The Crosstalk Between O-Glcnacylation and Phosphorylation in Insulin Signaling
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Cross-Talk Between Methylation and Phosphorylation in Lymphoid-Specific Helicase Drives Cancer Stem-Like Properties
Signal Transduction and Targeted Therapy www.nature.com/sigtrans ARTICLE OPEN The cross-talk between methylation and phosphorylation in lymphoid-specific helicase drives cancer stem-like properties Na Liu1,2,3, Rui Yang1,2, Ying Shi1,2, Ling Chen1,2, Yating Liu1,2, Zuli Wang1,2, Shouping Liu1,2, Lianlian Ouyang4, Haiyan Wang1,2, Weiwei Lai1,2, Chao Mao1,2, Min Wang1,2, Yan Cheng5, Shuang Liu4, Xiang Wang6, Hu Zhou7, Ya Cao1,2, Desheng Xiao1 and Yongguang Tao1,2,6 Posttranslational modifications (PTMs) of proteins, including chromatin modifiers, play crucial roles in the dynamic alteration of various protein properties and functions including stem-cell properties. However, the roles of Lymphoid-specific helicase (LSH), a DNA methylation modifier, in modulating stem-like properties in cancer are still not clearly clarified. Therefore, exploring PTMs modulation of LSH activity will be of great significance to further understand the function and activity of LSH. Here, we demonstrate that LSH is capable to undergo PTMs, including methylation and phosphorylation. The arginine methyltransferase PRMT5 can methylate LSH at R309 residue, meanwhile, LSH could as well be phosphorylated by MAPK1 kinase at S503 residue. We further show that the accumulation of phosphorylation of LSH at S503 site exhibits downregulation of LSH methylation at R309 residue, which eventually promoting stem-like properties in lung cancer. Whereas, phosphorylation-deficient LSH S503A mutant promotes the accumulation of LSH methylation at R309 residue and attenuates stem-like properties, indicating the critical roles of LSH PTMs in modulating stem-like properties. Thus, our study highlights the importance of the crosstalk between LSH PTMs in determining its activity and function in lung cancer stem-cell maintenance. -

Tyrosine Kinase – Role and Significance in Cancer
Int. J. Med. Sci. 2004 1(2): 101-115 101 International Journal of Medical Sciences ISSN 1449-1907 www.medsci.org 2004 1(2):101-115 ©2004 Ivyspring International Publisher. All rights reserved Review Tyrosine kinase – Role and significance in Cancer Received: 2004.3.30 Accepted: 2004.5.15 Manash K. Paul and Anup K. Mukhopadhyay Published:2004.6.01 Department of Biotechnology, National Institute of Pharmaceutical Education and Research, Sector-67, S.A.S Nagar, Mohali, Punjab, India-160062 Abstract Tyrosine kinases are important mediators of the signaling cascade, determining key roles in diverse biological processes like growth, differentiation, metabolism and apoptosis in response to external and internal stimuli. Recent advances have implicated the role of tyrosine kinases in the pathophysiology of cancer. Though their activity is tightly regulated in normal cells, they may acquire transforming functions due to mutation(s), overexpression and autocrine paracrine stimulation, leading to malignancy. Constitutive oncogenic activation in cancer cells can be blocked by selective tyrosine kinase inhibitors and thus considered as a promising approach for innovative genome based therapeutics. The modes of oncogenic activation and the different approaches for tyrosine kinase inhibition, like small molecule inhibitors, monoclonal antibodies, heat shock proteins, immunoconjugates, antisense and peptide drugs are reviewed in light of the important molecules. As angiogenesis is a major event in cancer growth and proliferation, tyrosine kinase inhibitors as a target for anti-angiogenesis can be aptly applied as a new mode of cancer therapy. The review concludes with a discussion on the application of modern techniques and knowledge of the kinome as means to gear up the tyrosine kinase drug discovery process. -

