The Vocal Organ of Hummingbirds Shows Convergence with Songbirds Tobias Riede & Christopher R
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
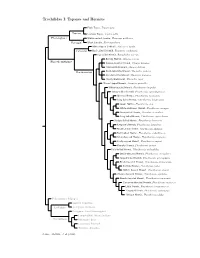
Topazes and Hermits
Trochilidae I: Topazes and Hermits Fiery Topaz, Topaza pyra Topazini Crimson Topaz, Topaza pella Florisuginae White-necked Jacobin, Florisuga mellivora Florisugini Black Jacobin, Florisuga fusca White-tipped Sicklebill, Eutoxeres aquila Eutoxerini Buff-tailed Sicklebill, Eutoxeres condamini Saw-billed Hermit, Ramphodon naevius Bronzy Hermit, Glaucis aeneus Phaethornithinae Rufous-breasted Hermit, Glaucis hirsutus ?Hook-billed Hermit, Glaucis dohrnii Threnetes ruckeri Phaethornithini Band-tailed Barbthroat, Pale-tailed Barbthroat, Threnetes leucurus ?Sooty Barbthroat, Threnetes niger ?Broad-tipped Hermit, Anopetia gounellei White-bearded Hermit, Phaethornis hispidus Tawny-bellied Hermit, Phaethornis syrmatophorus Mexican Hermit, Phaethornis mexicanus Long-billed Hermit, Phaethornis longirostris Green Hermit, Phaethornis guy White-whiskered Hermit, Phaethornis yaruqui Great-billed Hermit, Phaethornis malaris Long-tailed Hermit, Phaethornis superciliosus Straight-billed Hermit, Phaethornis bourcieri Koepcke’s Hermit, Phaethornis koepckeae Needle-billed Hermit, Phaethornis philippii Buff-bellied Hermit, Phaethornis subochraceus Scale-throated Hermit, Phaethornis eurynome Sooty-capped Hermit, Phaethornis augusti Planalto Hermit, Phaethornis pretrei Pale-bellied Hermit, Phaethornis anthophilus Stripe-throated Hermit, Phaethornis striigularis Gray-chinned Hermit, Phaethornis griseogularis Black-throated Hermit, Phaethornis atrimentalis Reddish Hermit, Phaethornis ruber ?White-browed Hermit, Phaethornis stuarti ?Dusky-throated Hermit, Phaethornis squalidus Streak-throated Hermit, Phaethornis rupurumii Cinnamon-throated Hermit, Phaethornis nattereri Little Hermit, Phaethornis longuemareus ?Tapajos Hermit, Phaethornis aethopygus ?Minute Hermit, Phaethornis idaliae Polytminae: Mangos Lesbiini: Coquettes Lesbiinae Coeligenini: Brilliants Patagonini: Giant Hummingbird Lampornithini: Mountain-Gems Tro chilinae Mellisugini: Bees Cynanthini: Emeralds Trochilini: Amazilias Source: McGuire et al. (2014).. -

Lista Roja De Las Aves Del Uruguay 1
Lista Roja de las Aves del Uruguay 1 Lista Roja de las Aves del Uruguay Una evaluación del estado de conservación de la avifauna nacional con base en los criterios de la Unión Internacional para la Conservación de la Naturaleza. Adrián B. Azpiroz, Laboratorio de Genética de la Conservación, Instituto de Investigaciones Biológicas Clemente Estable, Av. Italia 3318 (CP 11600), Montevideo ([email protected]). Matilde Alfaro, Asociación Averaves & Facultad de Ciencias, Universidad de la República, Iguá 4225 (CP 11400), Montevideo ([email protected]). Sebastián Jiménez, Proyecto Albatros y Petreles-Uruguay, Centro de Investigación y Conservación Marina (CICMAR), Avenida Giannattasio Km 30.5. (CP 15008) Canelones, Uruguay; Laboratorio de Recursos Pelágicos, Dirección Nacional de Recursos Acuáticos, Constituyente 1497 (CP 11200), Montevideo ([email protected]). Cita sugerida: Azpiroz, A.B., M. Alfaro y S. Jiménez. 2012. Lista Roja de las Aves del Uruguay. Una evaluación del estado de conservación de la avifauna nacional con base en los criterios de la Unión Internacional para la Conservación de la Naturaleza. Dirección Nacional de Medio Ambiente, Montevideo. Descargo de responsabilidad El contenido de esta publicación es responsabilidad de los autores y no refleja necesariamente las opiniones o políticas de la DINAMA ni de las organizaciones auspiciantes y no comprometen a estas instituciones. Las denominaciones empleadas y la forma en que aparecen los datos no implica de parte de DINAMA, ni de las organizaciones auspiciantes o de los autores, juicio alguno sobre la condición jurídica de países, territorios, ciudades, personas, organizaciones, zonas o de sus autoridades, ni sobre la delimitación de sus fronteras o límites. -

