Impact of Hypertension on Progression and Prognosis in Patients with COVID-19
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Mapping the Accessibility of Medical Facilities of Wuhan During the COVID-19 Pandemic
International Journal of Geo-Information Article Mapping the Accessibility of Medical Facilities of Wuhan during the COVID-19 Pandemic Zhenqi Zhou 1, Zhen Xu 1,* , Anqi Liu 1, Shuang Zhou 1, Lan Mu 2 and Xuan Zhang 2 1 Department of Landscape Architecture, College of Landscape Architecture, Nanjing Forestry University, Nanjing 210037, China; [email protected] (Z.Z.); [email protected] (A.L.); [email protected] (S.Z.) 2 Department of Geography, University of Georgia, Athens, GA 30602, USA; [email protected] (L.M.); [email protected] (X.Z.) * Correspondence: [email protected] Abstract: In December 2019, the coronavirus disease 2019 (COVID-19) pandemic attacked Wuhan, China. The city government soon strictly locked down the city, implemented a hierarchical diagnosis and treatment system, and took a series of unprecedented pharmaceutical and non-pharmaceutical measures. The residents’ access to the medical resources and the consequently potential demand– supply tension may determine effective diagnosis and treatment, for which travel distance and time are key indicators. Using the Application Programming Interface (API) of Baidu Map, we estimated the travel distance and time from communities to the medical facilities capable of treating COVID-19 patients, and we identified the service areas of those facilities as well. The results showed significant differences in service areas and potential loading across medical facilities. The accessibility of medical facilities in the peripheral areas was inferior to those in the central areas; there was spatial inequality of medical resources within and across districts; the amount of community healthcare Citation: Zhou, Z.; Xu, Z.; Liu, A.; Zhou, S.; Mu, L.; Zhang, X. -

Prediction of the Clinical Outcome of COVID-19 Patients Using T Lymphocyte Subsets with 340
medRxiv preprint doi: https://doi.org/10.1101/2020.04.06.20056127; this version posted April 16, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. It is made available under a CC-BY-ND 4.0 International license . Prediction of the clinical outcome of COVID-19 patients using T lymphocyte subsets with 340 cases from Wuhan, China: a retrospective cohort study and a web visualization tool Qibin Liu1, Xuemin Fang1, Shinichi Tokuno3, Ungil Chung4, Xianxiang Chen5, Xiyong Dai6, Xiaoyu Liu7, Feng Xu8, Bing Wang9, Peng Peng* 1 Wuhan Pulmonary Hospital, Wuhan Institute for Tuberculosis Control, No. 28 Baofeng Road, Qiaokou District, Wuhan City, Hubei Province, China; [email protected] 1 Graduate School of Health Innovation, Kanagawa University of Human Services, 2F Bldg.2-A, 3-25-10, Tonomachi Kawasaki-ku, Kawasaki City, Kanagawa 210-0821, Japan; [email protected] 3 Graduate School of Health Innovation, Kanagawa University of Human Services, 2F Bldg.2-A, 3-25-10, Tonomachi Kawasaki-ku, Kawasaki City, Kanagawa 210-0821, Japan; [email protected] 4 Graduate School of Health Innovation, Kanagawa University of Human Services, 2F Bldg.2-A, 3-25-10, Tonomachi Kawasaki-ku, Kawasaki City, Kanagawa 210-0821, Japan; [email protected] 5 Wuhan Pulmonary Hospital, Wuhan Institute for Tuberculosis Control, No. 28 Baofeng Road, Qiaokou District, Wuhan City, Hubei Province, China; [email protected] NOTE: This preprint reports new research that has not been certified by peer review and should not be used to guide clinical practice. -

