Emerging Applications of Metabolomic and Genomic Profiling in Diabetic Clinical Medicine
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Profiling Data
Compound Name DiscoveRx Gene Symbol Entrez Gene Percent Compound Symbol Control Concentration (nM) JNK-IN-8 AAK1 AAK1 69 1000 JNK-IN-8 ABL1(E255K)-phosphorylated ABL1 100 1000 JNK-IN-8 ABL1(F317I)-nonphosphorylated ABL1 87 1000 JNK-IN-8 ABL1(F317I)-phosphorylated ABL1 100 1000 JNK-IN-8 ABL1(F317L)-nonphosphorylated ABL1 65 1000 JNK-IN-8 ABL1(F317L)-phosphorylated ABL1 61 1000 JNK-IN-8 ABL1(H396P)-nonphosphorylated ABL1 42 1000 JNK-IN-8 ABL1(H396P)-phosphorylated ABL1 60 1000 JNK-IN-8 ABL1(M351T)-phosphorylated ABL1 81 1000 JNK-IN-8 ABL1(Q252H)-nonphosphorylated ABL1 100 1000 JNK-IN-8 ABL1(Q252H)-phosphorylated ABL1 56 1000 JNK-IN-8 ABL1(T315I)-nonphosphorylated ABL1 100 1000 JNK-IN-8 ABL1(T315I)-phosphorylated ABL1 92 1000 JNK-IN-8 ABL1(Y253F)-phosphorylated ABL1 71 1000 JNK-IN-8 ABL1-nonphosphorylated ABL1 97 1000 JNK-IN-8 ABL1-phosphorylated ABL1 100 1000 JNK-IN-8 ABL2 ABL2 97 1000 JNK-IN-8 ACVR1 ACVR1 100 1000 JNK-IN-8 ACVR1B ACVR1B 88 1000 JNK-IN-8 ACVR2A ACVR2A 100 1000 JNK-IN-8 ACVR2B ACVR2B 100 1000 JNK-IN-8 ACVRL1 ACVRL1 96 1000 JNK-IN-8 ADCK3 CABC1 100 1000 JNK-IN-8 ADCK4 ADCK4 93 1000 JNK-IN-8 AKT1 AKT1 100 1000 JNK-IN-8 AKT2 AKT2 100 1000 JNK-IN-8 AKT3 AKT3 100 1000 JNK-IN-8 ALK ALK 85 1000 JNK-IN-8 AMPK-alpha1 PRKAA1 100 1000 JNK-IN-8 AMPK-alpha2 PRKAA2 84 1000 JNK-IN-8 ANKK1 ANKK1 75 1000 JNK-IN-8 ARK5 NUAK1 100 1000 JNK-IN-8 ASK1 MAP3K5 100 1000 JNK-IN-8 ASK2 MAP3K6 93 1000 JNK-IN-8 AURKA AURKA 100 1000 JNK-IN-8 AURKA AURKA 84 1000 JNK-IN-8 AURKB AURKB 83 1000 JNK-IN-8 AURKB AURKB 96 1000 JNK-IN-8 AURKC AURKC 95 1000 JNK-IN-8 -

Epstein-Barr Virus Viral Protein Lmp1 Increases Rap1
EPSTEIN-BARR VIRUS VIRAL PROTEIN LMP1 INCREASES RAP1 SUMOYLATION, ENHANCING TELOMERE MAINTENANCE DURING TUMORIGENESIS By WYATT TYLER CRAMBLET B.S., Biology, Gordon State College, 2016 A Thesis Submitted to the Faculty of Mercer University School of Medicine in Partial Fulfillment of the Requirements for the Degree MASTER OF SCIENCE IN BIOMEDICAL SCIENCE Macon, GA 2018 EPSTEIN-BARR VIRUS VIRAL PROTEIN LMP1 INCREASES RAP1 SUMOYLATION, ENHANCING TELOMERE MAINTENANCE DURING TUMORIGENESIS By WYATT TYLER CRAMBLET Approved: ___________________________________________ Date _________ Gretchen L. Bentz, Ph.D. Faculty Advisor for Thesis ___________________________________________ Date _________ Richard O. McCann, Ph.D. Thesis Committee Member ___________________________________________ Date _________ Laura Silo-Suh, Ph.D. Thesis Committee Member ___________________________________________ Date _________ Jon Shuman, Ph.D. Thesis Committee Member ___________________________________________ Date _________ Jean Sumner, MD Dean, Mercer University School of Medicine ACKNOWLEDGEMENTS I would like to thank the National Institutes of Health for providing the funding for this project and Mercer University School of Medicine for the opportunity for discovery and knowledge this experience has provided me. My utmost respect and gratitude goes to Dr. Gretchen Bentz. Along the course of my research Dr. Bentz has always provided what I required to be successful: materials, focus, experience, and understanding. Her guidance at both the lab and the writing desk is what has allowed me to accomplish the greatest achievement of my academic career. I thank Dr. Richard McCann and Dr. Christy Bridges who have been the Directors of the Master of Science Biomedical Sciences Program. Their leadership has been invaluable to my peers and me for becoming the professionals we set out to be. -

