COVID-19 Vaccine Hesitancy Guide
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Executive Summary: Under the Surface: Covid-19 Vaccine Narratives, Misinformation and Data Deficits on Social Media
WWW.FIRSTDRAFTNEWS.ORG @FIRSTDRAFTNEWS Executive summary Under the surface: Covid-19 vaccine narratives, misinformation and data deficits on social media AUTHORS: RORY SMITH, SEB CUBBON AND CLAIRE WARDLE CONTRIBUTING RESEARCHERS: JACK BERKEFELD AND TOMMY SHANE WWW.FIRSTDRAFTNEWS.ORG @FIRSTDRAFTNEWS Mapping competing vaccine narratives across English, Spanish and Francophone social media This research demonstrates the complexity of the vaccine Cite this report: Smith, R., Cubbon, S. & Wardle, C. (2020). Under the information ecosystem, where a cacophony of voices and surface: Covid-19 vaccine narratives, misinformation & data deficits on narratives have coalesced to create an environment of extreme social media. First Draft. https://firstdraftnews.org/vaccine- uncertainty. Two topics are driving a large proportion of the narratives-report-summary- november-2020 current global vaccine discourse, especially around a Covid-19 vaccine: the “political and economic motives” of actors and institutions involved in vaccine development and the “safety, efficacy and necessity” concerns around vaccines. Narratives challenging the safety of vaccines have been perennial players in the online vaccine debate. Yet this research shows that narratives related to mistrust in the intentions of institutions and key figures surrounding vaccines are now driving as much of the online conversation and vaccine skepticism as safety concerns. This issue is compounded by the complexities and vulnerabilities of this information ecosystem. It is full of “data deficits” — situations where demand for information about a topic is high, but the supply of credible information is low — that are being exploited by bad actors. These data deficits complicate efforts to accurately make sense of the development of a Covid-19 vaccine and vaccines more generally. -

REALM Research Briefing: Vaccines, Variants, and Venitlation
Briefing: Vaccines, Variants, and Ventilation A Briefing on Recent Scientific Literature Focused on SARS-CoV-2 Vaccines and Variants, Plus the Effects of Ventilation on Virus Spread Dates of Search: 01 January 2021 through 05 July 2021 Published: 22 July 2021 This document synthesizes various studies and data; however, the scientific understanding regarding COVID-19 is continuously evolving. This material is being provided for informational purposes only, and readers are encouraged to review federal, state, tribal, territorial, and local guidance. The authors, sponsors, and researchers are not liable for any damages resulting from use, misuse, or reliance upon this information, or any errors or omissions herein. INTRODUCTION Purpose of This Briefing • Access to the latest scientific research is critical as libraries, archives, and museums (LAMs) work to sustain modified operations during the continuing severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic. • As an emerging event, the SARS-CoV-2 pandemic continually presents new challenges and scientific questions. At present, SARS-CoV-2 vaccines and variants in the US are two critical areas of focus. The effects of ventilation-based interventions on the spread of SARS-CoV-2 are also an interest area for LAMs. This briefing provides key information and results from the latest scientific literature to help inform LAMs making decisions related to these topics. How to Use This Briefing: This briefing is intended to provide timely information about SARS-CoV-2 vaccines, variants, and ventilation to LAMs and their stakeholders. Due to the evolving nature of scientific research on these topics, the information provided here is not intended to be comprehensive or final. -

The Hepatitis B Vaccine Is the Most Widely Used Vaccine in the World, with Over 1 Billion Doses Given
Hepatitis B Vaccine Protect Yourself and Those You Love What is Hepatitis B? Hepatitis B is the most common serious liver infection in the world. It is caused by the hepatitis B virus (HBV), which attacks liver cells and can lead to liver failure, cirrhosis (scarring), or liver cancer later in life. The virus is transmitted through direct contact with infected blood and bodily fluids, and from an infected woman to her newborn at birth. Is there a safe vaccine for hepatitis B? YES! The good news is that there is a safe and effective vaccine for hepatitis B. The vaccine is a series, typically given as three shots over a six-month period that will provide a lifetime of protection. You cannot get hepatitis B from the vaccine – there is no human blood or live virus in the vaccine. The hepatitis B vaccine is the most widely used vaccine in the world, with over 1 billion doses given. The hepatitis B vaccine is the first "anti-cancer" vaccine because it can help prevent liver cancer! Who should be vaccinated against hepatitis B? The World Health Organization (WHO) and the U.S. Centers for Disease Control and Prevention (CDC) recommend the hepatitis B vaccine for all newborns and children up to 18 years of age, and all high-risk adults. All infants should receive the first dose of the vaccine in the delivery room or in the first 24 hours of life, preferably within 12 hours. (CDC recommends the first dose within 12 hours vs. the WHO recommendation of 24 hours.) The HBV vaccine is recommended to anyone who is at high risk of infection. -