Broad-Tailed Hummingbird Coloration and Sun Orientation 1
1 Broad-tailed hummingbird coloration and sun orientation 1 Two ways to display: male hummingbirds show different 2 color-display tactics based on sun orientation 3 Running header: Broad-tailed hummingbird coloration and sun orientation 4 5 Richard K. Simpson1* and Kevin J. McGraw1 6 1School of Life Sciences, Arizona State University, Tempe, AZ 85287-4501 7 *Corresponding Author. Email: [email protected]; Phone: (480) 965-2593 8 9 ABSTRACT 10 Animals exhibit a diversity of ornaments and courtship behaviors, which often co- 11 occur and are used for communication. The sensory drive hypothesis states that these 12 traits evolved and vary due to interactions with each other, the environment, and signal 13 receiver. However, interactions between colorful ornaments and courtship behaviors, 14 specifically in relation to environmental variation, remain poorly understood. We studied 15 male iridescent plumage (gorgets), display behavior, and sun orientation during courtship 16 flights (shuttle displays) in broad-tailed hummingbirds (Selasphorus platycercus), to 17 understand how these traits interact in both space and time to produce the perceived 18 coloration of males. We also tested how gorget coloration varies among males based on 19 their plumage, behavioral, and morphological characteristics. In contrast with previous 20 work on other animals, we found that displaying males did not directionally face the sun, 21 but instead displayed on a continuum of solar orientation angles. The gorgets of males 22 who tended to face the sun during their displays appeared flashier (i.e. exhibited greater 23 color/brightness changes), brighter, and more colorful, whereas the gorgets of males who 2 Broad-tailed hummingbird coloration and sun orientation 24 tended to not face the sun were more consistently reflective (i.e. -

Tiny Fossil Sheds Light on Miniaturization of Birds
Retraction Tiny fossil sheds light on miniaturization of birds Roger B. J. Benson Nature 579, 199–200 (2020) In view of the fact that the authors of ‘Hummingbird-sized dinosaur from the Cretaceous period of Myanmar‘ (L. Xing et al. Nature 579, 245–249; 2020) are retracting their report, I wish to retract this News & Views article, which dealt with this study and was based on the accuracy and reproducibility of their data. Nature | 22 July 2020 ©2020 Spri nger Nature Li mited. All rights reserved. drives the assembly of DNA-PK and stimulates regulation of protein synthesis. And, although 3. Dragon, F. et al. Nature 417, 967–970 (2002). its catalytic activity in vitro, although does so further studies are required, we might have 4. Adelmant, G. et al. Mol. Cell. Proteom. 11, 411–421 (2012). much less efficiently than can DNA. taken a step closer to deciphering the 5. Britton, S., Coates, J. & Jackson, S. P. J. Cell Biol. 202, Taken together, these observations suggest mysterious ribosomopathies. 579–595 (2013). a model in which KU recruits DNA-PKcs to the 6. Barandun, J. et al. Nature Struct. Mol. Biol. 24, 944–953 (2017). small-subunit processome. In the case of Alan J. Warren is at the Cambridge Institute 7. Ma, Y. et al. Cell 108, 781–794 (2002). kinase-defective DNA-PK, the mutant enzyme’s for Medical Research, Hills Road, Cambridge 8. Yin, X. et al. Cell Res. 27, 1341–1350 (2017). inability to regulate its own activity gives the CB2 OXY, UK. 9. Sharif, H. et al. -

The Tarsometatarsus of the Middle Eocene Loon Colymbiculus Udovichenkoi
– 17 – Paleornithological Research 2013 Proceed. 8th Inter nat. Meeting Society of Avian Paleontology and Evolution Ursula B. Göhlich & Andreas Kroh (Eds) The tarsometatarsus of the Middle Eocene loon Colymbiculus udovichenkoi GERALD MAYR1, LEONID GOROBETS2 & EVGENIJ ZVONOK3 1 Senckenberg Research Institute and Natural History Museum Frankfurt, Frankfurt am Main, Germany; E-mail: [email protected] 2 Taras Shevchenko National University of Kiev, Dept. of Ecology and Environmental Protection, Kiev, Ukraine 3 Institute of Geological Sciences of NAS of Ukraine, Branch of Paleontology and Stratigraphy, Kiev, Ukraine Abstract — We describe the previously unknown tarsometatarsus of the earliest unambiguously identified loon, Colymbiculus udovichenkoi, from the Middle Eocene of the Ukraine. Except for being more elongate and apart from details of the hypotarsus morphology, the bone resembles the tarsometatarsus of the Early Miocene Colym- boides minutus. We consider the hypotarsus morphology of Colymbiculus to be plesiomorphic for Gaviiformes. Colymboides and crown group Gaviiformes are each characterized by an autapomorphic hypotarsus morphology, which precludes the former from being directly ancestral to the latter. The similarities shared by C. udovichenkoi and C. minutus, including their small size, are likely to be plesiomorphic for Gaviiformes. Although the disap- pearance of small stem group Gaviiformes may be related to the retreat of loons to cold Northern latitudes, more data are needed to firmly establish this hypothesis. We finally note that early Paleogene stem group Gaviiformes markedly differ from putative Late Cretaceous loons, whose identification needs to be verified by further fossil specimens. Key words: Colymbiculus, Colymboides, fossil birds, Gaviiformes, Eocene, Lutetian, Ukraine Introduction 1982 from the Early Miocene of the Czech Republic (ŠVEC 1982), the earliest stem line- Loons (Gaviiformes) have a fairly comprehen- age representative, being distinctly smaller than sive Neogene fossil record (OLSON 1985; MAYR its modern congeners. -

Systematic Relationships and Biogeography of the Tracheophone Suboscines (Aves: Passeriformes)
MOLECULAR PHYLOGENETICS AND EVOLUTION Molecular Phylogenetics and Evolution 23 (2002) 499–512 www.academicpress.com Systematic relationships and biogeography of the tracheophone suboscines (Aves: Passeriformes) Martin Irestedt,a,b,* Jon Fjeldsaa,c Ulf S. Johansson,a,b and Per G.P. Ericsona a Department of Vertebrate Zoology and Molecular Systematics Laboratory, Swedish Museum of Natural History, P.O. Box 50007, SE-104 05 Stockholm, Sweden b Department of Zoology, University of Stockholm, SE-106 91 Stockholm, Sweden c Zoological Museum, University of Copenhagen, Copenhagen, Denmark Received 29 August 2001; received in revised form 17 January 2002 Abstract Based on their highly specialized ‘‘tracheophone’’ syrinx, the avian families Furnariidae (ovenbirds), Dendrocolaptidae (woodcreepers), Formicariidae (ground antbirds), Thamnophilidae (typical antbirds), Rhinocryptidae (tapaculos), and Conop- ophagidae (gnateaters) have long been recognized to constitute a monophyletic group of suboscine passerines. However, the monophyly of these families have been contested and their interrelationships are poorly understood, and this constrains the pos- sibilities for interpreting adaptive tendencies in this very diverse group. In this study we present a higher-level phylogeny and classification for the tracheophone birds based on phylogenetic analyses of sequence data obtained from 32 ingroup taxa. Both mitochondrial (cytochrome b) and nuclear genes (c-myc, RAG-1, and myoglobin) have been sequenced, and more than 3000 bp were subjected to parsimony and maximum-likelihood analyses. The phylogenetic signals in the mitochondrial and nuclear genes were compared and found to be very similar. The results from the analysis of the combined dataset (all genes, but with transitions at third codon positions in the cytochrome b excluded) partly corroborate previous phylogenetic hypotheses, but several novel arrangements were also suggested. -