Amino Acid Building Block Models – in Brief
Amino Acid Building Block Models – In Brief Key Teaching Points for Amino Acid Building Block Models© Overall Student Learning Objective: What Dictates How a Protein Folds? Amino acids are the building blocks of proteins. All amino acids have an identical core structure consisting of an alpha-carbon, carboxyl group, amino group and R-group (sidechain). A linear chain of amino acids is a polypeptide. The primary sequence of a protein is the linear sequence of amino acids in a polypeptide. Proteins are made up of amino acid monomers linked together by peptide bonds. Peptide bond formation between amino acids results in the release of water (dehydration synthesis or condensation reaction). The protein backbone is characterized by the “N-C-C-N-C-C. .” pattern. The “ends” of the protein can be identified by the N-terminus (amino group) end and the C-terminus (carboxyl group) end. For a more complete lesson guide, please visit: http://www.3dmoleculardesigns.com/3DMD-Files/AABB/ContentsandAssembly.pdf Amino Acid Core Structure Build an amino acid according to the diagram to the right: 1. Identify the alpha carbon, amino group, carboxyl group and R-group (sidechain representation) in the structure you have constructed. Two amino acids can be chemically linked by a reaction called “condensation” or “dehydration synthesis” to form a dipeptide bond linking the two amino acids. A chain of amino acid units (monomers) linked together by peptide bonds is called a polypeptide. General Dipeptide Structure Construct a model of a dipeptide using the amino acid models previously built. 2. What are the products of the condensation reaction (dehydration synthesis)? 3. -

Principles of Protein Phosphorylation Biophysical Chemistry 1, Fall 2010
Principles of protein phosphorylation Biophysical Chemistry 1, Fall 2010 Signalling “cascades” Structural biology of phosphorylation Web assignment: http://pkr.genomics.purdue.edu Reversible protein phosphorylation Enzymatic reaction Posttranslational Control ΔG~12kcal/mol Kinases and phosphatases Phosphatase Kinase dephospho- phosphorylates rylates General Examples Signalling overview Cells are way too complex! MAPK/ERK Signaling Pathway RousExample:sarcoma virus Rous (RSV) sarcoma virus (RSV) gag - encodes capsid proteins pol - encodes reverse transcriptase env - encodes envelope proteins src - encodes a tyrosine kinase that attaches phosphate groups to the amino acid tyrosine in host cell proteins MutationsExample:, viruses Rous and cancer sarcoma virus (RSV) v-src lacks the C-terminal inhibitory phosphorylation site (tyrosine-527), and is therefore constitutively active as opposed to normal src (c-src) Continuous cell profileration tumor BiophysicsStructural of signalling Effect of Phosphorylation Phosphorylation is an important regulatory mechanism Can reversible attach/detach a phosphate and therefore switch “on”/”off” the function Effect of phosphorylation is manifold • Conformational change • Ordering/disordering • Electrostatic effects • Alternate binding behavior Signalling by reorientation Reorientation: A conformational switch Rmsd = 2.5Å DHP (red) and Z-Score = 4.6 DHPs74e (blue) (>3.6 same fold) Title: A phosphorylation-induced conformation change in dematin headpiece Author(s): Jiang ZHG, McKnight CJ Source: STRUCTURE Volume: 14 Issue: 2 Pages: 379-387 Published: FEB 2006 Order/disorderDisordering:transitions NtrC, a molecular switch upon phosphorylation Orange-yellow: unphosphorylated NtrC blue-cyan: phosphorylated NtrC Volkman et al., Science 2001, 291, 2429-33 Alternate Binding: SRC SH2 Src/SH2 interactions: binding vs release domain binding ExpectedCan conformationalwe understand effects (predict) the effect of phosphorylation Electrostatics Hydrogen bonding Size Mutational analysis Experimental data Lubman, O.Y. -

Phosphorylation of Chicken Protein Tyrosine Phosphatase 1 by Casein Kinase II in Vitro
EXPERIMENTAL and MOLECULAR MEDICINE, Vol. 29, No 4, 229-233, December 1997 Phosphorylation of chicken protein tyrosine phosphatase 1 by casein kinase II in vitro Eun Joo Jung,1 Kee Ryeon Kang1 and Introduction Yoon-Se Kang1,2 The phosphorylation of protein tyrosine residues is an early event in signal transduction initiated by binding of 1 Department of Biochemistry and Gyeongsang Institute of Cancer growth factors and hormones to their cognate receptors Research, College of Medicine, Gyeongsang National University, and it leads to regulation of cellular activities which include Chinju 660-280, Korea proliferation, differentiation, and also malignant transfor- 2 Corresponding author mation of cells (Hunter, 1989; Ullirich and Schlessinger, Accepted 17 November 1997 1990; Cantley et al., 1991). Under normal conditions, the level of tyrosine phosphorylation within a cell is determined by a balance between the actions of protein tyrosine Abbreviations: CPTP, chicken protein tyrosine phosphatase; HPTP1B, human placenta kinases (PTKs) and protein tyrosine phosphatases (PTPs) protein tyrosine phosphatase 1B; CKII, casein kinase II; MAP kinase, mitogen-activated (Hunter, 1989; Fischer et al., 1991; Trowbridge, 1991). protein kinase; GST, glutathione S-transferase; pNPP, p-nitrophenyl phosphate; EGF, PTPs do not simply reverse the action of tyrosine kinases, epidermal growth factor but rather, PTP itself may play a central role in cellular regulation. PTPs are generally classified as transmem- brane (receptor-type) and nontransmembrane (nonrecep- tor-type) enzymes based on the presence or absence of extracellular and transmembrane portions of their predicted sequence (Fischer et al., 1991). Because the activity of Abstract tyrosine kinase can be controlled by phosphorylation, it has been postulated that PTP activity may be regulated The phosphorylation and dephosphorylation of by phosphorylation as well. -