Immunizations
Medicine Summer 2015 | Volume 14, Number 2 encephalitis tetanus RUBELLA MUMPS meningococus pertussis ENCEPHALITIS MEASLES PERTUSSIS ROTAVIRUS meningococus MUMPS meningococus MEASLES tuberculosis RUBELLA diphtheria CHICKENPOX meningococus CHICKENPOX pneumococus meningococus rotavirus ENCEPHALITIS PNEUMOCOCUS FEVER TETANUS DIPHTHERIA TETANUS hepatitismumps POLIO encephalitis hepatitis measles pneumococus POLIO DIPHTHERIA FEVER CHICKENPOX tuberculosisRUBELLA tuberculosis diphtheria DIPHTHERIA meningococusMUMPS pneumococus encephalitis ENCEPHALITIS polio TETANUS diphtheria pertussis polio hepatitisRUBELLA meningococus ENCEPHALITIS DIPHTHERIA MEASLES YELLOW RUBELLA TETANUS measles Immunizations MEASLESpolio meningococus PERTUSSIS MEASLES RUBELLA measles tetanus rubella YELLOW tuberculosisENCEPHALITIS pneumococus CHICKENPOX RUBELLA measles What physicians rubella pertussis PNEUMOCOCUS need to know encephalitis diphtheria MUMPS meningococustetanus pneumococus measles rubella tetanus measles rotavirus tetanus rubellaPOLIOmeningococus hepatitisHEPATITISFEVER PERTUSSIS RUBELLA FEVER hepatitis FEVER HEPATITIS chickenpox poliorubella encephalitisTETANUSpertussis rubella DIPHTHERIA rotavirus tuberculosis FEVERHEPATITIS POLIO yellow DIPHTHERIAYELLOW polio pneumococus YELLOW polio ENCEPHALITIS chickenpoxrubellaRUBELLA rotavirus ENCEPHALITIS pertussis ROTAVIRUS tetanus chickenpox hepatitisFEVER encephalitis ENCEPHALITIS POLIO rotavirus ROTAVIRUS DIPHTHERIA YELLOW pertussis PNEUMOCOCUS PERTUSSIS RUBELLA polio CHICKENPOX ROTAVIRUS pneumococus CHICKENPOXCHICKENPOX -

SCIENCE and HEALTH MISINFORMATION in the DIGITAL AGE: How Does It Spread? Who Is Vulnerable? How Do We Fight It?
SCIENCE AND HEALTH MISINFORMATION IN THE DIGITAL AGE: How does it spread? Who is vulnerable? How do we fight it? ERIC MERKLEY & PETER LOEWEN J U LY, 2021 ABOUT PPF Good Policy. Better Canada. The Public Policy Forum builds bridges among diverse participants in the policy-making process and gives them a platform to examine issues, offer new perspectives and feed fresh ideas into critical policy discussions. We believe good policy is critical to making a better Canada—a country that’s cohesive, prosperous and secure. We contribute by: . Conducting research on critical issues . Convening candid dialogues on research subjects . Recognizing exceptional leaders Our approach—called Inclusion to Conclusion—brings emerging and established voices to policy conversations, which informs conclusions that identify obstacles to success and pathways forward. PPF is an independent, non-partisan charity whose members are a diverse group of private, public and non-profit organizations. ppforum.ca @ppforumca © 2021, Public Policy Forum 1400 - 130 Albert Street Ottawa, ON, Canada, K1P 5G4 613.238.7858 ISBN: 978-1-77452-085-7 WITH THANKS TO OUR PARTNERS ABOUT THE AUTHORS ERIC MERKLEY Eric Merkley (PhD, UBC) is an Assistant Professor in the Department of Political Science at the University of Toronto. He was the lead survey analyst of the Media Ecosystem Observatory, which studied the Canadian information ecosystem and public opinion during the COVID-19 pandemic, and the Digital Democracy Project, which studied misinformation and public attitudes during the 2019 Canadian federal election. His research focuses on the link between political elite behaviour, the news media, and public opinion. -