A Genome-Wide Association Study of Idiopathic Dilated Cardiomyopathy in African Americans
Journal of Personalized Medicine Article A Genome-Wide Association Study of Idiopathic Dilated Cardiomyopathy in African Americans Huichun Xu 1,* ID , Gerald W. Dorn II 2, Amol Shetty 3, Ankita Parihar 1, Tushar Dave 1, Shawn W. Robinson 4, Stephen S. Gottlieb 4 ID , Mark P. Donahue 5, Gordon F. Tomaselli 6, William E. Kraus 5,7 ID , Braxton D. Mitchell 1,8 and Stephen B. Liggett 9,* 1 Division of Endocrinology, Diabetes and Nutrition, Department of Medicine, University of Maryland School of Medicine, Baltimore, MD 21201, USA; [email protected] (A.P.); [email protected] (T.D.); [email protected] (B.D.M.) 2 Center for Pharmacogenomics, Department of Internal Medicine, Washington University School of Medicine, St. Louis, MO 63110, USA; [email protected] 3 Institute for Genome Sciences, University of Maryland School of Medicine, Baltimore, MD 21201, USA; [email protected] 4 Division of Cardiovascular Medicine, University of Maryland School of Medicine, Baltimore, MD 21201, USA; [email protected] (S.W.R.); [email protected] (S.S.G.) 5 Division of Cardiology, Department of Medicine, Duke University Medical Center, Durham, NC 27708, USA; [email protected] (M.P.D.); [email protected] (W.E.K.) 6 Department of Medicine, Division of Cardiology, Johns Hopkins University, Baltimore, MD 21218, USA; [email protected] 7 Duke Molecular Physiology Institute, Duke University Medical Center, Durham, NC 27701, USA 8 Geriatrics Research and Education Clinical Center, Baltimore Veterans Administration -

1 Kinobead Profiling Reveals Reprogramming of B-Cell Receptor Signaling in Response to Therapy Within Primary CLL Cells. Linley
bioRxiv preprint doi: https://doi.org/10.1101/841312; this version posted November 14, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Kinobead profiling reveals reprogramming of B-cell receptor signaling in response to therapy within primary CLL cells. Linley AJ1, Griffin R2, Cicconi S2, D’Avola A3$, MacEwan DJ4, Pettit AR1, Kalakonda N1, Packham G3, Prior IA5, Slupsky JR1. 1. Department of Molecular and Clinical Cancer Medicine, Institute of Translational Medicine, University of Liverpool, Sherrington Building, Ashton Street, Liverpool, UK. 2. CRUK Clinical Trials Unit, University of Liverpool, Waterhouse Building, Ashton Street, Liverpool. 3. Southampton Cancer Research UK Centre, Cancer Sciences Unit, Faculty of Medicine, University of Southampton, Southampton, UK. 4. Department of Molecular and Clinical Pharmacology, Institute of Translational Medicine, University of Liverpool, Sherrington Building, Ashton Street, Liverpool, UK. 5. Department of Cellular and Molecular Physiology, Institute of Translational Medicine, University of Liverpool, Nuffield Wing, Crown Street, Liverpool, UK. $ Current Address: The Francis Crick Institute, 1 Midland Road, London, UK. Corresponding author: Dr Adam J Linley, Department of Molecular and Clinical Cancer Medicine, Institute of Translational Medicine, University of Liverpool, Sherrington Building, Ashton Street, Liverpool, UK; [email protected]; +44(0)151 794 5310 Running head: Therapy brings about BCR signal changes. Key points 1. sIgM signaling patterns alter following in vivo therapy using either chemoimmunotherapy or ibrutinib. 2. Kinobeads provide a novel method for high-resolution investigation of signaling in primary CLL cells. 1 bioRxiv preprint doi: https://doi.org/10.1101/841312; this version posted November 14, 2019. -

Camk1d Kinase: a Potential Target for Breast Cancer Therapy by Jasmeen Kaur Sethi
