator subunits often affect expression primar- studies have shown that the transcriptional coactivator function of ily of target genes and pathways controlled by their corresponding the Mediator involves direct ligand-dependent interactions of the transcription factor(s) (9). In the case of nuclear receptors, the MED1 subunit, through its two classical LxxLL motifs, with the re- Mediator interactions are ligand- and AF-2-dependent and ceptor AF2 domain. However, despite the strong in vitro evidence, mediated through the LxxLL motifs in the MED1 (a.k.a. there currently is little information regarding in vivo functions of TRAP220/PBP/DRIP205) subunit (10–13). Importantly, in rela- the LxxLL motifs either in MED1 or in other coactivators. -

A Pathogenic Ctbp1 Missense Mutation Causes Altered Cofactor Binding and Transcriptional Activity
neurogenetics (2019) 20:129–143 https://doi.org/10.1007/s10048-019-00578-1 ORIGINAL ARTICLE A pathogenic CtBP1 missense mutation causes altered cofactor binding and transcriptional activity David B. Beck1 & T. Subramanian2 & S. Vijayalingam2 & Uthayashankar R. Ezekiel3 & Sandra Donkervoort4 & Michele L. Yang5 & Holly A. Dubbs6 & Xilma R. Ortiz-Gonzalez7 & Shenela Lakhani8 & Devorah Segal9 & Margaret Au10 & John M. Graham Jr10 & Sumit Verma11 & Darrel Waggoner12 & Marwan Shinawi13 & Carsten G. Bönnemann4 & Wendy K. Chung14 & G. Chinnadurai2 Received: 23 October 2018 /Revised: 18 March 2019 /Accepted: 9 April 2019 /Published online: 30 April 2019 # Springer-Verlag GmbH Germany, part of Springer Nature 2019 Abstract We previously reported a pathogenic de novo p.R342W mutation in the transcriptional corepressor CTBP1 in four independent patients with neurodevelopmental disabilities [1]. Here, we report the clinical phenotypes of seven additional individuals with the same recurrent de novo CTBP1 mutation. Within this cohort, we identified consistent CtBP1-related phenotypes of intellectual disability, ataxia, hypotonia, and tooth enamel defects present in most patients. The R342W mutation in CtBP1 is located within a region implicated in a high affinity-binding cleft for CtBP-interacting proteins. Unbiased proteomic analysis demonstrated reduced interaction of several chromatin-modifying factors with the CtBP1 W342 mutant. Genome-wide transcriptome analysis in human glioblastoma cell lines expressing -CtBP1 R342 (wt) or W342 mutation revealed changes in the expression profiles of genes controlling multiple cellular processes. Patient-derived dermal fibroblasts were found to be more sensitive to apoptosis during acute glucose deprivation compared to controls. Glucose deprivation strongly activated the BH3-only pro-apoptotic gene NOXA, suggesting a link between enhanced cell death and NOXA expression in patient fibroblasts. -

Table S1 the Four Gene Sets Derived from Gene Expression Profiles of Escs and Differentiated Cells
Table S1 The four gene sets derived from gene expression profiles of ESCs and differentiated cells Uniform High Uniform Low ES Up ES Down EntrezID GeneSymbol EntrezID GeneSymbol EntrezID GeneSymbol EntrezID GeneSymbol 269261 Rpl12 11354 Abpa 68239 Krt42 15132 Hbb-bh1 67891 Rpl4 11537 Cfd 26380 Esrrb 15126 Hba-x 55949 Eef1b2 11698 Ambn 73703 Dppa2 15111 Hand2 18148 Npm1 11730 Ang3 67374 Jam2 65255 Asb4 67427 Rps20 11731 Ang2 22702 Zfp42 17292 Mesp1 15481 Hspa8 11807 Apoa2 58865 Tdh 19737 Rgs5 100041686 LOC100041686 11814 Apoc3 26388 Ifi202b 225518 Prdm6 11983 Atpif1 11945 Atp4b 11614 Nr0b1 20378 Frzb 19241 Tmsb4x 12007 Azgp1 76815 Calcoco2 12767 Cxcr4 20116 Rps8 12044 Bcl2a1a 219132 D14Ertd668e 103889 Hoxb2 20103 Rps5 12047 Bcl2a1d 381411 Gm1967 17701 Msx1 14694 Gnb2l1 12049 Bcl2l10 20899 Stra8 23796 Aplnr 19941 Rpl26 12096 Bglap1 78625 1700061G19Rik 12627 Cfc1 12070 Ngfrap1 12097 Bglap2 21816 Tgm1 12622 Cer1 19989 Rpl7 12267 C3ar1 67405 Nts 21385 Tbx2 19896 Rpl10a 12279 C9 435337 EG435337 56720 Tdo2 20044 Rps14 12391 Cav3 545913 Zscan4d 16869 Lhx1 19175 Psmb6 12409 Cbr2 244448 Triml1 22253 Unc5c 22627 Ywhae 12477 Ctla4 69134 2200001I15Rik 14174 Fgf3 19951 Rpl32 12523 Cd84 66065 Hsd17b14 16542 Kdr 66152 1110020P15Rik 12524 Cd86 81879 Tcfcp2l1 15122 Hba-a1 66489 Rpl35 12640 Cga 17907 Mylpf 15414 Hoxb6 15519 Hsp90aa1 12642 Ch25h 26424 Nr5a2 210530 Leprel1 66483 Rpl36al 12655 Chi3l3 83560 Tex14 12338 Capn6 27370 Rps26 12796 Camp 17450 Morc1 20671 Sox17 66576 Uqcrh 12869 Cox8b 79455 Pdcl2 20613 Snai1 22154 Tubb5 12959 Cryba4 231821 Centa1 17897 -

Modes of Interaction of KMT2 Histone H3 Lysine 4 Methyltransferase/COMPASS Complexes with Chromatin
cells Review Modes of Interaction of KMT2 Histone H3 Lysine 4 Methyltransferase/COMPASS Complexes with Chromatin Agnieszka Bochy ´nska,Juliane Lüscher-Firzlaff and Bernhard Lüscher * ID Institute of Biochemistry and Molecular Biology, Medical School, RWTH Aachen University, Pauwelsstrasse 30, 52057 Aachen, Germany; [email protected] (A.B.); jluescher-fi[email protected] (J.L.-F.) * Correspondence: [email protected]; Tel.: +49-241-8088850; Fax: +49-241-8082427 Received: 18 January 2018; Accepted: 27 February 2018; Published: 2 March 2018 Abstract: Regulation of gene expression is achieved by sequence-specific transcriptional regulators, which convey the information that is contained in the sequence of DNA into RNA polymerase activity. This is achieved by the recruitment of transcriptional co-factors. One of the consequences of co-factor recruitment is the control of specific properties of nucleosomes, the basic units of chromatin, and their protein components, the core histones. The main principles are to regulate the position and the characteristics of nucleosomes. The latter includes modulating the composition of core histones and their variants that are integrated into nucleosomes, and the post-translational modification of these histones referred to as histone marks. One of these marks is the methylation of lysine 4 of the core histone H3 (H3K4). While mono-methylation of H3K4 (H3K4me1) is located preferentially at active enhancers, tri-methylation (H3K4me3) is a mark found at open and potentially active promoters. Thus, H3K4 methylation is typically associated with gene transcription. The class 2 lysine methyltransferases (KMTs) are the main enzymes that methylate H3K4. KMT2 enzymes function in complexes that contain a necessary core complex composed of WDR5, RBBP5, ASH2L, and DPY30, the so-called WRAD complex. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated.