Plant Ecophysiology
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Mycodiversity Studies in Selected Ecosystems of Greece: 5
Uploaded — May 2011 [Link page — MYCOTAXON 115: 535] Expert reviewers: Giuseppe Venturella, Solomon P. Wasser Mycodiversity studies in selected ecosystems of Greece: 5. Basidiomycetes associated with woods dominated by Castanea sativa (Nafpactia Mts., central Greece) ELIAS POLEMIS1, DIMITRIS M. DIMOU1,3, LEONIDAS POUNTZAS4, DIMITRIS TZANOUDAKIS2 & GEORGIOS I. ZERVAKIS1* 1 [email protected], [email protected] Agricultural University of Athens, Lab. of General & Agricultural Microbiology Iera Odos 75, 11855 Athens, Greece 2 University of Patras, Dept. of Biology, Panepistimioupoli, 26500 Rion, Greece 3 Koritsas 10, 15343 Agia Paraskevi, Greece 4 Technological Educational Institute of Mesologgi, 30200 Mesologgi, Greece Abstract — Very scarce literature data are available on the macrofungi associated with sweet chestnut trees (Castanea sativa, Fagaceae). We report here the results of an inventory of basidiomycetes, which was undertaken in the region of Nafpactia Mts., central Greece. The investigated area, with woods dominated by C. sativa, was examined for the first time in respect to its mycodiversity. One hundred and four species belonging in 54 genera were recorded. Fifteen species (Conocybe pseudocrispa, Entoloma nitens, Lactarius glaucescens, Lichenomphalia velutina, Parasola schroeteri, Pholiotina coprophila, Russula alutacea, R. azurea, R. pseudoaeruginea, R. pungens, R. vitellina, Sarcodon glaucopus, Tomentella badia, T. fibrosa and Tubulicrinis sororius) are reported for the first time from Greece. In addition, 33 species constitute new habitats/hosts/substrates records. Key words — biodiversity, macromycete, Mediterranean, mushroom Introduction Castanea sativa Mill., Fagaceae (sweet chestnut) generally prefers north- facing slopes where the rainfall is greater than 600 mm, on moderately acid soils (pH 4.5–6.5) with a light texture. It covers ca. -

Intraspecific Facilitation: Generality, Mechanisms and Effects
Intraspecific facilitation: generality, mechanisms and effects Dissertation der Mathematisch-Naturwissenschaftlichen Fakultät der Eberhard Karls Universität Tübingen zur Erlangung des Grades eines Doktors der Naturwissenschaften (Dr. rer. nat.) vorgelegt von Ruichang Zhang aus Shandong, China Tübingen 2017 Gedruckt mit Genehmigung der Mathematisch-Naturwissenschaftlichen Fakultät der Eberhard Karls Universität Tübingen. Tag der mündlichen Qualifikation: 07. April. 2017 Dekan: Prof. Dr. Wolfgang Rosenstiel 1. Berichterstatter: Prof. Dr. Katja Tielbörger 2. Berichterstatter: Prof. Dr. Oliver Bossdorf 2 Table of contents Acknowledgments 4 Declaration of my own working contribution to the present thesis 5 Abstract 6 Chapter 1: 8 General introduction Chapter 2: 15 A review on reciprocity and asymmetry/symmetry in facilitation: the foundation for exploring population-level consequences of facilitation Chapter 3: 45 Intraspecific facilitation: responses and effects of conspecific genotypes with different stress tolerance Chapter 4: 63 Density-dependence tips the balance between facilitation and competition along stress gradients Chapter 5: 80 Facilitation alters allometric responses of plants to density Chapter 6: 94 General conclusions Bibliography: 101 Appendices: 127 Curriculum vitae: 188 3 Acknowledgement I spent four years of my life on the thesis and now it is my pleasure to acknowledge and thank all the people who helped me during this period. I would like to acknowledge the financial support from China Scholarship Council (CSC). Without this funding source my stay in Germany and research could not have been possible. I am tremendously grateful to my supervisor Prof. Dr. Katja Tielbörger for her guidance and support during the past four years. A huge thank also goes to Prof. Dr. Oliver Bossdorf for sharing the greenhouse and stimulating discussion. -

