The Ants of Oklahoma Master of Science
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Delsinne, T., Sonet, G. & Donoso, D.A. 2015. Two New
European Journal of Taxonomy 143: 1–35 ISSN 2118-9773 http://dx.doi.org/10.5852/ejt.2015.143 www.europeanjournaloftaxonomy.eu 2015 · Delsinne T., Sonet G. & Donoso D.A. This work is licensed under a Creative Commons Attribution 3.0 License. Research article urn:lsid:zoobank.org:pub:CA2B7F29-C4C3-4BCC-A65B-40733C856652 Two new species of Leptanilloides Mann, 1823 (Formicidae: Dorylinae) from the Andes of southern Ecuador Thibaut DELSINNE 1,4,*, Gontran SONET 2 & David A. DONOSO 1,3 1 Universidad Técnica Particular de Loja (UTPL), Departamento de Ciencias Naturales, Museo de Colecciones Biológicas (MUTPL), Loja, Ecuador. 2 Royal Belgian Institute of Natural Sciences, Joint Experimental Molecular Unit (JEMU) – OD Taxonomy and Phylogeny, Vautierstraat 29, B-1000 Brussels, Belgium. 3 Universidad de Cuenca, Facultad de Ciencias, Agropecuarias, Ave. 12 de Abril s/n, Cuenca, Ecuador * Corresponding author: [email protected] 2 E-mail: [email protected] 3 E-mail: [email protected] 4 urn:lsid:zoobank.org:author:BA819CA7-A1BC-43AC-94A3-9DBB2D89D634 2 urn:lsid:zoobank.org:author:BA7353F8-5296-4968-9D2D-D474EF8FCEB6 3 urn:lsid:zoobank.org:author:BD09BA33-A9F8-4F77-A5B3-F277062FB286 Abstract. Two new species of Leptanilloides are described: L. copalinga Delsinne & Donoso sp. nov. and L. prometea Delsinne & Donoso sp. nov., based on workers collected in the leaf litter and soil of the Andes of southern Ecuador. Both species belong to the L. biconstricta species-group (formally diagnosed here). The metatibial gland, considered a synapomorphy for Dorylinae, is observed in L. prometea sp. nov. but seems absent in L. copalinga sp. -
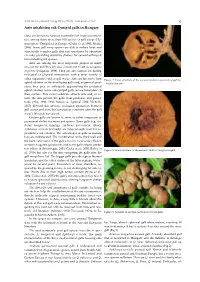
Ants Inhabiting Oak Cynipid Galls in Hungary
North-Western Journal of Zoology 2020, vol.16 (1) - Correspondence: Notes 95 Ants inhabiting oak Cynipid galls in Hungary Oaks are known to harbour extremely rich insect communi- ties, among them more than 100 species of gall wasps (Hy- menoptera: Cynipidae) in Europe (Csóka et al. 2005, Melika 2006). Some gall wasp species are able to induce large and structurally complex galls that can sometimes be abundant on oaks, providing attractive shelters for several arthropod taxa including ant species. Ants are among the most important players in many ecosystems and they are also considered to act as ecosystem engineers (Folgarait, 1998). They are also famous for having ecological or physical interactions with a great variety of other organisms, such as gall wasps. Ants are known to tend Figure 1. Inner structure of the asexual Andricus quercustozae gall in- aphid colonies on the developing galls and, as general pred- habited by ants. ators, they prey on arthropods approaching the protected aphid colonies. Some oak cynipid galls secrete honeydew on their surface. This sweet substrate attracts ants and, in re- turn, the ants protect the galls from predators and parasi- toids (Abe, 1988, 1992; Inouye & Agrawal 2004; Nicholls, 2017). Beyond this obvious ecological interaction between gall wasps and ants, this association continues after the gall wasp’s life cycle has ceased. Certain galls are known to serve as either temporary or permanent shelter for many ant species. Some galls (e.g. An- dricus hungaricus (Hartig), Andricus quercustozae (Bosc), Aphelonyx cerricola (Giraud)) are large enough even for re- productive ant colonies. The advantages of galls as nesting logs are multifaceted. -

Predicting Future Coexistence in a North American Ant Community Sharon Bewick1,2, Katharine L