CONFIDENTIAL CaMK1D kinase: a potential target for breast cancer therapy by Jasmeen Kaur Sethi A thesis submitted to the University of Birmingham for the degree of MRes in Cancer Sciences. School of Cancer Research College of Medical and Dental Sciences University of Birmingham August 2014 University of Birmingham Research Archive e-theses repository This unpublished thesis/dissertation is copyright of the author and/or third parties. The intellectual property rights of the author or third parties in respect of this work are as defined by The Copyright Designs and Patents Act 1988 or as modified by any successor legislation. Any use made of information contained in this thesis/dissertation must be in accordance with that legislation and must be properly acknowledged. Further distribution or reproduction in any format is prohibited without the permission of the copyright holder. CONFIDENTIAL Abstract Basal like breast cancer (BLBC) is a highly aggressive and invasive subtype of triple negative breast cancer. BLBC patients are offered empirical treatments, which is associated with poor patient prognosis. The recent study of the genomic and transcriptomic background of 2000 breast cancers together with the discovery of a 10p13 chromosomal gain in BLBC suggest that a kinase known as CaMK1D serves as a driver for disease progression. In-house testing of small molecule compounds using structure-based techniques have already exhibited selective inhibition of the CaMK1D oncogenic state locking it in an autoinhibitory state. Here, we developed a cell-based model system for assays required to evaluate these inhibitors in vitro. BLBC cell lines, HCC70 and MDA-MB-231, were stably transformed using lentiviral transfections to express varying CaMK1D protein levels; knockdown, basal and overexpression. -

Site-Specific Identification and Quantitation of Endogenous SUMO Modifications Under Native Conditions
ARTICLE DOI: 10.1038/s41467-017-01271-3 OPEN Site-specific identification and quantitation of endogenous SUMO modifications under native conditions Ryan J. Lumpkin1, Hongbo Gu2, Yiying Zhu2, Marilyn Leonard3, Alla S. Ahmad 1, Karl R. Clauser4, Jesse G. Meyer 1, Eric J. Bennett 3 & Elizabeth A. Komives1 fi fi 1234567890 Small ubiquitin-like modi er (SUMO) modi cation regulates numerous cellular processes. Unlike ubiquitin, detection of endogenous SUMOylated proteins is limited by the lack of naturally occurring protease sites in the C-terminal tail of SUMO proteins. Proteome-wide detection of SUMOylation sites on target proteins typically requires ectopic expression of mutant SUMOs with introduced tryptic sites. Here, we report a method for proteome-wide, site-level detection of endogenous SUMOylation that uses α-lytic protease, WaLP. WaLP digestion of SUMOylated proteins generates peptides containing SUMO-remnant diglycyl- lysine (KGG) at the site of SUMO modification. Using previously developed immuno-affinity isolation of KGG-containing peptides followed by mass spectrometry, we identified 1209 unique endogenous SUMO modification sites. We also demonstrate the impact of protea- some inhibition on ubiquitin and SUMO-modified proteomes using parallel quantitation of ubiquitylated and SUMOylated peptides. This methodological advancement enables deter- mination of endogenous SUMOylated proteins under completely native conditions. 1 Department of Chemistry and Biochemistry, University of California, San Diego, La Jolla, CA 92093-0378, USA. 2 Proteomic Service Group Cell Signaling Technology, 3 Trask Lane, Danvers, MA 01923, USA. 3 Cell and Developmental Biology, University of California, San Diego, La Jolla, CA 92093-0380, USA. 4 Broad Institute of MIT and Harvard, 415 Main St, Cambridge, MA 02142, USA. -

