Department of Mental Health Parameters 3.8 for Use of Psychotropic Medication in Children and Adolescents
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

HALDOL Brand of Haloperidol Injection (For Immediate Release) WARNING Increased Mortality in Elderly Patients with Dementia
HALDOL® brand of haloperidol injection (For Immediate Release) WARNING Increased Mortality in Elderly Patients with Dementia-Related Psychosis Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of seventeen placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. HALDOL Injection is not approved for the treatment of patients with dementia-related psychosis (see WARNINGS). DESCRIPTION Haloperidol is the first of the butyrophenone series of major antipsychotics. The chemical designation is 4-[4-(p-chlorophenyl)-4-hydroxypiperidino] 4’-fluorobutyrophenone and it has the following structural formula: HALDOL (haloperidol) is available as a sterile parenteral form for intramuscular injection. The injection provides 5 mg haloperidol (as the lactate) and lactic acid for pH adjustment between 3.0 – 3.6. -

Pindolol of the Activation of Postsynaptic 5-HT1A Receptors
Potentiation by (-)Pindolol of the Activation of Postsynaptic 5-HT1A Receptors Induced by Venlafaxine Jean-Claude Béïque, Ph.D., Pierre Blier, M.D., Ph.D., Claude de Montigny, M.D., Ph.D., and Guy Debonnel, M.D. The increase of extracellular 5-HT in brain terminal regions antagonist WAY 100635 (100 g/kg, i.v.). A short-term produced by the acute administration of 5-HT reuptake treatment with VLX (20 mg/kg/day ϫ 2 days) resulted in a inhibitors (SSRI’s) is hampered by the activation of ca. 90% suppression of the firing activity of 5-HT neurons somatodendritic 5-HT1A autoreceptors in the raphe nuclei. in the dorsal raphe nucleus. This was prevented by the The present in vivo electrophysiological studies were coadministration of (-)pindolol (15 mg/kg/day ϫ 2 days). undertaken, in the rat, to assess the effects of the Taken together, these results indicate that (-)pindolol coadministration of venlafaxine, a dual 5-HT/NE reuptake potentiated the activation of postsynaptic 5-HT1A receptors inhibitor, and (-)pindolol on pre- and postsynaptic 5-HT1A resulting from 5-HT reuptake inhibition probably by receptor function. The acute administration of venlafaxine blocking the somatodendritic 5-HT1A autoreceptor, but not and of the SSRI paroxetine (5 mg/kg, i.v.) induced a its postsynaptic congener. These results support and extend suppression of the firing activity of dorsal hippocampus CA3 previous findings providing a biological substratum for the pyramidal neurons. This effect of venlafaxine was markedly efficacy of pindolol as an accelerating strategy in major potentiated by a pretreatment with (-)pindolol (15 mg/kg, depression. -
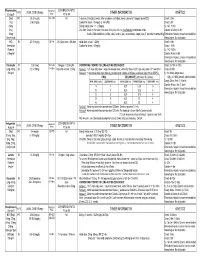
Medication Conversion Chart
Fluphenazine FREQUENCY CONVERSION RATIO ROUTE USUAL DOSE (Range) (Range) OTHER INFORMATION KINETICS Prolixin® PO to IM Oral PO 2.5-20 mg/dy QD - QID NA ↑ dose by 2.5mg/dy Q week. After symptoms controlled, slowly ↓ dose to 1-5mg/dy (dosed QD) Onset: ≤ 1hr 1mg (2-60 mg/dy) Caution for doses > 20mg/dy (↑ risk EPS) Cmax: 0.5hr 2.5mg Elderly: Initial dose = 1 - 2.5mg/dy t½: 14.7-15.3hr 5mg Oral Soln: Dilute in 2oz water, tomato or fruit juice, milk, or uncaffeinated carbonated drinks Duration of Action: 6-8hr 10mg Avoid caffeinated drinks (coffee, cola), tannics (tea), or pectinates (apple juice) 2° possible incompatibilityElimination: Hepatic to inactive metabolites 5mg/ml soln Hemodialysis: Not dialyzable HCl IM 2.5-10 mg/dy Q6-8 hr 1/3-1/2 po dose = IM dose Initial dose (usual): 1.25mg Onset: ≤ 1hr Immediate Caution for doses > 10mg/dy Cmax: 1.5-2hr Release t½: 14.7-15.3hr 2.5mg/ml Duration Action: 6-8hr Elimination: Hepatic to inactive metabolites Hemodialysis: Not dialyzable Decanoate IM 12.5-50mg Q2-3 wks 10mg po = 12.5mg IM CONVERTING FROM PO TO LONG-ACTING DECANOATE: Onset: 24-72hr (4-72hr) Long-Acting SC (12.5-100mg) (1-4 wks) Round to nearest 12.5mg Method 1: 1.25 X po daily dose = equiv decanoate dose; admin Q2-3wks. Cont ½ po daily dose X 1st few mths Cmax: 48-96hr 25mg/ml Method 2: ↑ decanoate dose over 4wks & ↓ po dose over 4-8wks as follows (accelerate taper for sx of EPS): t½: 6.8-9.6dy (single dose) ORAL DECANOATE (Administer Q 2 weeks) 15dy (14-100dy chronic administration) ORAL DOSE (mg/dy) ↓ DOSE OVER (wks) INITIAL DOSE (mg) TARGET DOSE (mg) DOSE OVER (wks) Steady State: 2mth (1.5-3mth) 5 4 6.25 6.25 0 Duration Action: 2wk (1-6wk) Elimination: Hepatic to inactive metabolites 10 4 6.25 12.5 4 Hemodialysis: Not dialyzable 20 8 6.25 12.5 4 30 8 6.25 25 4 40 8 6.25 25 4 Method 3: Admin equivalent decanoate dose Q2-3wks. -