Predicting future coexistence in a North American ant community Sharon Bewick1,2, Katharine L. Stuble1, Jean-Phillipe Lessard3,4, Robert R. Dunn5, Frederick R. Adler6,7 & Nathan J. Sanders1,3 1Department of Ecology and Evolutionary Biology, University of Tennessee, Knoxville, Tennessee 2National Institute for Mathematical and Biological Synthesis, University of Tennessee, Knoxville, Tennessee 3Center for Macroecology, Evolution and Climate, Natural History Museum of Denmark, University of Copenhagen, Copenhagen, Denmark 4Quebec Centre for Biodiversity Science, Department of Biology, McGill University, Montreal, Quebec, Canada 5Department of Biological Sciences, North Carolina State University, Raleigh, North Carolina 6Department of Mathematics, University of Utah, Salt Lake City, Utah 7Department of Biology, University of Utah, Salt Lake City, Utah Keywords Abstract Ant communities, climate change, differential equations, mechanistic models, species Global climate change will remodel ecological communities worldwide. How- interactions. ever, as a consequence of biotic interactions, communities may respond to cli- mate change in idiosyncratic ways. This makes predictive models that Correspondence incorporate biotic interactions necessary. We show how such models can be Sharon Bewick, Department of Biology, constructed based on empirical studies in combination with predictions or University of Maryland, College Park, MD assumptions regarding the abiotic consequences of climate change. Specifically, 20742, USA. Tel: 724-833-4459; we consider a well-studied ant community in North America. First, we use his- Fax: 301-314-9358; E-mail: [email protected] torical data to parameterize a basic model for species coexistence. Using this model, we determine the importance of various factors, including thermal Funding Information niches, food discovery rates, and food removal rates, to historical species coexis- RRD and NJS were supported by DOE-PER tence. -

NEST STRUCTURE and FLIGHTS of the ANT Formica Obscuriventris Mayr
NEST STRUCTURE AND FLIGHTS OF THE ANT Formica obscuriventris Mayr BY MARY TALBOT The Edwin S. George Reserve, University of Michigan Nineteen colonies of Formica obscuriventris and Panicum sphaerocarpon Ell.) with scattered were discovered between 1951 and 1962 in the forbs (Verbascum thapsus L., Asclepias syriaca two square miles of the Edwin S . George Reserve L., Circium arvense (L.) Scop.). Oaks of the in southern Michigan. This paper describes flight nearby woods held a thick moist layer of fallen activities of males and females, atmospheric leaves, but on the sunny slope the leaves col- conditions which allowed or prevented flights lected abundantly only where held by logs and and the type of nest in which the winged ants their branches . developed and from which they flew. The matted leaf layers piled against the largest Nest Distribution and Structure log were used extensively for housing larvae and pupae. From here they could be moved Most of the 19 colonies were located at places readily to take advantage of fluctuating moisture where woods and fields met ; six were at woods' and heat. Brood was also found within the log, edges, three in small clearings, one at a fence row just under it, and in chambers extending down and two among small clumps of trees in fields. into the soil from 2 to 6 inches . At various Six were in a sandy strip between marsh and times brood was found under leaves along 30 woods, and one was on a ridge of an open oak- feet of the main log. Covered runways extended hickory wood . -