The Effects of Acute Nicotinamide Riboside Supplementation
THE EFFECTS OF ACUTE NICOTINAMIDE RIBOSIDE SUPPLEMENTATION ON SUBSTRATE UTILISATION AND 5KM TIME-TRIAL PERFORMANCE By ELIZABETH LOUISE GRAY A thesis submitted to The University of Birmingham for the degree of MASTERS BY RESEARCH School of Sport, Exercise and Rehabilitation Sciences College of Life and Environmental Studies University of Birmingham August 2018 University of Birmingham Research Archive e-theses repository This unpublished thesis/dissertation is copyright of the author and/or third parties. The intellectual property rights of the author or third parties in respect of this work are as defined by The Copyright Designs and Patents Act 1988 or as modified by any successor legislation. Any use made of information contained in this thesis/dissertation must be in accordance with that legislation and must be properly acknowledged. Further distribution or reproduction in any format is prohibited without the permission of the copyright holder. ABSTRACT Nicotinamide Riboside (NR) administration has been shown to increase fat oxidation and improve endurance performance in rodents, whilst recent research has proven it is safe for human consumption. The present study aimed to investigate the influence of acute NR supplementation on substrate utilisation and exercise performance in humans. In this counter-balanced, crossover design study, eleven recreationally-active males performed a 60-minute bout of cycling at 55% VO2max, followed by a 5km time-trial. Participants completed this twice during visits separated by at least one week, once following the consumption of 1000mg NR, and the other following placebo consumption. The contribution of fat oxidation to total substrate utilisation was not significantly different between the NR and placebo conditions during steady-state exercise (22.3±9.0% and 19.6±7.3%, respectively; p < 0.05). -

Introduction to Proteins and Amino Acids Introduction
Introduction to Proteins and Amino Acids Introduction • Twenty percent of the human body is made up of proteins. Proteins are the large, complex molecules that are critical for normal functioning of cells. • They are essential for the structure, function, and regulation of the body’s tissues and organs. • Proteins are made up of smaller units called amino acids, which are building blocks of proteins. They are attached to one another by peptide bonds forming a long chain of proteins. Amino acid structure and its classification • An amino acid contains both a carboxylic group and an amino group. Amino acids that have an amino group bonded directly to the alpha-carbon are referred to as alpha amino acids. • Every alpha amino acid has a carbon atom, called an alpha carbon, Cα ; bonded to a carboxylic acid, –COOH group; an amino, –NH2 group; a hydrogen atom; and an R group that is unique for every amino acid. Classification of amino acids • There are 20 amino acids. Based on the nature of their ‘R’ group, they are classified based on their polarity as: Classification based on essentiality: Essential amino acids are the amino acids which you need through your diet because your body cannot make them. Whereas non essential amino acids are the amino acids which are not an essential part of your diet because they can be synthesized by your body. Essential Non essential Histidine Alanine Isoleucine Arginine Leucine Aspargine Methionine Aspartate Phenyl alanine Cystine Threonine Glutamic acid Tryptophan Glycine Valine Ornithine Proline Serine Tyrosine Peptide bonds • Amino acids are linked together by ‘amide groups’ called peptide bonds. -