Influence of Tree Species on Richness and Diversity of Epigeous Fungal
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Archive Ouverte en Sciences de l'Information et de la Communication fungal ecology 4 (2011) 22e31 available at www.sciencedirect.com journal homepage: www.elsevier.com/locate/funeco Influence of tree species on richness and diversity of epigeous fungal communities in a French temperate forest stand Marc BUE´Ea,*, Jean-Paul MAURICEb, Bernd ZELLERc, Sitraka ANDRIANARISOAc, Jacques RANGERc,Re´gis COURTECUISSEd, Benoıˆt MARC¸AISa, Franc¸ois LE TACONa aINRA Nancy, UMR INRA/UHP 1136 Interactions Arbres/Microorganismes, 54280 Champenoux, France bGroupe Mycologique Vosgien, 18 bis, place des Cordeliers, 88300 Neufchaˆteau, France cINRA Nancy, UR 1138 Bioge´ochimie des Ecosyste`mes Forestiers, 54280 Champenoux, France dUniversite´ de Lille, Faculte´ de Pharmacie, F59006 Lille, France article info abstract Article history: Epigeous saprotrophic and ectomycorrhizal (ECM) fungal sporocarps were assessed during Received 30 September 2009 7 yr in a French temperate experimental forest site with six 30-year-old mono-specific Revision received 10 May 2010 plantations (four coniferous and two hardwood plantations) and one 150-year-old native Accepted 21 July 2010 mixed deciduous forest. A total of 331 fungal species were identified. Half of the fungal Available online 6 October 2010 species were ECM, but this proportion varied slightly by forest composition. The replace- Corresponding editor: Anne Pringle ment of the native forest by mono-specific plantations, including native species such as beech and oak, considerably altered the diversity of epigeous ECM and saprotrophic fungi. Keywords: Among the six mono-specific stands, fungal diversity was the highest in Nordmann fir and Conifer plantation Norway spruce plantations and the lowest in Corsican pine and Douglas fir plantations. -

Phylogenetic Analyses of Juniperus Species in Turkey and Their Relations with Other Juniperus Based on Cpdna Supervisor: Prof
MOLECULAR PHYLOGENETIC ANALYSES OF JUNIPERUS L. SPECIES IN TURKEY AND THEIR RELATIONS WITH OTHER JUNIPERS BASED ON cpDNA A THESIS SUBMITTED TO THE GRADUATE SCHOOL OF NATURAL AND APPLIED SCIENCES OF MIDDLE EAST TECHNICAL UNIVERSITY BY AYSUN DEMET GÜVENDİREN IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY IN BIOLOGY APRIL 2015 Approval of the thesis MOLECULAR PHYLOGENETIC ANALYSES OF JUNIPERUS L. SPECIES IN TURKEY AND THEIR RELATIONS WITH OTHER JUNIPERS BASED ON cpDNA submitted by AYSUN DEMET GÜVENDİREN in partial fulfillment of the requirements for the degree of Doctor of Philosophy in Department of Biological Sciences, Middle East Technical University by, Prof. Dr. Gülbin Dural Ünver Dean, Graduate School of Natural and Applied Sciences Prof. Dr. Orhan Adalı Head of the Department, Biological Sciences Prof. Dr. Zeki Kaya Supervisor, Dept. of Biological Sciences METU Examining Committee Members Prof. Dr. Musa Doğan Dept. Biological Sciences, METU Prof. Dr. Zeki Kaya Dept. Biological Sciences, METU Prof.Dr. Hayri Duman Biology Dept., Gazi University Prof. Dr. İrfan Kandemir Biology Dept., Ankara University Assoc. Prof. Dr. Sertaç Önde Dept. Biological Sciences, METU Date: iii I hereby declare that all information in this document has been obtained and presented in accordance with academic rules and ethical conduct. I also declare that, as required by these rules and conduct, I have fully cited and referenced all material and results that are not original to this work. Name, Last name : Aysun Demet GÜVENDİREN Signature : iv ABSTRACT MOLECULAR PHYLOGENETIC ANALYSES OF JUNIPERUS L. SPECIES IN TURKEY AND THEIR RELATIONS WITH OTHER JUNIPERS BASED ON cpDNA Güvendiren, Aysun Demet Ph.D., Department of Biological Sciences Supervisor: Prof. -