Rare Disruptive Variants in the DISC1 Interactome and Regulome: Association with Cognitive Ability and Schizophrenia
OPEN Molecular Psychiatry (2018) 23, 1270–1277 www.nature.com/mp ORIGINAL ARTICLE Rare disruptive variants in the DISC1 Interactome and Regulome: association with cognitive ability and schizophrenia S Teng1,2,10, PA Thomson3,4,10, S McCarthy1,10, M Kramer1, S Muller1, J Lihm1, S Morris3, DC Soares3, W Hennah5, S Harris3,4, LM Camargo6, V Malkov7, AM McIntosh8, JK Millar3, DH Blackwood8, KL Evans4, IJ Deary4,9, DJ Porteous3,4 and WR McCombie1 Schizophrenia (SCZ), bipolar disorder (BD) and recurrent major depressive disorder (rMDD) are common psychiatric illnesses. All have been associated with lower cognitive ability, and show evidence of genetic overlap and substantial evidence of pleiotropy with cognitive function and neuroticism. Disrupted in schizophrenia 1 (DISC1) protein directly interacts with a large set of proteins (DISC1 Interactome) that are involved in brain development and signaling. Modulation of DISC1 expression alters the expression of a circumscribed set of genes (DISC1 Regulome) that are also implicated in brain biology and disorder. Here we report targeted sequencing of 59 DISC1 Interactome genes and 154 Regulome genes in 654 psychiatric patients and 889 cognitively-phenotyped control subjects, on whom we previously reported evidence for trait association from complete sequencing of the DISC1 locus. Burden analyses of rare and singleton variants predicted to be damaging were performed for psychiatric disorders, cognitive variables and personality traits. The DISC1 Interactome and Regulome showed differential association across the phenotypes tested. After family-wise error correction across all traits (FWERacross), an increased burden of singleton disruptive variants in the Regulome was associated with SCZ (FWERacross P=0.0339). -

Role of Ubiquitin and SUMO in Intracellular Trafficking
Curr. Issues Mol. Biol. (2020) 35: 99-108. Role of Ubiquitin and SUMO in Intracellular Trafcking Maria Sundvall1,2,3* 1Institute of Biomedicine, University of Turku, Turku, Finland. 2Western Cancer Centre FICAN West, Turku University Hospital, Turku, Finland. 3Department of Oncology and Radiotherapy, University of Turku, Turku, Finland. *Correspondence: [email protected] htps://doi.org/10.21775/cimb.035.099 Abstract behaviour. In addition to e.g. chemical groups, Precise location of proteins at a given time within such as phosphate groups, proteins can be modi- a cell is essential to convey specifc signals and fed by small polypeptides, such as ubiquitin and result in a relevant functional outcome. Small SUMO. Ubiquitin and SUMO share a similar ubiquitin-like modifcations, such as ubiquitin three-dimensional structure and are principally and SUMO, represent a delicate and diverse way covalently linked at lysine (K) residues of sub- to transiently regulate intracellular trafcking strate proteins by a similar conjugation pathway events of existing proteins in cells. Trafcking (Hershko et al., 1998; Hay, 2013). Unlike ubiq- of multiple proteins is controlled reversibly by uitin, SUMO is preferentially atached at SUMO ubiquitin and/or SUMO directly or indirectly via consensus sites ΨKxE in substrates Ψ = hydropho- regulation of transport machinery components. bic residue with high preference for I or V, x = any Regulation is dynamic and multilayered, involv- amino acid) under steady state conditions, but ing active crosstalk and interdependence between under stress conditions in particular more non- post-translational modifcations. However, in consensus sites are SUMOylated (Hendriks et al., most cases regulation appears very complex, and 2016). -