Seed Ecology and Grassland Resilience: the Case Study of Campo Rupestre
Université d’Avignon et des Pays de Universidade Federal de Minas Gerais Vaucluse École doctorale 536 Programa de Pós Graduação em «Sciences et Agrosciences» Biologia Vegetal THESIS presented as a requirement for obtaining a doctorate degree from the Federal University of Minas Gerais and the Avignon Université (Cotutella agreeement) SEED ECOLOGY AND GRASSLAND RESILIENCE: THE CASE STUDY OF CAMPO RUPESTRE ANDRÉ JARDIM ARRUDA Estimated date for thesis defense on 30 August 2019 Grégory MAHY Dr., Professor Reporter Gembloux AgroBio Tech, Université de Liège, Belgique Giselda DURIGAN Dr., Researcher Reporter Instituto Florestal do Estado de São Paulo, Brazil Thierry DUTOIT Dr., Researcher Examiner Avignon Université, France Alessandra FIDELIS Dr., Adjunct Professor, H.D.R. Examiner Universidade Estadual Paulista, Brazil Tatiana CORNELISSEN Dr., Adjunct Professor, H.D.R. Examiner Universidade Federal de Minas Gerais, Brazil Jeronimo SANSEVERO Dr., Adjunct Professor, H.D.R. Examiner Universidade Federal Rural do Rio de Janeiro, Brazil Elise BUISSON Dr., Adjunct Professor, H.D.R. Co-advisor Avignon Université, France Dr., Professor Advisor Fernando SILVEIRA Universidade Federal de Minas Gerais, Brazil This thesis was prepared at the Institut Méditerranéen de Biodiversité et d'Écologie and at the Laboratory of Ecology and Evolution of Tropical Plant I ANDRÉ JARDIM ARRUDA SEED ECOLOGY AND GRASSLAND RESILIENCE: THE CASE STUDY OF CAMPO RUPESTRE Tese apresentada ao Programa de Pós-Graduação em Biologia Vegetal do Departamento de Botânica do Instituto -

Hymenoptera: Formicidae)
SYSTEMATICS Phylogenetic Analysis of Aphaenogaster Supports the Resurrection of Novomessor (Hymenoptera: Formicidae) 1 B. B. DEMARCO AND A. I. COGNATO Department of Entomology, Michigan State University, 288 Farm Lane, East Lansing, MI 48824. Ann. Entomol. Soc. Am. 108(2): 201–210 (2015); DOI: 10.1093/aesa/sau013 ABSTRACT The ant genus Aphaenogaster Mayr is an ecologically diverse group that is common throughout much of North America. Aphaenogaster has a complicated taxonomic history due to variabil- ity of taxonomic characters. Novomessor Emery was previously synonymized with Aphaenogaster, which was justified by the partial mesonotal suture observed in Aphaenogaster ensifera Forel. Previous studies using Bayesian phylogenies with molecular data suggest Aphaenogaster is polyphyletic. Convergent evolution and retention of ancestral similarities are two major factors contributing to nonmonophyly of Aphaenogaster. Based on 42 multistate morphological characters and five genes, we found Novomessor more closely related to Veromessor Forel and that this clade is sister to Aphaenogaster. Our results confirm the validity of Novomessor stat. r. as a separate genus, and it is resurrected based on the combi- nation of new DNA, morphological, behavioral, and ecological data. KEY WORDS Aphaenogaster, Novomessor, phylogenetics, resurrection Introduction phylogenetic analyses resolved Aphaenogaster as polyphyletic, including Messor Forel, 1890 and Sten- The ant genus Aphaenogaster Mayr, 1853 is a speciose amma (Brady et al. 2006, Moreau and Bell 2013). group,whichhasnotbeentaxonomicallyreviewedin Ward (2011) suggested that convergent evolution and over 60 years (Creighton 1950). Aphaenogaster con- retention of ancestral similarities were two major fac- tains 227 worldwide species (Bolton 2006), with 23 tors contributing to polyphyly of Aphaenogaster. valid North American species reduced from 31 original Aphaenogaster taxonomy was further complicated species descriptions. -