Here You Can Taste Wuhan Featured Food
Contents Basic Mandarin Chinese Words and Phrases............................................... 2 Useful Sayings....................................................................................... 2 In Restaurants....................................................................................... 3 Numbers................................................................................................3 Dinning and cafes..........................................................................................5 Eating Out in Wuhan.............................................................................5 List of Restaurants and Food Streets (sort by distance).......................6 4 Places Where You Can Taste Wuhan Featured Food.........................7 Restaurants and cafes in Walking Distance........................................11 1 Basic Mandarin Chinese Words and Phrases Useful Sayings nǐ hǎo Hello 你 好 knee how zài jiàn Goodbye 再 见 zi gee’en xiè xiè Thank You 谢 谢! sheh sheh bú yòng le, xiè xiè No, thanks. 不 用 了,谢 谢 boo yong la, sheh sheh bú yòng xiè You are welcome. 不 用 谢 boo yong sheh wǒ jiào… My name is… 我 叫… wore jeow… shì Yes 是 shr bú shì No 不 是 boo shr hǎo Good 好 how bù hǎo Bad 不 好 boo how duì bù qǐ Excuse Me 对 不 起 dway boo chee wǒ tīng bù dǒng I do not understand 我 听 不 懂 wore ting boo dong duō shǎo qián How much? 多 少 钱? dor sheow chen Where is the xǐ shǒu jiān zài nǎ lǐ See-sow-jian zai na-lee washroom? 洗 手 间 在 哪 里 2 In Restaurants In China, many people call a male waiter as handsome guy and a female waitress as beautiful girl. It is also common to call “fú wù yuan” for waiters of both genders. cài dān Menu 菜 单 tsai dan shuài gē Waiter(Handsome) 帅 哥 shuai ge měi nǚ Waitress(Beautiful) 美 女 may nyu fú wù yuán Waiter/Waitress 服 务 员 fu woo yuan wǒ xiǎng yào zhè ge 我 I would like this. -

Ill-Health and Ill Treatment of Gong Shengliang
AI Index: ASA 17/008/2005 Distrib: PG/SC To: Health Professionals From: Health and Human Rights Team Date: 17 March 2005 MEDICAL ACTION CHINA: Ill-health and ill treatment of Gong Shengliang Amnesty International (AI) is seriously concerned for the life and well being of Gong Shengliang, a Protestant pastor and leader of the banned "Huanan (South China) Church", who is imprisoned in Hongshan prison, Hubei Province. Gong Shengliang's latest health setbacks are compounded by the continual ill-treatment he has received since his imprisonment in 2001. During his imprisonment, Gong Shengliang has suffered continual ill-treatment. According to letters that he wrote in the first two weeks of December 2004, which have only recently been published on the internet, Gong Shengliang describes how he was suffering from a debilitating stomach complaint, and how he had been able to only take a little food and water for at least 11 days. Gong Shengliang had been treated at the prison hospital five times and on 6 December 2004 asked to be admitted to a hospital outside the prison. However, this request was refused by the prison authorities, who, on one occasion, threatened Gong Shengliang with what were described as "heavy-handed measures". The prison authorities later agreed to Gong Shengliang’s request and he then received treatment at a provincial hospital and an army hospital, following which he was diagnosed with a duodenal ulcer. Throughout this time, Gong Shengliang had been refused visits from his family. Background information Gong Shengliang -

CHINA VANKE CO., LTD.* 萬科企業股份有限公司 (A Joint Stock Company Incorporated in the People’S Republic of China with Limited Liability) (Stock Code: 2202)
Hong Kong Exchanges and Clearing Limited and The Stock Exchange of Hong Kong Limited take no responsibility for the contents of this announcement, make no representation as to its accuracy or completeness and expressly disclaim any liability whatsoever for any loss howsoever arising from or in reliance upon the whole or any part of the contents of this announcement. CHINA VANKE CO., LTD.* 萬科企業股份有限公司 (A joint stock company incorporated in the People’s Republic of China with limited liability) (Stock Code: 2202) 2019 ANNUAL RESULTS ANNOUNCEMENT The board of directors (the “Board”) of China Vanke Co., Ltd.* (the “Company”) is pleased to announce the audited results of the Company and its subsidiaries for the year ended 31 December 2019. This announcement, containing the full text of the 2019 Annual Report of the Company, complies with the relevant requirements of the Rules Governing the Listing of Securities on The Stock Exchange of Hong Kong Limited in relation to information to accompany preliminary announcement of annual results. Printed version of the Company’s 2019 Annual Report will be delivered to the H-Share Holders of the Company and available for viewing on the websites of The Stock Exchange of Hong Kong Limited (www.hkexnews.hk) and of the Company (www.vanke.com) in April 2020. Both the Chinese and English versions of this results announcement are available on the websites of the Company (www.vanke.com) and The Stock Exchange of Hong Kong Limited (www.hkexnews.hk). In the event of any discrepancies in interpretations between the English version and Chinese version, the Chinese version shall prevail, except for the financial report prepared in accordance with International Financial Reporting Standards, of which the English version shall prevail. -