Discovery of Potent and Selective MRCK Inhibitors with Therapeutic
Published OnlineFirst January 30, 2018; DOI: 10.1158/0008-5472.CAN-17-2870 Cancer Translational Science Research Discovery of Potent and Selective MRCK Inhibitors with Therapeutic Effect on Skin Cancer Mathieu Unbekandt1, Simone Belshaw2, Justin Bower2, Maeve Clarke2, Jacqueline Cordes2, Diane Crighton2, Daniel R. Croft2, Martin J. Drysdale2, Mathew J. Garnett3, Kathryn Gill2, Christopher Gray2, David A. Greenhalgh4, James A.M. Hall3, Jennifer Konczal2, Sergio Lilla5, Duncan McArthur2, Patricia McConnell2, Laura McDonald2, Lynn McGarry6, Heather McKinnon2, Carol McMenemy4, Mokdad Mezna2, Nicolas A. Morrice5, June Munro1, Gregory Naylor1, Nicola Rath1, Alexander W. Schuttelkopf€ 2, Mairi Sime2, and Michael F. Olson1,7 Abstract The myotonic dystrophy–related Cdc42-binding kinases an autophosphorylation site, enabling development of a phos- MRCKa and MRCKb contribute to the regulation of actin–myosin phorylation-sensitive antibody tool to report on MRCKa status in cytoskeleton organization and dynamics, acting in concert with tumor specimens. In a two-stage chemical carcinogenesis model the Rho-associated coiled-coil kinases ROCK1 and ROCK2. The of murine squamous cell carcinoma, topical treatments reduced absence of highly potent and selective MRCK inhibitors has MRCKa S1003 autophosphorylation and skin papilloma out- resulted in relatively little knowledge of the potential roles of growth. In parallel work, we validated a phospho-selective anti- these kinases in cancer. Here, we report the discovery of the body with the capability to monitor drug pharmacodynamics. azaindole compounds BDP8900 and BDP9066 as potent and Taken together, our findings establish an important oncogenic selective MRCK inhibitors that reduce substrate phosphorylation, role for MRCK in cancer, and they offer an initial preclinical proof leading to morphologic changes in cancer cells along with inhi- of concept for MRCK inhibition as a valid therapeutic strategy. -

Systems Genetics Identifies a Role for Cacna2d1 Regulation in Elevated
Supplementary Dataset 1: Gene set enrichment analysis (GSEA) input Gene Symbol Gene name Score Cacna2d1 calcium channel, voltage-dependent, alpha 2/delta subunit 1 1 Pten phosphatase and tensin homolog (mutated in multiple advanced cancers 1) 0.8113829 Rora RAR-related orphan receptor A 0.7830256 Nfia nuclear factor I/A 0.77796155 Plxna2 plexin A2 0.7689365 Rwdd4a RWD domain containing 4A 0.75700486 Znf608 zinc finger protein 608 0.7334024 Huwe1 HECT, UBA and WWE domain containing 1 0.73015225 Prkg1 protein kinase, cGMP-dependent, type I 0.7254574 Rab39b RAB39B, member RAS oncogene family 0.71763426 St3gal2 ST3 beta-galactoside alpha-2,3-sialyltransferase 2 0.70345706 Lpgat1 lysophosphatidylglycerol acyltransferase 1 0.7029922 potassium voltage-gated channel, shaker-related subfamily, member 1 (episodic ataxia Kcna1 0.7010396 with myokymia) Ppp2ca protein phosphatase 2 (formerly 2A), catalytic subunit, alpha isoform 0.69497764 Maoa monoamine oxidase A 0.6936078 Socs6 suppressor of cytokine signaling 6 0.6922545 Abcc5 ATP-binding cassette, sub-family C (CFTR/MRP), member 5 0.6845195 Hspa4 heat shock 70kDa protein 4 0.6812371 Dctn4 dynactin 4 (p62) 0.68075943 Lats2 LATS, large tumor suppressor, homolog 2 (Drosophila) 0.67761165 Lin7a lin-7 homolog A (C. elegans) 0.673076 Itga9 integrin, alpha 9 0.6707624 Isoc1 isochorismatase domain containing 1 0.6701377 Cyfip2 cytoplasmic FMR1 interacting protein 2 0.6698394 Asip agouti signaling protein, nonagouti homolog (mouse) 0.6682124 Trpm7 transient receptor potential cation channel, subfamily M, -