I. Studies of Volatile Compounds from Ants Ii
I. STUDIES OF VOLATILE COMPOUNDS FROM ANTS II. DEGRADATION STUDIES AND STRUCTURE PROOF OF CIS, CIS~NEPETALACTONE By DONALD JOHN MCGURK :1.J I Bachelor of Science University of Nebraska Lincoln, Nebraska 1962 Submitted to the faculty of the Graduate College of the Oklahoma State University in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY July~ 1968 OKlAHOMA STATE UNIVERSITY LI BRAF-cY JAN ~O 1969 ,. ,t--•f,. , ; ... • ' , , ........, ...., ~ # ~· . I. STUDIES OF VOLATILE COMPOUNDS FROM ANTS II. DEGRADATION STUDIES AND STRUCTURE PROOF OF CIS, CIS-NEPETALACTONE Thesis Approved: D t2 ~n Dean of the Graduate College 696365 ii ACKNOWLEDGMENTS I am deeply grateful for the encouragement and guidance provided by Dro Eo Jo Eisenbraun during the conduct of this investigationo I wish to thank Dr. W. A. Drew, Dr. Jerry Young, Dr. and Mrs. D. E. Bryan and especially Dr. Ken Vick for the collection, identification, and behavioral testing of ants. Appreciation is extended to Dro George Waller for the use of the mass spectrometer, to Dro Earl Mitchell and Mro Keith Kinneberg for their assistance in taking spectra, and to the National Science Founda tion which supported these studies through grant NSF GB-32750 I particularly wish to thank Dr. Pat Flanagan who provided the nmr spectra and assisted in their interpretation and Mrs. Jennifer Frost who assisted in the ant studies. Finally, I extend my deepest thanks to the graduate students at Oklahoma State who encouraged me to start to write this dissertation. iii TABLE OF CONTENTS Chapter Page PART I, STUDIES.OF VOLATILE COMPOUNDS FROM ANTS I. -
Arkansas Academy of Science
Journal of the CODEN: AKASO ISBN: 0097-4374 ARKANSAS ACADEMY OF SCIENCE VOLUME 61 2007 Library Rate ARKANSAS ACADEMY OF SCIENCE ARKANSAS TECH UNIVERSITY DEPARTMENT OF PHYSICAL SCIENCES 1701 N. BOULDER RUSSELLVILLE. AR 72801-2222 Arkansas Academy ofScience, Dept. of Physical Sciences, Arkansas Tech University PAST PRESIDENTS OF THE ARKANSAS ACADEMY OF SCIENCE Charles Brookover, 1917 C. E. Hoffman, 1959 Paul Sharrah, 1984 Dwight M. Moore, 1932-33, 64 N. D. Buffaloe, 1960 William L. Evans, 1985 Flora Haas, 1934 H. L. Bogan, 1961 Gary Heidt, 1986 H. H. Hyman, 1935 Trumann McEver, 1962 Edmond Bacon, 1987 L. B. Ham, 1936 Robert Shideler, 1963 Gary Tucker, 1988 W. C. Muon, 1937 L. F. Bailey, 1965 David Chittenden, 1989 M. J. McHenry, 1938 James H. Fribourgh, 1966 Richard K. Speairs, Jr. 1990 T. L. Smith, 1939 Howard Moore, 1967 Robert Watson, 1991 P. G. Horton, 1940 John J. Chapman, 1968 Michael W. Rapp, 1992 I. A. Willis, 1941-42 Arthur Fry, 1969 Arthur A. Johnson, 1993 L. B. Roberts, 1943-44 M. L. Lawson, 1970 George Harp, 1994 JeffBanks, 1945 R. T. Kirkwood, 1971 James Peck, 1995 H. L. Winburn, 1946-47 George E. Templeton, 1972 Peggy R. Dorris, 1996 E. A. Provine, 1948 E. B. Wittlake, 1973 Richard Kluender, 1997 G. V. Robinette, 1949 Clark McCarty, 1974 James Daly, 1998 John R. Totter, 1950 Edward Dale, 1975 Rose McConnell, 1999 R. H. Austin, 1951 Joe Guenter, 1976 Mostafa Hemmati, 2000 E. A. Spessard, 1952 Jewel Moore, 1977 Mark Draganjac, 2001 Delbert Swartz, 1953 Joe Nix, 1978 John Rickett, 2002 Z. -

Download Download