The People's Liberation Army's 37 Academic Institutions the People's
The People’s Liberation Army’s 37 Academic Institutions Kenneth Allen • Mingzhi Chen Printed in the United States of America by the China Aerospace Studies Institute ISBN: 9798635621417 To request additional copies, please direct inquiries to Director, China Aerospace Studies Institute, Air University, 55 Lemay Plaza, Montgomery, AL 36112 Design by Heisey-Grove Design All photos licensed under the Creative Commons Attribution-Share Alike 4.0 International license, or under the Fair Use Doctrine under Section 107 of the Copyright Act for nonprofit educational and noncommercial use. All other graphics created by or for China Aerospace Studies Institute E-mail: [email protected] Web: http://www.airuniversity.af.mil/CASI Twitter: https://twitter.com/CASI_Research | @CASI_Research Facebook: https://www.facebook.com/CASI.Research.Org LinkedIn: https://www.linkedin.com/company/11049011 Disclaimer The views expressed in this academic research paper are those of the authors and do not necessarily reflect the official policy or position of the U.S. Government or the Department of Defense. In accordance with Air Force Instruction 51-303, Intellectual Property, Patents, Patent Related Matters, Trademarks and Copyrights; this work is the property of the U.S. Government. Limited Print and Electronic Distribution Rights Reproduction and printing is subject to the Copyright Act of 1976 and applicable treaties of the United States. This document and trademark(s) contained herein are protected by law. This publication is provided for noncommercial use only. Unauthorized posting of this publication online is prohibited. Permission is given to duplicate this document for personal, academic, or governmental use only, as long as it is unaltered and complete however, it is requested that reproductions credit the author and China Aerospace Studies Institute (CASI). -

Infection in Wuhan, China
DR. WEI GUO (Orcid ID : 0000-0001-8686-3779) Article type : Letter to the Editor Patterns of HIV and SARS-CoV-2 co-infection in Wuhan, China Wei Guo1,2*, Fangzhao Ming3*, Yong Feng4*, Qian Zhang5, Pingzhen Mo6, Lian Liu7, Ming Gao8, Weiming Tang9§ and Ke Liang6§ 1. Department of Pathology, Zhongnan Hospital of Wuhan University, Wuhan, China 2. Department of Pathology, School of Basic Medical Sciences, Wuhan University, Wuhan, China 3. Wuchang District Center for Disease Control and Prevention,Wuhan, China 4. State Key Laboratory of Virology/Department of Medical Microbiology, School of Basic Medical Sciences, Wuhan University, Wuhan, China 5. Qingshan District Center for Disease Control and Prevention,Wuhan, China 6. Department of Infectious Diseases, Zhongnan Hospital of Wuhan University, Wuhan University, Hubei, China 7. Caidian District Center for Disease Control and Prevention, Wuhan, China 8. Xinzhou District Center for Disease Control and Prevention, Wuhan, China 9. Dermatology Hospital, Southern Medical University, and the University of North Carolina at Chapel Hill Project-China, Guangzhou, China * These authors equally contributed to this manuscript and are co-first authors. This article has been accepted for publication and undergone full peer review but has not been through the copyediting, typesetting, pagination and proofreading process, which may lead to differencesAccepted Article between this version and the Version of Record. Please cite this article as doi: 10.1002/jia2.25568 This article is protected by copyright. All rights reserved § These authors contributed equally. Correspondence to: Dr. Ke Liang, Department of Infectious Diseases Zhongnan Hospital in Wuhan University, Wuhan, 430071, China; Email: [email protected] Keywords: Clinical patterns; HIV; SARS-CoV-2; Co-infection; COVID-19; CD4+ T lymphocyte count; lymphopenia Accepted Article This article is protected by copyright. -