Schizophrenia Care Guide
August 2015 CCHCS/DHCS Care Guide: Schizophrenia SUMMARY DECISION SUPPORT PATIENT EDUCATION/SELF MANAGEMENT GOALS ALERTS Minimize frequency and severity of psychotic episodes Suicidal ideation or gestures Encourage medication adherence Abnormal movements Manage medication side effects Delusions Monitor as clinically appropriate Neuroleptic Malignant Syndrome Danger to self or others DIAGNOSTIC CRITERIA/EVALUATION (PER DSM V) 1. Rule out delirium or other medical illnesses mimicking schizophrenia (see page 5), medications or drugs of abuse causing psychosis (see page 6), other mental illness causes of psychosis, e.g., Bipolar Mania or Depression, Major Depression, PTSD, borderline personality disorder (see page 4). Ideas in patients (even odd ideas) that we disagree with can be learned and are therefore not necessarily signs of schizophrenia. Schizophrenia is a world-wide phenomenon that can occur in cultures with widely differing ideas. 2. Diagnosis is made based on the following: (Criteria A and B must be met) A. Two of the following symptoms/signs must be present over much of at least one month (unless treated), with a significant impact on social or occupational functioning, over at least a 6-month period of time: Delusions, Hallucinations, Disorganized Speech, Negative symptoms (social withdrawal, poverty of thought, etc.), severely disorganized or catatonic behavior. B. At least one of the symptoms/signs should be Delusions, Hallucinations, or Disorganized Speech. TREATMENT OPTIONS MEDICATIONS Informed consent for psychotropic -

THE USE of MIRTAZAPINE AS a HYPNOTIC O Uso Da Mirtazapina Como Hipnótico Francisca Magalhães Scoralicka, Einstein Francisco Camargosa, Otávio Toledo Nóbregaa
ARTIGO ESPECIAL THE USE OF MIRTAZAPINE AS A HYPNOTIC O uso da mirtazapina como hipnótico Francisca Magalhães Scoralicka, Einstein Francisco Camargosa, Otávio Toledo Nóbregaa Prescription of approved hypnotics for insomnia decreased by more than 50%, whereas of antidepressive agents outstripped that of hypnotics. However, there is little data on their efficacy to treat insomnia, and many of these medications may be associated with known side effects. Antidepressants are associated with various effects on sleep patterns, depending on the intrinsic pharmacological properties of the active agent, such as degree of inhibition of serotonin or noradrenaline reuptake, effects on 5-HT1A and 5-HT2 receptors, action(s) at alpha-adrenoceptors, and/or histamine H1 sites. Mirtazapine is a noradrenergic and specific serotonergic antidepressive agent that acts by antagonizing alpha-2 adrenergic receptors and blocking 5-HT2 and 5-HT3 receptors. It has high affinity for histamine H1 receptors, low affinity for dopaminergic receptors, and lacks anticholinergic activity. In spite of these potential beneficial effects of mirtazapine on sleep, no placebo-controlled randomized clinical trials of ABSTRACT mirtazapine in primary insomniacs have been conducted. Mirtazapine was associated with improvements in sleep on normal sleepers and depressed patients. The most common side effects of mirtazapine, i.e. dry mouth, drowsiness, increased appetite and increased body weight, were mostly mild and transient. Considering its use in elderly people, this paper provides a revision about studies regarding mirtazapine for sleep disorders. KEYWORDS: sleep; antidepressive agents; sleep disorders; treatment� A prescrição de hipnóticos aprovados para insônia diminuiu em mais de 50%, enquanto de antidepressivos ultrapassou a dos primeiros. -