Behavioral Ecology Symposium ’96: Cushing 165 MYRMECOMORPHY AND MYRMECOPHILY IN SPIDERS: A REVIEW PAULA E. CUSHING The College of Wooster Biology Department 931 College Street Wooster, Ohio 44691 ABSTRACT Myrmecomorphs are arthropods that have evolved a morphological resemblance to ants. Myrmecophiles are arthropods that live in or near ant nests and are considered true symbionts. The literature and natural history information about spider myrme- comorphs and myrmecophiles are reviewed. Myrmecomorphy in spiders is generally considered a type of Batesian mimicry in which spiders are gaining protection from predators through their resemblance to aggressive or unpalatable ants. Selection pressure from spider predators and eggsac parasites may trigger greater integration into ant colonies among myrmecophilic spiders. Key Words: Araneae, symbiont, ant-mimicry, ant-associates RESUMEN Los mirmecomorfos son artrópodos que han evolucionado desarrollando una seme- janza morfológica a las hormigas. Los Myrmecófilos son artrópodos que viven dentro o cerca de nidos de hormigas y se consideran verdaderos simbiontes. Ha sido evaluado la literatura e información de historia natural acerca de las arañas mirmecomorfas y mirmecófilas . El myrmecomorfismo en las arañas es generalmente considerado un tipo de mimetismo Batesiano en el cual las arañas están protegiéndose de sus depre- dadores a través de su semejanza con hormigas agresivas o no apetecibles. La presión de selección de los depredadores de arañas y de parásitos de su saco ovopositor pueden inducir una mayor integración de las arañas mirmecófílas hacia las colonias de hor- migas. Myrmecomorphs and myrmecophiles are arthropods that have evolved some level of association with ants. Myrmecomorphs were originally referred to as myrmecoids by Donisthorpe (1927) and are defined as arthropods that mimic ants morphologically and/or behaviorally. -

Influence of Small-Scale Disturbances by Kangaroo Rats on Chihuahuan Desert Ants
Oecologia (2000) 125:142–149 © Springer-Verlag 2000 R.L. Schooley · B.T. Bestelmeyer · J.F. Kelly Influence of small-scale disturbances by kangaroo rats on Chihuahuan Desert ants Received: 11 October 1999 / Accepted: 11 March 2000 Abstract Banner-tailed kangaroo rats (Dipodomys Introduction spectabilis) are prominent ecosystem engineers that build large mounds that influence the spatial structuring Natural disturbances create environmental patchiness of fungi, plants, and some ground-dwelling animals. that can strongly influence organisms and ecological Ants are diverse and functionally important components processes (Watt 1947; Wiens 1976; Pickett and White of arid ecosystems; some species are also ecosystem en- 1985; Turner 1987). In particular, patch disturbances can gineers. We investigated the effects of patch disturbances affect patterns of species richness and community struc- created by D. spectabilis mounds on ant assemblages in ture by increasing habitat heterogeneity and permitting a Chihuahuan Desert grassland in southern New Mexico the coexistence of species with differing competitive and by using pitfall traps in a paired design (mound vs. ma- colonization abilities (Horn and MacArthur 1972; trix). Although the disturbances did not alter species Denslow 1985; Huston 1994). In terrestrial systems, the richness or harbor unique ant communities relative to the disturbances created by the activities of burrowing mam- matrix, they did alter species composition; the abun- mals are important generators of spatial heterogeneity dances of 6 of 26 species were affected. The distur- (Huntley and Inouye 1988; Whicker and Detling 1988; bances might also act to disrupt spatial patterning of ants Hansell 1993). These effects are especially evident in ar- caused by other environmental gradients. -

(Hymenoptera: Formicidae) by Diethe Ortius