Citric Acid Cycle
CHEM464 / Medh, J.D. The Citric Acid Cycle Citric Acid Cycle: Central Role in Catabolism • Stage II of catabolism involves the conversion of carbohydrates, fats and aminoacids into acetylCoA • In aerobic organisms, citric acid cycle makes up the final stage of catabolism when acetyl CoA is completely oxidized to CO2. • Also called Krebs cycle or tricarboxylic acid (TCA) cycle. • It is a central integrative pathway that harvests chemical energy from biological fuel in the form of electrons in NADH and FADH2 (oxidation is loss of electrons). • NADH and FADH2 transfer electrons via the electron transport chain to final electron acceptor, O2, to form H2O. Entry of Pyruvate into the TCA cycle • Pyruvate is formed in the cytosol as a product of glycolysis • For entry into the TCA cycle, it has to be converted to Acetyl CoA. • Oxidation of pyruvate to acetyl CoA is catalyzed by the pyruvate dehydrogenase complex in the mitochondria • Mitochondria consist of inner and outer membranes and the matrix • Enzymes of the PDH complex and the TCA cycle (except succinate dehydrogenase) are in the matrix • Pyruvate translocase is an antiporter present in the inner mitochondrial membrane that allows entry of a molecule of pyruvate in exchange for a hydroxide ion. 1 CHEM464 / Medh, J.D. The Citric Acid Cycle The Pyruvate Dehydrogenase (PDH) complex • The PDH complex consists of 3 enzymes. They are: pyruvate dehydrogenase (E1), Dihydrolipoyl transacetylase (E2) and dihydrolipoyl dehydrogenase (E3). • It has 5 cofactors: CoASH, NAD+, lipoamide, TPP and FAD. CoASH and NAD+ participate stoichiometrically in the reaction, the other 3 cofactors have catalytic functions. -
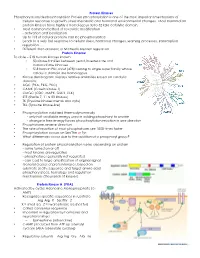
Protein Kinases Phosphorylation/Dephosphorylation Protein Phosphorylation Is One of the Most Important Mechanisms of Cellular Re
Protein Kinases Phosphorylation/dephosphorylation Protein phosphorylation is one of the most important mechanisms of cellular responses to growth, stress metabolic and hormonal environmental changes. Most mammalian protein kinases have highly a homologous 30 to 32 kDa catalytic domain. • Most common method of reversible modification - activation and localization • Up to 1/3 of cellular proteins can be phosphorylated • Leads to a very fast response to cellular stress, hormonal changes, learning processes, transcription regulation .... • Different than allosteric or Michealis Menten regulation Protein Kinome To date – 518 human kinases known • 50 kinase families between yeast, invertebrate and mammaliane kinomes • 518 human PKs, most (478) belong to single super family whose catalytic domain are homologous. • Kinase dendrogram displays relative similarities based on catalytic domains. • AGC (PKA, PKG, PKC) • CAMK (Casein kinase 1) • CMGC (CDC, MAPK, GSK3, CLK) • STE (Sterile 7, 11 & 20 kinases) • TK (Tryosine kinases memb and cyto) • TKL (Tyrosine kinase-like) • Phosphorylation stabilized thermodynamically - only half available energy used in adding phosphoryl to protein - change in free energy forces phosphorylation reaction in one direction • Phosphatases reverse direction • The rate of reaction of most phosphatases are 1000 times faster • Phosphorylation occurs on Ser/The or Tyr • What differences occur due to the addition of a phosphoryl group? • Regulation of protein phosphorylation varies depending on protein - some turned on or off -

Dynamic Metabolic Zonation of the Hepatic Glucose Metabolism Is Accomplished by Sinusoidal Plasma Gradients of Nutrients and Hormones
ORIGINAL RESEARCH published: 12 December 2018 doi: 10.3389/fphys.2018.01786 Dynamic Metabolic Zonation of the Hepatic Glucose Metabolism Is Accomplished by Sinusoidal Plasma Gradients of Nutrients and Hormones Nikolaus Berndt 1,2 and Hermann-Georg Holzhütter 1* 1 Computational Biochemistry Group, Institute of Biochemistry, Charite—University Medicine Berlin, Berlin, Germany, 2 Institute for Computational and Imaging Science in Cardiovascular Medicine, Charite—University Medicine Berlin, Berlin, Germany Being the central metabolic organ of vertebrates, the liver possesses the largest repertoire of metabolic enzymes among all tissues and organs. Almost all metabolic pathways are resident in the parenchymal cell, hepatocyte, but the pathway capacities may largely differ depending on the localization of hepatocytes within the liver acinus-a phenomenon that is commonly referred to as metabolic zonation. Metabolic zonation is rather dynamic since gene expression patterns of metabolic enzymes may change in response to nutrition, Edited by: drugs, hormones and pathological states of the liver (e.g., fibrosis and inflammation). Steven Dooley, Universitätsmedizin Mannheim, This fact has to be ultimately taken into account in mathematical models aiming at Medizinische Fakultät Mannheim, the prediction of metabolic liver functions in different physiological and pathological Universität Heidelberg, Germany settings. Here we present a spatially resolved kinetic tissue model of hepatic glucose Reviewed by: metabolism which includes zone-specific temporal changes of enzyme abundances Adil Mardinoglu, Chalmers University of Technology, which are driven by concentration gradients of nutrients, hormones and oxygen along Sweden the hepatic sinusoids. As key modulators of enzyme expression we included oxygen, Rolf Gebhardt, Leipzig University, Germany glucose and the hormones insulin and glucagon which also control enzyme activities *Correspondence: by cAMP-dependent reversible phosphorylation. -