Extract of Juniperus Indica Bertol Synergizes with Cisplatin to Inhibit
molecules Article Extract of Juniperus indica Bertol Synergizes with Cisplatin to Inhibit Oral Cancer Cell Growth via Repression of Cell Cycle Progression and Activation of the Caspase Cascade Xiao-Fan Huang 1,2, Kai-Fu Chang 1,2 , Shan-Chih Lee 3,4, Chia-Yu Li 5, Hung-Hsiu Liao 2, Ming-Chang Hsieh 2,6,* and Nu-Man Tsai 2,6,* 1 Institute of Medicine, Chung Shan Medical University, Taichung 40201, Taiwan; [email protected] (X.-F.H.); [email protected] (K.-F.C.) 2 Department of Medical Laboratory and Biotechnology, Chung Shan Medical University, Taichung 40201, Taiwan; [email protected] 3 Department of Medical Imaging and Radiological Sciences, Chung Shan Medical University, Taichung 40201, Taiwan; [email protected] 4 Department of Medical Imaging, Chung Shan Medical University Hospital, Taichung 40201, Taiwan 5 Department of Life and Death, Nanhua University, Chiayi 62249, Taiwan; [email protected] 6 Clinical Laboratory, Chung Shan Medical University Hospital, Taichung 40201, Taiwan * Correspondence: [email protected] (M.-C.H.); [email protected] (N.-M.T.); Tel.: +886-4-2473-0022 (ext. 12411) (N.-M.T.); Fax: +886-4-2324-8171 (N.-M.T.) Academic Editors: Halina Ekiert and Agnieszka Szopa Received: 21 May 2020; Accepted: 12 June 2020; Published: 13 June 2020 Abstract: Oral cancer—a type of head and neck cancer—is estimated to be the fifth most common cancer in Taiwan. However, efficacious therapies for oral cancer are still lacking due to drug resistance and recurrence. Consequently, the identification of new anticancer agents for clinical treatment is needed. -

Himalayan Aromatic Medicinal Plants: a Review of Their Ethnopharmacology, Volatile Phytochemistry, and Biological Activities
medicines Review Himalayan Aromatic Medicinal Plants: A Review of their Ethnopharmacology, Volatile Phytochemistry, and Biological Activities Rakesh K. Joshi 1, Prabodh Satyal 2 and Wiliam N. Setzer 2,* 1 Department of Education, Government of Uttrakhand, Nainital 263001, India; [email protected] 2 Department of Chemistry, University of Alabama in Huntsville, Huntsville, AL 35899, USA; [email protected] * Correspondence: [email protected]; Tel.: +1-256-824-6519; Fax: +1-256-824-6349 Academic Editor: Lutfun Nahar Received: 24 December 2015; Accepted: 3 February 2016; Published: 19 February 2016 Abstract: Aromatic plants have played key roles in the lives of tribal peoples living in the Himalaya by providing products for both food and medicine. This review presents a summary of aromatic medicinal plants from the Indian Himalaya, Nepal, and Bhutan, focusing on plant species for which volatile compositions have been described. The review summarizes 116 aromatic plant species distributed over 26 families. Keywords: Jammu and Kashmir; Himachal Pradesh; Uttarakhand; Nepal; Sikkim; Bhutan; essential oils 1. Introduction The Himalya Center of Plant Diversity [1] is a narrow band of biodiversity lying on the southern margin of the Himalayas, the world’s highest mountain range with elevations exceeding 8000 m. The plant diversity of this region is defined by the monsoonal rains, up to 10,000 mm rainfall, concentrated in the summer, altitudinal zonation, consisting of tropical lowland rainforests, 100–1200 m asl, up to alpine meadows, 4800–5500 m asl. Hara and co-workers have estimated there to be around 6000 species of higher plants in Nepal, including 303 species endemic to Nepal and 1957 species restricted to the Himalayan range [2–4]. -