A DOLICHODERUS TASCHENBERGI QUEEN FOUND IN A POLYGYNOUS COLONY OF D. PLAGIATUS (HYMENOPTERA: FORMICIDAE) BY DIETHE ORTIUS Theodor-Boveri-Institut LS ftir Verhaltensphysiologie und Soziobiologie Am Hubland, D-97074 Wtirzburg, Germany ABSTRACT Very little is known about colony founding strategies and social organization of the four North American species of the ant genus Dolichoderus. I here report the finding of a Dolichoderus taschen- bergi queen in a colony of D. plagiatus, which suggests parasitic colony founding may occur occasionally in Dolichoderus taschen- bergi. In addition, the colony contained three reproductively active queens of D. plagiatus, indicating that this species is facultatively polygynous. INTRODUCTION The ant genus Dolichoderus Lund is represented by four species in North America (Creighton, 1950): mariae Forel, plagiatus Mayr, pustulatus Mayr, and taschenbergi Mayr. Except for studies by Kannowski (1959, 1967) on the flight activities of these species and a review by Johnson (1989) of their distribution and nest sites, very little is known about the life histories of North American Dolichoderus. Several authors (Wheeler, 1905a; Cole, 1940; Carter, 1962a, b; Wheeler and Wheeler, 1963) have described below-ground nest structures and leaf litter nests for all four North American Dolichoderus. D. plagiatus is considered to be monogy- nous (Kannowski, 1967) and nuptial flights are known to occur between mid June and July (Kannowski, 1959). Among the 12 colonies censused by Kannowski (1967), one colony contained two queens, which, however, he did not dis- sect. The majority of the D. plagiatus colonies investigated by Manuscript received 4 March 1995. 147 148 Psyche [Vol. 102 Kannowski (1967) were monogynous, and the single polygynous colony found was interpreted as either a consequence of pleometrotic colony foundation (primary polygyny) or the adoption of young mated queens (secondary polygyny). -

Zootaxa, Crematogaster Pygmaea (Hymenoptera
Zootaxa 2075: 45–54 (2009) ISSN 1175-5326 (print edition) www.mapress.com/zootaxa/ Article ZOOTAXA Copyright © 2009 · Magnolia Press ISSN 1175-5334 (online edition) Crematogaster pygmaea (Hymenoptera: Formicidae: Myrmicinae), a highly polygynous and polydomous Crematogaster from northeastern Brazil YVES QUINET1, RACHID HAMIDI2, MARIO XAVIER RUIZ-GONZALEZ3, JEAN-CHRISTOPHE de BISEAU4 & JOHN T. LONGINO5 1Laboratório de Entomologia, Instituto Superior de Ciências Biomédicas, Universidade Estadual do Ceará, 60740-000 Fortaleza, Brazil; E-mail: [email protected] 2,4Servide d'Eco-Ethologie Evolutive CP 160/12, Université Libre de Bruxelles, 1050 Bruxelles, Belgium. E-mail: [email protected], [email protected], 3Laboratoire Evolution et Diversité Biologique, UMR-CNRS 5174, Université Paul Sabatier, 31062 Toulouse cedex 9, France. E-mail: [email protected] 5The Evergreen State College, Olympia, Washington 98505, USA. E-mail: [email protected] Abstract Crematogaster pygmaea is revived from synonymy under C. abstinens and newly characterized as a ground-nesting ant from northeastern Brazil. It is a habitat specialist in coastal and tabuleiro zones, where it forms extensive polydomous and polygynous colonies that nest in the soil. Workers forage extensively on honeydew and extrafloral nectar, and foraging continues day and night, although it is depressed at ground temperatures above 32°C. Gyne production occurs at the beginning of the rainy season, while male production starts in the dry season, some months before gyne production. Key words: Brazil, Ceará, Formicidae, Myrmicinae, Crematogaster, polygyny, polydomy Introduction The genus Crematogaster occurs throughout the tropical and subtropical regions of the world, and it is particularly diverse and abundant in the Neotropics.