The Role of Sirtuin 2 Activation by Nicotinamide Phosphoribosyltransferase in the Aberrant Proliferation and Survival of Myeloid Leukemia Cells
Acute Myeloid Leukemia Articles and Brief Reports The role of sirtuin 2 activation by nicotinamide phosphoribosyltransferase in the aberrant proliferation and survival of myeloid leukemia cells Lan Dan, 1,4 Olga Klimenkova, 1 Maxim Klimiankou, 1 Jan-Henning Klusman, 2 Marry M. van den Heuvel-Eibrink, 3 Dirk Reinhardt, 2 Karl Welte, 1 and Julia Skokowa 1 1Department of Molecular Hematopoiesis, Children’s Hospital, Hannover Medical School, Hannover, Germany; 2Department of Pediatric Hematology and Oncology, Children’s Hospital, Hannover Medical School, Hannover, Germany; and 3Department of Pediatric Oncology and Hematology, Erasmus MC-Sophia Children’s Hospital, Rotterdam, The Netherlands; 4Department of Pediatrics, The First Affiliated Hospital of GuangXi Medical University, NanNing, China ABSTRACT Acknowledgments: we thank Background A. Gigina, A. Müller Brechlin Inhibitors of nicotinamide phosphoribosyltransferase have recently been validated as therapeu - and M. Reuter for their excellent tic targets in leukemia, but the mechanism of leukemogenic transformation downstream of this technical assistance. enzyme is unclear. Manuscript received on Design and Methods September 14, 2011. Revised version arrived on November 21, Here, we evaluated whether nicotinamide phosphoribosyltransferase’s effects on aberrant pro - 2011. Manuscript accepted liferation and survival of myeloid leukemic cells are dependent on sirtuin and delineated the on December 19, 2011. downstream signaling pathways operating during this process. Correspondence: Results Karl Welte, Department of We identified significant upregulation of sirtuin 2 and nicotinamide phosphoribosyltransferase Molecular Hematopoiesis, Hannover levels in primary acute myeloid leukemia blasts compared to in hematopoietic progenitor cells Medical School, Carl-Neuberg from healthy individuals. Importantly, specific inhibition of nicotinamide phosphoribosyltrans - Str. 1, 30625 Hannover, ferase or sirtuin 2 significantly reduced proliferation and induced apoptosis in human acute Germany. -

Β-Catenin Signaling Dynamics Regulate Cell Fate in Differentiating Neural Stem Cells
β-Catenin signaling dynamics regulate cell fate in differentiating neural stem cells Alyssa B. Rosenblooma, Marcin Tarczynski a, Nora Lama, Ravi S. Kaneb,1, Lukasz J. Bugaja,c,1, and David V. Schaffera,d,e,f,1 aDepartment of Bioengineering, University of California, Berkeley, CA 94720; bSchool of Chemical & Biomolecular Engineering, Georgia Institute of Technology, Atlanta, GA 30332; cDepartment of Bioengineering, University of Pennsylvania, Philadelphia, PA 19104; dDepartment of Chemical and Biomolecular Engineering, University of California, Berkeley, CA 94720; eDepartment of Molecular and Cell Biology, University of California, Berkeley, CA 94720; and fHelen Wills Neuroscience Institute, University of California, Berkeley, CA 94720 Edited by Randall T. Moon, University of Washington, Seattle, WA, and approved September 21, 2020 (received for review May 4, 2020) Stem cells undergo differentiation in complex and dynamic environ- of how signaling dynamics impact cellular function. Optogenetics ments wherein instructive signals fluctuate on various timescales. has recently emerged as a field in which light—which can readily Thus, cells must be equipped to properly respond to the timing of be varied in intensity, space, and time—is harnessed to precisely signals, for example, to distinguish sustained signaling from transient modulate cell-signaling pathways. In this approach, light-sensitive noise. However, how stem cells respond to dynamic variations in proteins are engineered to interface with specific signaling path- differentiation cues is not well characterized. Here, we use optoge- ways, and the subsequent introduction of such an optogenetic netic activation of β-catenin signaling to probe the dynamic responses construct into cells renders the signaling pathway responsive to of differentiating adult neural stem cells (NSCs).