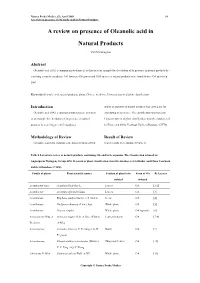
A Review on Presence of Oleanolic Acid in Natural Products
Natura Proda Medica, (2), April 2009 64 A review on presence of Oleanolic acid in Natural Products A review on presence of Oleanolic acid in Natural Products YEUNG Ming Fai Abstract Oleanolic acid (OA), a common phytochemical, is chosen as an example for elucidation of its presence in natural products by searching scientific databases. 146 families, 698 genera and 1620 species of natural products were found to have OA up to Sep 2007. Keywords Oleanolic acid, natural products, plants, Chinese medicine, Linnaeus system of plant classification Introduction and/or its saponins in natural products was carried out for Oleanolic acid (OA), a common phytochemical, is chosen elucidating its pressence. The classification was based on as an example for elucidation of its presence in natural Linnaeus system of plant classification from the databases of products by searching scientific databases. SciFinder and China Yearbook Full-text Database (CJFD). Methodology of Review Result of Review Literature search for isolation and characterization of OA Search results were tabulated (Table 1). Table 1 Literature review of natural products containing OA and/or its saponins. The classification is based on Angiosperm Phylogeny Group APG II system of plant classification from the databases of SciFinder and China Yearbook Full-text Database (CJFD). Family of plants Plant scientific names Position of plant to be Form of OA References isolated isolated Acanthaceae Juss. Acanthus illicifolius L. Leaves OA [1-2] Acanthaceae Avicennia officinalis Linn. Leaves OA [3] Acanthaceae Blepharis sindica Stocks ex T. Anders Seeds OA [4] Acanthaceae Dicliptera chinensis (Linn.) Juss. Whole plant OA [5] Acanthaceae Justicia simplex Whole plant OA saponins [6] Actinidiaceae Gilg. -

Chemical Elements in Ascomycetes and Basidiomycetes
Chemical elements in Ascomycetes and Basidiomycetes The reference mushrooms as instruments for investigating bioindication and biodiversity Roberto Cenci, Luigi Cocchi, Orlando Petrini, Fabrizio Sena, Carmine Siniscalco, Luciano Vescovi Editors: R. M. Cenci and F. Sena EUR 24415 EN 2011 1 The mission of the JRC-IES is to provide scientific-technical support to the European Union’s policies for the protection and sustainable development of the European and global environment. European Commission Joint Research Centre Institute for Environment and Sustainability Via E.Fermi, 2749 I-21027 Ispra (VA) Italy Legal Notice Neither the European Commission nor any person acting on behalf of the Commission is responsible for the use which might be made of this publication. Europe Direct is a service to help you find answers to your questions about the European Union Freephone number (*): 00 800 6 7 8 9 10 11 (*) Certain mobile telephone operators do not allow access to 00 800 numbers or these calls may be billed. A great deal of additional information on the European Union is available on the Internet. It can be accessed through the Europa server http://europa.eu/ JRC Catalogue number: LB-NA-24415-EN-C Editors: R. M. Cenci and F. Sena JRC65050 EUR 24415 EN ISBN 978-92-79-20395-4 ISSN 1018-5593 doi:10.2788/22228 Luxembourg: Publications Office of the European Union Translation: Dr. Luca Umidi © European Union, 2011 Reproduction is authorised provided the source is acknowledged Printed in Italy 2 Attached to this document is a CD containing: • A PDF copy of this document • Information regarding the soil and mushroom sampling site locations • Analytical data (ca, 300,000) on total samples of soils and mushrooms analysed (ca, 10,000) • The descriptive statistics for all genera and species analysed • Maps showing the distribution of concentrations of inorganic elements in mushrooms • Maps showing the distribution of concentrations of inorganic elements in soils 3 Contact information: Address: Roberto M. -