Does Curcumin Or Pindolol Potentiate Fluoxetine's Antidepressant Effect
Research Paper Does Curcumin or Pindolol Potentiate Fluoxetine’s Antidepressant Effect by a Pharmacokinetic or Pharmacodynamic Interaction? H. A. S. MURAD*, M. I. SULIAMAN1, H. ABDALLAH2 AND MAY ABDULSATTAR1 Department of Pharmacology, Faculty of Medicine, Rabigh, King Abdulaziz University (KAU), Jeddah-21589, Saudi Arabia (SA) and Faculty of Medicine, Ain Shams University, Cairo-11541, Egypt. 1Department of Pharmacology, Faculty of Medicine, KAU, Jeddah-21589, SA. 2Department of Natural Products, Faculty of Pharmacy, KAU, Jeddah-21589, SA and Department of Pharmacognosy, Faculty of Pharmacy, Cairo University, Cairo-11541, Egypt Murad, et al.: Potentiation of Fluoxetine by Curcumin This study was designed to study potentiation of fluoxetine’s antidepressant effect by curcumin or pindolol. Twenty eight groups of mice (n=8) were used in three sets of experiments. In the first set, 9 groups were subjected to the forced swimming test after being treated intraperitoneally with three vehicles, fluoxetine (5 and 20 mg/kg), curcumin (20 mg/kg), pindolol (32 mg/kg), curcumin+fluoxetine (5 mg/kg) and pindolol+fluoxetine (5 mg/kg). One hour after the test, serum and brain fluoxetine and norfluoxetine levels were measured in mice receiving fluoxetine (5 and 20 mg/kg), curcumin+fluoxetine (5 mg/kg) and pindolol+fluoxetine (5 mg/kg). In the second set, the test was done after pretreatment with p-chlorophenylalanine. In the third set, the locomotor activity was measured. The immobility duration was significantly decreased in fluoxetine (20 mg/kg), curcumin (20 mg/kg), curcumin+fluoxetine (5 mg/kg) and pindolol+fluoxetine (5 mg/kg) groups. These decreases were reversed with p-chlorophenylalanine. -

Serotonin and Drug-Induced Therapeutic Responses in Major Depression, Obsessive–Compulsive and Panic Disorders Pierre Blier, M.D., Ph.D
Serotonin and Drug-Induced Therapeutic Responses in Major Depression, Obsessive–Compulsive and Panic Disorders Pierre Blier, M.D., Ph.D. and Claude de Montigny, M.D., Ph.D. The therapeutic effectiveness of antidepressant drugs in major development of treatment strategies producing greater efficacy depression was discovered by pure serendipity. It took over 20 and more rapid onset of antidepressant action; that, is lithium years before the neurobiological modifications that could addition and pindolol combination, respectively. It is expected mediate the antidepressive response were put into evidence. that the better understanding recently obtained of the Indeed, whereas the immediate biochemical effects of these mechanism of action of certain antidepressant drugs in drugs had been well documented, their antidepressant action obsessive– compulsive and panic disorders will also lead to generally does not become apparent before 2 to 3 weeks of more effective treatment strategies for those disorders. treatment. The different classes of antidepressant [Neuropsychopharmacology 21:91S–98S, 1999] treatments were subsequently shown to enhance serotonin © 1999 American College of Neuropsychopharmacology. neurotransmission albeit via different pre- and postsynaptic Published by Elsevier Science Inc. mechanisms. Clinical trials based on this hypothesis led to the KEY WORDS: Antidepressant; Electroconvulsive shocks; electroconvulsive shock treatment (ECT), which was Lithium; Pindolol; Hippocampus; Orbito-frontal cortex generally given only to hospitalized patients. It took several years after the demonstration of the In the late 1950s, the therapeutic effect of certain drugs antidepressant effect of iproniazide and imipramine to in major depression was discovered by serendipity. An understand that these drugs could interfere with the ca- antidepressant response was observed in patients with tabolism of monoamines. -