Tinamiformes – Falconiformes
LIST OF THE 2,008 BIRD SPECIES (WITH SCIENTIFIC AND ENGLISH NAMES) KNOWN FROM THE A.O.U. CHECK-LIST AREA. Notes: "(A)" = accidental/casualin A.O.U. area; "(H)" -- recordedin A.O.U. area only from Hawaii; "(I)" = introducedinto A.O.U. area; "(N)" = has not bred in A.O.U. area but occursregularly as nonbreedingvisitor; "?" precedingname = extinct. TINAMIFORMES TINAMIDAE Tinamus major Great Tinamou. Nothocercusbonapartei Highland Tinamou. Crypturellus soui Little Tinamou. Crypturelluscinnamomeus Thicket Tinamou. Crypturellusboucardi Slaty-breastedTinamou. Crypturellus kerriae Choco Tinamou. GAVIIFORMES GAVIIDAE Gavia stellata Red-throated Loon. Gavia arctica Arctic Loon. Gavia pacifica Pacific Loon. Gavia immer Common Loon. Gavia adamsii Yellow-billed Loon. PODICIPEDIFORMES PODICIPEDIDAE Tachybaptusdominicus Least Grebe. Podilymbuspodiceps Pied-billed Grebe. ?Podilymbusgigas Atitlan Grebe. Podicepsauritus Horned Grebe. Podicepsgrisegena Red-neckedGrebe. Podicepsnigricollis Eared Grebe. Aechmophorusoccidentalis Western Grebe. Aechmophorusclarkii Clark's Grebe. PROCELLARIIFORMES DIOMEDEIDAE Thalassarchechlororhynchos Yellow-nosed Albatross. (A) Thalassarchecauta Shy Albatross.(A) Thalassarchemelanophris Black-browed Albatross. (A) Phoebetriapalpebrata Light-mantled Albatross. (A) Diomedea exulans WanderingAlbatross. (A) Phoebastriaimmutabilis Laysan Albatross. Phoebastrianigripes Black-lootedAlbatross. Phoebastriaalbatrus Short-tailedAlbatross. (N) PROCELLARIIDAE Fulmarus glacialis Northern Fulmar. Pterodroma neglecta KermadecPetrel. (A) Pterodroma -

REGUA Bird List July 2020.Xlsx
Birds of REGUA/Aves da REGUA Updated July 2020. The taxonomy and nomenclature follows the Comitê Brasileiro de Registros Ornitológicos (CBRO), Annotated checklist of the birds of Brazil by the Brazilian Ornithological Records Committee, updated June 2015 - based on the checklist of the South American Classification Committee (SACC). Atualizado julho de 2020. A taxonomia e nomenclatura seguem o Comitê Brasileiro de Registros Ornitológicos (CBRO), Lista anotada das aves do Brasil pelo Comitê Brasileiro de Registros Ornitológicos, atualizada em junho de 2015 - fundamentada na lista do Comitê de Classificação da América do Sul (SACC). -

Project Scientific Progress Report Study Site
Project Ecology of plant-hummingbird interactions along an elevational gradient Scientific Progress Report Project leader: Catherine Graham, Swiss Federal Research Institute Principal investigator: María Alejandra Maglianesi, Universidad Estatal a Distancia Coordinator: Emanuel Brenes Rodríguez, Universidad Estatal a Distancia Study site Las Nubes Biological Reserve York University San José, Costa Rica January, 2020 1 INTRODUCTION A primary aim of community ecology is to identify the processes that govern species assemblages across environmental gradients (McGill et al. 2006), allowing us to understand why biodiversity is non-randomly distributed on Earth. Mutualistic interactions such as those between plants and their animal pollinators are the major biodiversity component from which the integrity of ecosystems depends (Valiente-Banuet et al. 2015). The interdependence of plant and pollinators can be assessed using a network approach, which is a powerful tool to analyze the complexity of ecological systems (Ings et al. 2009), especially in highly diversified tropical regions. Mountain regions provide pronounced environmental gradients across relatively small spatial scales and have proved to be a suitable model system to investigate patterns and determinants of species diversity and community structure (Körner 2000, Sanders and Rahbek 2012). Although some studies have investigated the variation in plant–pollinator interaction networks across elevational gradients (Ramos-Jiliberto et al. 2010, Benadi et al. 2013), such studies are still scarce, particularly in the tropics. In the Neotropics, hummingbirds (Trochilidae) are considered to be effective pollinators (Castellanos et al. 2003). They have been classified into two distinct groups: hermits and non-hermits, which differ mainly in their elevational distribution and their level of specialization on floral resources, i.e., the proportion of floral resources available in the community that is used by species (Stiles 1978). -