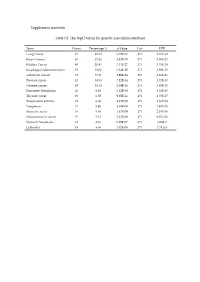
Supplement Materials Table S1. the Top15 Terms for Genetic Association
Supplement materials Table S1. The top15 terms for genetic association database. Term Count Percentage % p-Value List FDR Lung Cancer 73 25.34 6.70E-36 271 1.16E-32 Breast Cancer 67 23.26 2.92E-30 271 5.06E-27 Bladder Cancer 60 20.83 3.33E-27 271 5.78E-24 Esophageal adenocarcinoma 52 18.06 6.24E-29 271 1.08E-25 Colorectal Cancer 51 17.71 4.99E-24 271 8.66E-21 Prostate cancer 42 14.58 7.12E-14 271 1.23E-10 Ovarian cancer 39 13.54 2.04E-16 271 3.89E-13 Pancreatic Neoplasms 20 6.94 1.45E-08 271 2.52E-05 Thyroid cancer 19 6.59 9.98E-11 271 1.73E-07 Rheumatoid arthritis 19 6.60 8.82E-08 271 1.53E-04 Neoplasms 17 5.90 4.58E-08 271 7.94E-05 Stomach cancer 16 5.56 1.17E-09 271 2.03E-06 Head and neck cancer 15 5.21 5.23E-09 271 9.07E-06 Stomach Neoplasms 14 4.86 6.20E-07 271 1.08E-3 Leukemia 14 4.86 1.02E-06 271 1.78 E-3 Table S2. The results of the reverse docking for AutoDock Vina. PDB Protein Gene_Name Uniprot_ID Score 4TVJ Poly [ADP-ribose] polymerase 2 PARP2 Q9UGN5 -10.80 2DQ7 Proto-oncogene tyrosine-protein kinase Fyn FYN P06241 -10.00 4KIK Inhibitor of nuclear factor kappa-B kinase IKBKB O14920 -10.00 subunit beta 3EQR Activated CDC42 kinase 1 ACK1/TNK2 Q07912 -9.90 3MTF Activin receptor type-1 ACVR1 Q04771 -9.90 4GV0 Poly [ADP-ribose] polymerase 3 PARP3 Q9Y6F1 -9.90 1M6I Programmed cell death protein 8 AIF O95831 -9.80 1UA2 Cell division protein kinase 7 CDK7 P50613 -9.80 1Z6T Apoptotic protease activating factor 1 APAF1 O14727 -9.80 1ZXM DNA topoisomerase II TOPII P11388 -9.80 2WOU Serine/Threonine-protein kinase NEK7 NEK7 Q8TDX7 -9.60 -

Full Text (PDF)
medRxiv preprint doi: https://doi.org/10.1101/2020.12.29.20248986; this version posted January 4, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. It is made available under a CC-BY-NC-ND 4.0 International license . Insights into the molecular mechanism of anticancer drug ruxolitinib repurposable in COVID-19 therapy Manisha Mandal Department of Physiology, MGM Medical College, Kishanganj-855107, India Email: [email protected], ORCID: https://orcid.org/0000-0002-9562-5534 Shyamapada Mandal* Department of Zoology, University of Gour Banga, Malda-732103, India Email: [email protected], ORCID: https://orcid.org/0000-0002-9488-3523 *Corresponding author: Email: [email protected]; [email protected] Abstract Due to non-availability of specific therapeutics against COVID-19, repurposing of approved drugs is a reasonable option. Cytokines imbalance in COVID-19 resembles cancer; exploration of anti-inflammatory agents, might reduce COVID-19 mortality. The current study investigates the effect of ruxolitinib treatment in SARS-CoV-2 infected alveolar cells compared to the uninfected one from the GSE5147507 dataset. The protein-protein interaction network, biological process and functional enrichment of differentially expressed genes were studied using STRING App of the Cytoscape software and R programming tools. The present study indicated that ruxolitinib treatment elicited similar response equivalent to that of SARS-CoV-2 uninfected situation by inducing defense response in host against virus infection by RLR and NOD like receptor pathways. Further, the effect of ruxolitinib in SARS- CoV-2 infection was mainly caused by significant suppression of IFIH1, IRF7 and MX1 genes as well as inhibition of DDX58/IFIH1-mediated induction of interferon- I and -II signalling.