NORPRAMIN® (Desipramine Hydrochloride Tablets USP)
NORPRAMIN® (desipramine hydrochloride tablets USP) Suicidality and Antidepressant Drugs Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of NORPRAMIN or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. NORPRAMIN is not approved for use in pediatric patients. (See WARNINGS: Clinical Worsening and Suicide Risk, PRECAUTIONS: Information for Patients, and PRECAUTIONS: Pediatric Use.) DESCRIPTION NORPRAMIN® (desipramine hydrochloride USP) is an antidepressant drug of the tricyclic type, and is chemically: 5H-Dibenz[bƒ]azepine-5-propanamine,10,11-dihydro-N-methyl-, monohydrochloride. 1 Reference ID: 3536021 Inactive Ingredients The following inactive ingredients are contained in all dosage strengths: acacia, calcium carbonate, corn starch, D&C Red No. 30 and D&C Yellow No. 10 (except 10 mg and 150 mg), FD&C Blue No. 1 (except 25 mg, 75 mg, and 100 mg), hydrogenated soy oil, iron oxide, light mineral oil, magnesium stearate, mannitol, polyethylene glycol 8000, pregelatinized corn starch, sodium benzoate (except 150 mg), sucrose, talc, titanium dioxide, and other ingredients. -

Guidance on the Use of Mood Stabilizers for the Treatment of Bipolar Affective Disorder Version 2
Guidance on the use of mood stabilizers for the treatment of bipolar affective disorder Version 2 RATIFYING COMMITTEE DRUGS AND THERAPEUTICS GROUP DATE RATIFIED July 2015 REPLACES Version 1 dated July 2013 NEXT REVIEW DATE July 2017 POLICY AUTHORS Jules Haste, Lead Pharmacist, Brighton and Hove Members of the Pharmacy Team (contributors are listed overleaf) If you require this document in an alternative format, i.e. easy read, large text, audio or Braille please contact the pharmacy team on 01243 623349 Page 1 of 49 Contributors Jed Hewitt, Chief Pharmacist - Governance & Professional Practice James Atkinson, Pharmacist Team Leader Mental Health and Community Services Miguel Gomez, Lead Pharmacist, Worthing. Hilary Garforth, Lead Pharmacist, Chichester. Pauline Daw, Lead Pharmacist (CRHTs & AOT), East Sussex. Iftekhar Khan, Lead Pharmacist (S&F Service), East Sussex. Graham Brown, Lead Pharmacist CAMHS & EIS. Gus Fernandez, Specialist Pharmacist MI and MH Lisa Stanton, Specialist Pharmacist Early Intervention Services & Learning Disabilities. Nana Tomova, Specialist Pharmacist, Crawley. Page 2 of 49 Section Title Page Number Introduction and Key Points 4 1. General principles in the treatment of acute mania 6 2. General principles in the treatment of bipolar depression 8 3. General principles in long term treatment 10 4. Rapid cycling 13 5. Physical health 13 6. Treatment in special situations 6.1 Pregnancy 15 6.2 Breast-feeding 17 6.3 Older adults 19 6.4 Children and adolescents 22 6.5 Learning disabilities 29 6.6 Cardiac dysfunction 30 6.7 Renal dysfunction 34 6.8 Hepatic dysfunction 37 6.9 Epilepsy 41 7. The risk of switching to mania with antidepressants 43 8. -

A Drug Repositioning Approach Identifies Tricyclic Antidepressants As Inhibitors of Small Cell Lung Cancer and Other Neuroendocrine Tumors
Published OnlineFirst September 26, 2013; DOI: 10.1158/2159-8290.CD-13-0183 RESEARCH ARTICLE A Drug Repositioning Approach Identifi es Tricyclic Antidepressants as Inhibitors of Small Cell Lung Cancer and Other Neuroendocrine Tumors Nadine S. Jahchan 1 , 2 , Joel T. Dudley 1 , Pawel K. Mazur 1 , 2 , Natasha Flores 1 , 2 , Dian Yang 1 , 2 , Alec Palmerton 1 , 2 , Anne-Flore Zmoos 1 , 2 , Dedeepya Vaka 1 , 2 , Kim Q.T. Tran 1 , 2 , Margaret Zhou 1 , 2 , Karolina Krasinska 3 , Jonathan W. Riess 4 , Joel W. Neal 5 , Purvesh Khatri 1 , 2 , Kwon S. Park 1 , 2 , Atul J. Butte 1 , 2 , and Julien Sage 1 , 2 Downloaded from cancerdiscovery.aacrjournals.org on September 29, 2021. © 2013 American Association for Cancer Research. Published OnlineFirst September 26, 2013; DOI: 10.1158/2159-8290.CD-13-0183 ABSTRACT Small cell lung cancer (SCLC) is an aggressive neuroendocrine subtype of lung cancer with high mortality. We used a systematic drug repositioning bioinformat- ics approach querying a large compendium of gene expression profi les to identify candidate U.S. Food and Drug Administration (FDA)–approved drugs to treat SCLC. We found that tricyclic antidepressants and related molecules potently induce apoptosis in both chemonaïve and chemoresistant SCLC cells in culture, in mouse and human SCLC tumors transplanted into immunocompromised mice, and in endog- enous tumors from a mouse model for human SCLC. The candidate drugs activate stress pathways and induce cell death in SCLC cells, at least in part by disrupting autocrine survival signals involving neurotransmitters and their G protein–coupled receptors. The candidate drugs inhibit the growth of other neuroendocrine tumors, including pancreatic neuroendocrine tumors and Merkel cell carcinoma. -