High Speed Rail: Wuhan Urban Garden 5-Day Trip
High Speed Rail: Wuhan Urban Garden 5-Day Trip Day 1 Itinerary Suggested Transportation Hong Kong → Wuhan High Speed Rail [Hong Kong West Kowloon Station → Wuhan Railway Station] To hotel: Recommend to stay in a hotel by the river in Wuchang District. Metro: From Wuhan Railway Hotel for reference: Station, take Metro Line 4 The Westin Wuhan Wuchang Hotel towards Huangjinkou. Address: 96 Linjiang Boulevard, Wuchang District, Wuhan Change to Line 2 at Hongshan Square Station towards Tianhe International Airport. Get off at Jiyuqiao Station and walk for about 7 minutes. (Total travel time about 46 minutes) Taxi: About 35 minutes. Enjoy lunch near the hotel On foot: Walk for about 5 minutes Restaurant for reference: Zhen Bafang Hot Pot from the hotel. Address: No. 43 & 44, Building 12-13, Qianjin Road, Wanda Plaza, Jiyu Bridge, Wuchang District, Wuhan Stand the Test of Time: Yellow Crane Tower Bus: Walk for about 4 minutes from the restaurant to Jiyuqiao Metro Station. Take bus 804 towards Nanhu Road Jiangnan Village. Get off at Yue Ma Chang Station and walk for about 6 minutes. (Total travel time about 37 minutes) Taxi: About 15 minutes. Known as “The No. 1 Tower in the World”, the Yellow Crane Tower is a landmark for Wuhan City and Hubei Province and a must-see attraction. The tower was built in the Three Kingdoms era and was named after its erection on Huangjiji, a submerged rock. Well-known ancient characters such as Li Bai, Bai Juyi, Lu You and Yue Fei had all referenced the tower in their poetry works. -

Download Article (PDF)
Advances in Social Science, Education and Humanities Research, volume 505 6th International Conference on Social Science and Higher Education (ICSSHE 2020) Exploration and Reflection on the Library Emergency Services of Colleges and Universities under COVID-19 —An Investigation on Anti-Epidemic Service in Hubei Colleges and Universities Library Lang Chen1 and Chi Zhang1,* 1Huazhong University of Science and Technology Library, Wuhan, 430074, China * Corresponding author. Email: [email protected] ABSTRACT In the course of novel coronavirus pneumonia (COVID-19) epidemic, the library service guarantee measures in colleges and universities were summarized and analyzed by investigating the official websites and WeChat public accounts of 32 public colleges and universities in Hubei Province. This work reflected on colleges and universities library service work in the special period of epidemic from three aspects and tried to put forward reasonable suggestions. It is expected to offer references and thinking for colleges and universities library emergency services when facing major public health emergencies. Keywords: Novel coronavirus pneumonia; Colleges and universities library; Service; Research; Electronic resources of provincial working committee. They quickly organized forces to donate more than 10000 copies of literature, art 1. INTRODUCTION and other leisure books to the shelter hospitals and From the end of 2019 to the beginning of 2020, the isolation points in Wuchang District and Qiaokou District, outbreak of COVID-19 was concentrated in Wuhan and and offered free electronic books, audio books and other spread rapidly across the country. This is a serious online reading resources. Also, they offer spiritual food for infectious disease which broke out again after SARS in COVID-19 patients and greatly encourage patients' morale. -
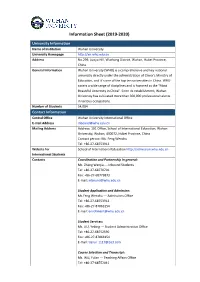
Information Sheet (2019-2020)
Information Sheet (2019-2020) University Information Name of Institution Wuhan University University Homepage http://en.whu.edu.cn Address No.299, Luojia Hill, Wuchang District, Wuhan, Hubei Province, China General Information Wuhan University (WHU) is a comprehensive and key national university directly under the administration of China’s Ministry of Education, and it’s one of the top ten universities in China. WHU covers a wide range of disciplines and is honored as the "Most Beautiful University in China". Since its establishment, Wuhan University has cultivated more than 300,000 professional talents in various occupations. Number of Students 54,084 Contact Information Central Office Wuhan University International Office E-mail Address [email protected] Mailing Address Address: 101 Office, School of International Education, Wuhan University, Wuhan, 430072, Hubei Province, China Contact person: Ms. Feng Wenzhu Tel: +86-27-68753912 Website for School of International Education http://admission.whu.edu.cn International Students Contacts Coordination and Partnership in general: Ms. Zhang Wenjia --- Inbound Students Tel: +86-27-68770701 Fax: +86-27-68779873 E-mail: [email protected] Student Application and Admission: Ms.Feng Wenzhu --- Admissions Office Tel: +86-27-68753912 Fax: +86-27-87863154 E-mail: [email protected] Student Services: Ms. LIU, Yebing --- Student Administration Office Tel: +86-27-68752590 Fax: +86-27-87863154 E-mail: [email protected] Course Selection and Transcript: Ms. WU, Yulan --- Teaching Affairs Office Tel: +86-27-68752041 E-mail: [email protected] International Students School of International Education Management Please note: There are two sections involved in dealing with exchange student affairs in WHU—the office of international affairs and the School of International Education (SIE). -