Phd. Thesis Sana Jabeen.Pdf
ECTOMYCORRHIZAL FUNGAL COMMUNITIES ASSOCIATED WITH HIMALAYAN CEDAR FROM PAKISTAN A dissertation submitted to the University of the Punjab in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY in BOTANY by SANA JABEEN DEPARTMENT OF BOTANY UNIVERSITY OF THE PUNJAB LAHORE, PAKISTAN JUNE 2016 TABLE OF CONTENTS CONTENTS PAGE NO. Summary i Dedication iii Acknowledgements iv CHAPTER 1 Introduction 1 CHAPTER 2 Literature review 5 Aims and objectives 11 CHAPTER 3 Materials and methods 12 3.1. Sampling site description 12 3.2. Sampling strategy 14 3.3. Sampling of sporocarps 14 3.4. Sampling and preservation of fruit bodies 14 3.5. Morphological studies of fruit bodies 14 3.6. Sampling of morphotypes 15 3.7. Soil sampling and analysis 15 3.8. Cleaning, morphotyping and storage of ectomycorrhizae 15 3.9. Morphological studies of ectomycorrhizae 16 3.10. Molecular studies 16 3.10.1. DNA extraction 16 3.10.2. Polymerase chain reaction (PCR) 17 3.10.3. Sequence assembly and data mining 18 3.10.4. Multiple alignments and phylogenetic analysis 18 3.11. Climatic data collection 19 3.12. Statistical analysis 19 CHAPTER 4 Results 22 4.1. Characterization of above ground ectomycorrhizal fungi 22 4.2. Identification of ectomycorrhizal host 184 4.3. Characterization of non ectomycorrhizal fruit bodies 186 4.4. Characterization of saprobic fungi found from fruit bodies 188 4.5. Characterization of below ground ectomycorrhizal fungi 189 4.6. Characterization of below ground non ectomycorrhizal fungi 193 4.7. Identification of host taxa from ectomycorrhizal morphotypes 195 4.8. -
![Downloaded from EMBL [25]](https://docslib.b-cdn.net/cover/3379/downloaded-from-embl-25-1133379.webp)
Downloaded from EMBL [25]
BMC Microbiology BioMed Central Methodology article Open Access Fungal-specific PCR primers developed for analysis of the ITS region of environmental DNA extracts Kendall J Martin*1 and Paul T Rygiewicz2 Address: 1Dynamac Corporation, National Health and Environmental Effects Research Laboratory, Corvallis, OR USA and 2USEPA National Health and Environmental Effects Research Laboratory, Corvallis, OR, USA Email: Kendall J Martin* - [email protected]; Paul T Rygiewicz - [email protected] * Corresponding author Published: 18 May 2005 Received: 30 November 2004 Accepted: 18 May 2005 BMC Microbiology 2005, 5:28 doi:10.1186/1471-2180-5-28 This article is available from: http://www.biomedcentral.com/1471-2180/5/28 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. PCRprimersbasidiomycetesascomycetesInternal Transcribed Spacer (ITS)X/T rapid extraction method Abstract Background: The Internal Transcribed Spacer (ITS) regions of fungal ribosomal DNA (rDNA) are highly variable sequences of great importance in distinguishing fungal species by PCR analysis. Previously published PCR primers available for amplifying these sequences from environmental samples provide varying degrees of success at discriminating against plant DNA while maintaining a broad range of compatibility. Typically, it has been necessary to use multiple primer sets to accommodate the range of fungi under study, potentially creating artificial distinctions for fungal sequences that amplify with more than one primer set. Results: Numerous sequences for PCR primers were tested to develop PCR assays with a wide range of fungal compatibility and high discrimination from plant DNA. -

Jardín Botánico Chagual De Santiago