Dieter Thomas Tietze Editor How They Arise, Modify and Vanish
Fascinating Life Sciences Dieter Thomas Tietze Editor Bird Species How They Arise, Modify and Vanish Fascinating Life Sciences This interdisciplinary series brings together the most essential and captivating topics in the life sciences. They range from the plant sciences to zoology, from the microbiome to macrobiome, and from basic biology to biotechnology. The series not only highlights fascinating research; it also discusses major challenges associated with the life sciences and related disciplines and outlines future research directions. Individual volumes provide in-depth information, are richly illustrated with photographs, illustrations, and maps, and feature suggestions for further reading or glossaries where appropriate. Interested researchers in all areas of the life sciences, as well as biology enthusiasts, will find the series’ interdisciplinary focus and highly readable volumes especially appealing. More information about this series at http://www.springer.com/series/15408 Dieter Thomas Tietze Editor Bird Species How They Arise, Modify and Vanish Editor Dieter Thomas Tietze Natural History Museum Basel Basel, Switzerland ISSN 2509-6745 ISSN 2509-6753 (electronic) Fascinating Life Sciences ISBN 978-3-319-91688-0 ISBN 978-3-319-91689-7 (eBook) https://doi.org/10.1007/978-3-319-91689-7 Library of Congress Control Number: 2018948152 © The Editor(s) (if applicable) and The Author(s) 2018. This book is an open access publication. Open Access This book is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made. -

Guia Para Observação Das Aves Do Parque Nacional De Brasília
See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/234145690 Guia para observação das aves do Parque Nacional de Brasília Book · January 2011 CITATIONS READS 0 629 4 authors, including: Mieko Kanegae Fernando Lima Favaro Federal University of Rio de Janeiro Instituto Chico Mendes de Conservação da Bi… 7 PUBLICATIONS 74 CITATIONS 17 PUBLICATIONS 69 CITATIONS SEE PROFILE SEE PROFILE All content following this page was uploaded by Fernando Lima Favaro on 28 May 2014. The user has requested enhancement of the downloaded file. Brasília - 2011 GUIA PARA OBSERVAÇÃO DAS AVES DO PARQUE NACIONAL DE BRASÍLIA Aílton C. de Oliveira Mieko Ferreira Kanegae Marina Faria do Amaral Fernando de Lima Favaro Fotografia de Aves Marcelo Pontes Monteiro Nélio dos Santos Paulo André Lima Borges Brasília, 2011 GUIA PARA OBSERVAÇÃO DAS AVES DO APRESENTAÇÃO PARQUE NACIONAL DE BRASÍLIA É com grande satisfação que apresento o Guia para Observação REPÚblica FEDERATiva DO BRASIL das Aves do Parque Nacional de Brasília, o qual representa um importante instrumento auxiliar para os observadores de aves que frequentam ou que Presidente frequentarão o Parque, para fins de lazer (birdwatching), pesquisas científicas, Dilma Roussef treinamentos ou em atividades de educação ambiental. Este é mais um resultado do trabalho do Centro Nacional de Pesquisa e Vice-Presidente Conservação de Aves Silvestres - CEMAVE, unidade descentralizada do Instituto Michel Temer Chico Mendes de Conservação da Biodiversidade (ICMBio) e vinculada à Diretoria de Conservação da Biodiversidade. O Centro tem como missão Ministério do Meio Ambiente - MMA subsidiar a conservação das aves brasileiras e dos ambientes dos quais elas Izabella Mônica Vieira Teixeira dependem. -

Wildlife Ecology Provincial Resources
MANITOBA ENVIROTHON WILDLIFE ECOLOGY PROVINCIAL RESOURCES !1 ACKNOWLEDGEMENTS We would like to thank: Olwyn Friesen (PhD Ecology) for compiling, writing, and editing this document. Subject Experts and Editors: Barbara Fuller (Project Editor, Chair of Test Writing and Education Committee) Lindsey Andronak (Soils, Research Technician, Agriculture and Agri-Food Canada) Jennifer Corvino (Wildlife Ecology, Senior Park Interpreter, Spruce Woods Provincial Park) Cary Hamel (Plant Ecology, Director of Conservation, Nature Conservancy Canada) Lee Hrenchuk (Aquatic Ecology, Biologist, IISD Experimental Lakes Area) Justin Reid (Integrated Watershed Management, Manager, La Salle Redboine Conservation District) Jacqueline Monteith (Climate Change in the North, Science Consultant, Frontier School Division) SPONSORS !2 Introduction to wildlife ...................................................................................7 Ecology ....................................................................................................................7 Habitat ...................................................................................................................................8 Carrying capacity.................................................................................................................... 9 Population dynamics ..............................................................................................................10 Basic groups of wildlife ................................................................................11