Repeated Lysergic Acid Diethylamide in an Animal Model of Depression
JOP0010.1177/0269881114531666Journal of PsychopharmacologyBuchborn et al. 531666research-article2014 Original Paper Repeated lysergic acid diethylamide in an animal model of depression: Normalisation of learning behaviour and Journal of Psychopharmacology 2014, Vol. 28(6) 545 –552 hippocampal serotonin 5-HT signalling © The Author(s) 2014 2 Reprints and permissions: sagepub.co.uk/journalsPermissions.nav DOI: 10.1177/0269881114531666 jop.sagepub.com Tobias Buchborn, Helmut Schröder, Volker Höllt and Gisela Grecksch Abstract A re-balance of postsynaptic serotonin (5-HT) receptor signalling, with an increase in 5-HT1A and a decrease in 5-HT2A signalling, is a final common pathway multiple antidepressants share. Given that the 5-HT1A/2A agonist lysergic acid diethylamide (LSD), when repeatedly applied, selectively downregulates 5-HT2A, but not 5-HT1A receptors, one might expect LSD to similarly re-balance the postsynaptic 5-HT signalling. Challenging this idea, we use an animal model of depression specifically responding to repeated antidepressant treatment (olfactory bulbectomy), and test the antidepressant-like properties of repeated LSD treatment (0.13 mg/kg/d, 11 d). In line with former findings, we observe that bulbectomised rats show marked deficits in active avoidance learning. These deficits, similarly as we earlier noted with imipramine, are largely reversed by repeated LSD administration. Additionally, bulbectomised rats exhibit distinct anomalies of monoamine receptor signalling in hippocampus and/or frontal cortex; 35 from these, only the hippocampal decrease in 5-HT2 related [ S]-GTP-gamma-S binding is normalised by LSD. Importantly, the sham-operated rats do not profit from LSD, and exhibit reduced hippocampal 5-HT2 signalling. As behavioural deficits after bulbectomy respond to agents classified as antidepressants only, we conclude that the effect of LSD in this model can be considered antidepressant-like, and discuss it in terms of a re-balance of hippocampal 5-HT2/5-HT1A signalling. -

Medication: Trazodone (Desyrel) 50 Mg
Trazodone COMPLEX CHRONIC DISEASES PROGRAM Medication Handout Date: May 15, 2018 Medication: Trazodone 50 mg What is trazodone: Trazodone is an antidepressant that is now used for insomnia; it helps with both falling asleep and staying asleep. Expected Benefit: As a sleep aid, you should notice a benefit on the first night within about 30 minutes of taking the medication. Watch for possible side effects: This list of side effects is important for you to be aware of however, it is also important to remember that not all side effects happen to everyone. If you have problems with these side effects talk with your doctor or pharmacist: Hangover effect (drowsiness that continues after waking up in the morning) Dizziness Dry mouth Headache Nausea Stopping the medication: Stopping trazodone is not usually a problem as there is no withdrawal effect you are taking it regularly at higher doses than prescribed below. Please ask your doctor or pharmacist before stopping the medication. Rebound insomnia is not usually a problem which makes trazodone a good option for taking a sleeping aid as needed How to use this medication: Take this medication with or without food Dosing Schedule: Start with 12.5 mg (¼ tablet) or 25 mg (½ tablet) at bedtime Increase the dose by ¼ or ½ a tablet every night until: o You can fall asleep, and/or o Stay asleep The usual effective dose is 50 – 150 mg at bedtime (1 – 3 tablets) Do not continue increasing the dose if you experience a hangover effect Talk with your doctor if you are still having problems with sleep, as you may need a different medication.