Chagual/EDITORIAL JARDÍN BOTÁNICO CHAGUAL DE SANTIAGO un año de la creación de la Corporación que avala su quehacer, y siguiendo los lineamientos del BGCI (Botanic Gardens Conservation International, Inglaterra), el Jardín Botánico Chagual de Santiago es un proyecto de educación A y conservación de flora nativa de Chile que empieza a concretarse. Hasta fechas muy recientes los términos patrimonio nacional o patrimonio de la humanidad se aplicaban exclusivamente a obras creadas por el hombre. Sin embargo, dada la presión y deterioro al que se encuentran sometidos los recursos naturales este concepto se ha ampliado, extendiéndose a entidades del mundo natural. Así, hoy en día, bosques, dunas, humedales y hasta especies individuales constituyen entes de valor patrimonial y su preservación es valorada como expresión de cultura . Impulsados por el desafío de colaborar a preservar la naturaleza y avalados por su reconocida experiencia en la aclimatación y propagación de plantas fuera de sus lugares de origen, los jardines botánicos inician un importante protagonismo en la educación, al relevar el valor que tienen las plantas y sus hábitat como sistema y soporte de la vida. En ese sentido, promueven la conservación de plantas nativas amenazadas, difunden prácticas y conocimientos hortícolas, rescatan el legado dejado por las culturas ancestrales -en relación al uso de las plantas- y desarrollan variados programas de educación escolar y educación para adultos. Chile es un país de larga y rica tradición botánica. Apoyándose en esa trayectoria, y conciente que el conocimiento científico de la naturaleza contribuye a una actitud de respeto y cuidado por ella, el Jardín Botánico Chagual se ha propuesto -como uno de sus objetivos más ambiciosos- contribuir al desarrollo de una cultura de la naturaleza capaz de apreciar y conservar la diversidad vegetal y florística de la zona de clima mediterráneo de Chile. -
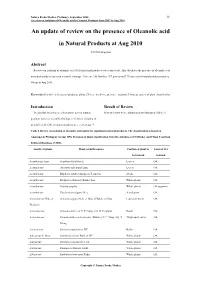
An Update of Review on the Presence of Oleanolic Acid in Natural Products at Aug 2010
Natura Proda Medica, Prelimary, September 2010 11 A review on isolation of Oleanolic acid in Natural Products from 2007 to Aug 2010 An update of review on the presence of Oleanolic acid in Natural Products at Aug 2010 YEUNG Ming Fai Abstract Reviews on isolation of oleanolic acid (OA) in natural products were carried out. This elucidates the presence of oleanolic acid in natural products based on scientific findings. There are 158 families, 767 genera and 1710 species of natural products isolated OA up to Aug 2010. Keywords Oleanolic acid, natural products, plants, Chinese medicine, presence, isolation, Linnaeus system of plant classification Introduction Result of Review To elucidate the presence of oleanolic acid in natural Review results were collaborated and tabulated (Table 1). products based on scientific findings, reviews on isolation of oleanolic acid (OA) in natural products were carried out 1-2. Table 1 Review on isolation of oleanolic acid and/or its saponins in natural products. The classification is based on Angiosperm Phylogeny Group APG II system of plant classification from the databases of SciFinder and China Yearbook Full-text Database (CJFD). Family of plants Plant scientific names Position of plant to Form of OA be isolated isolated Acanthaceae Juss. Acanthus illicifolius L. Leaves OA Acanthaceae Avicennia officinalis Linn. Leaves OA Acanthaceae Blepharis sindica Stocks ex T. Anders Seeds OA Acanthaceae Dicliptera chinensis (Linn.) Juss. Whole plants OA Acanthaceae Justicia simplex Wholeplants OAsaponins Acanthaceae Gendarussa vulgaris Nees Aerial parts OA Actinidiaceae Gilg. et Actinidia arguta (Sieb. et Zucc.) Planch. ex Miq. Leaves or stems OA Werderm. Actinidiaceae Actinidia deliciosa C.