Effects of Improving Primary Health Care Workers' Knowledge About Public Health Services in Rural China: a Comparative Study of Blended Learning and Pure E-Learning
JOURNAL OF MEDICAL INTERNET RESEARCH Zhan et al Original Paper Effects of Improving Primary Health Care Workers' Knowledge About Public Health Services in Rural China: A Comparative Study of Blended Learning and Pure E-Learning Xingxin Zhan, PhD; Zhixia Zhang, PhD; Fang Sun, MPH; Qian Liu, MMSc; Weijun Peng, MMSc; Heng Zhang, MMSc; Weirong Yan, PhD Department of Epidemiology and Biostatistics, School of Public Health, Tongji Medical College, Huazhong University of Science & Technology, Wuhan, China Corresponding Author: Weirong Yan, PhD Department of Epidemiology and Biostatistics School of Public Health Tongji Medical College, Huazhong University of Science & Technology No.13 Hangkong Road Qiaokou District Wuhan, 430030 China Phone: 86 (027)83650713 Fax: 86 (027)83693763 Email: [email protected] Abstract Background: Primary health care workers (PHCWs) are a major force in delivering basic public health services (BPHS) in rural China. It is necessary to take effective training approaches to improve PHCWs' competency on BPHS. Both electronic learning (e-learning) and blended learning have been widely used in the health workers' education. However, there is limited evidence on the effects of blended learning in comparison with pure e-learning. Objective: The aim of this study was to evaluate the effects of a blended-learning approach for rural PHCWs in improving their knowledge about BPHS as well as training satisfaction in comparison with a pure e-learning approach. Methods: The study was conducted among PHCWs in 6 rural counties of Hubei Province, China, between August 2013 and April 2014. Three counties were randomly allocated blended-learning courses (29 township centers or 612 PHCWsÐthe experimental group), and three counties were allocated pure e-learning courses (31 township centers or 625 PHCWsÐthe control group). -

A Brief Evolutionary History of TCM Culture in Hubei— from the Pre
2017 3rd Annual International Conference on Modern Education and Social Science (MESS 2017) ISBN: 978-1-60595-450-9 A Brief Evolutionary History of TCM Culture in Hubei— From the Pre-Qin Period to the Qing Dynasty Ming-An HUANG1,a, He-Rong MAO2,b*, Hui WEN3,c and Chong WEN4,d 1Center for TCM Development and Research, Hubei University of TCM, No. 1, West Huang Jiahu Road, Hongshan District, Wuhan City, Hubei Province, 430065, PRC. 2*Center for Compilation and Translation of TCM Literature, School of Foreign Languages, Hubei University of TCM, No. 1, West Huang Jiahu Road, Hongshan District, Wuhan City, Hubei Province, 430065, PRC. 3Department of Finance/Center for Military Medicine, Academy of Military Economics, No.122, Luo Jiadun, Qiaokou District, Wuhan City, Hubei Province, 430035, PRC. 4Department of TCM Rehabilitation, the Central Hospital of Wuhan, No. 26, Shengli Street, Jiang’an District, Wuhan City, Hubei Province, 430014, PRC. [email protected],[email protected]* [email protected],[email protected] *He-Rong MAO, Center for Compilation and Translation of TCM Literature, School of Foreign Languages, Hubei University of TCM, No. 1, West Huang Jiahu Road, Hongshan District, Wuhan City, Hubei Province, 430065, PRC. Telephone: +86-13986187098, E-mail: [email protected] Keywords: Evolutionary History, TCM Culture, Hubei Abstract: This paper tries to analyze the rules of evolution and development of TCM culture in Hubei in history. By exploring the rich materials from the authentic history books and local annals and interpreting the great medical thoughts of representative TCM masters lived in Hubei, we find that TCM culture has been well inherited and carried forward in Hubei and Hubei enjoys a respected position in the holistic development of TCM culture in China.