Vaccine Hesitancy
WHY CHILDREN WORKSHOP ON IMMUNIZATIONS ARE NOT VACCINATED? VACCINE HESITANCY José Esparza MD, PhD - Adjunct Professor, Institute of Human Virology, University of Maryland School of Medicine, Baltimore, MD, USA - Robert Koch Fellow, Robert Koch Institute, Berlin, Germany - Senior Advisor, Global Virus Network, Baltimore, MD, USA. Formerly: - Bill & Melinda Gates Foundation, Seattle, WA, USA - World Health Organization, Geneva, Switzerland The value of vaccination “The impact of vaccination on the health of the world’s people is hard to exaggerate. With the exception of safe water, no other modality has had such a major effect on mortality reduction and population growth” Stanley Plotkin (2013) VACCINES VAILABLE TO PROTECT AGAINST MORE DISEASES (US) BASIC VACCINES RECOMMENDED BY WHO For all: BCG, hepatitis B, polio, DTP, Hib, Pneumococcal (conjugated), rotavirus, measles, rubella, HPV. For certain regions: Japanese encephalitis, yellow fever, tick-borne encephalitis. For some high-risk populations: typhoid, cholera, meningococcal, hepatitis A, rabies. For certain immunization programs: mumps, influenza Vaccines save millions of lives annually, worldwide WHAT THE WORLD HAS ACHIEVED: 40 YEARS OF INCREASING REACH OF BASIC VACCINES “Bill Gates Chart” 17 M GAVI 5.6 M 4.2 M Today (ca 2015): <5% of children in GAVI countries fully immunised with the 11 WHO- recommended vaccines Seth Berkley (GAVI) The goal: 50% of children in GAVI countries fully immunised by 2020 Seth Berkley (GAVI) The current world immunization efforts are achieving: • Equity between high and low-income countries • Bringing the power of vaccines to even the world’s poorest countries • Reducing morbidity and mortality in developing countries • Eliminating and eradicating disease WHY CHILDREN ARE NOT VACCINATED? •Vaccines are not available •Deficient health care systems •Poverty •Vaccine hesitancy (reticencia a la vacunacion) VACCINE HESITANCE: WHO DEFINITION “Vaccine hesitancy refers to delay in acceptance or refusal of vaccines despite availability of vaccination services. -
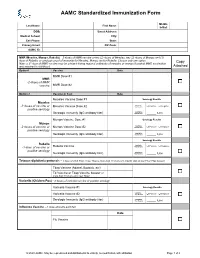
AAMC Standardized Immunization Form
AAMC Standardized Immunization Form Middle Last Name: First Name: Initial: DOB: Street Address: Medical School: City: Cell Phone: State: Primary Email: ZIP Code: AAMC ID: MMR (Measles, Mumps, Rubella) – 2 doses of MMR vaccine or two (2) doses of Measles, two (2) doses of Mumps and (1) dose of Rubella; or serologic proof of immunity for Measles, Mumps and/or Rubella. Choose only one option. Copy Note: a 3rd dose of MMR vaccine may be advised during regional outbreaks of measles or mumps if original MMR vaccination was received in childhood. Attached Option1 Vaccine Date MMR Dose #1 MMR -2 doses of MMR vaccine MMR Dose #2 Option 2 Vaccine or Test Date Measles Vaccine Dose #1 Serology Results Measles Qualitative -2 doses of vaccine or Measles Vaccine Dose #2 Titer Results: Positive Negative positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Mumps Vaccine Dose #1 Serology Results Mumps Qualitative -2 doses of vaccine or Mumps Vaccine Dose #2 Titer Results: Positive Negative positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Serology Results Rubella Qualitative Positive Negative -1 dose of vaccine or Rubella Vaccine Titer Results: positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Tetanus-diphtheria-pertussis – 1 dose of adult Tdap; if last Tdap is more than 10 years old, provide date of last Td or Tdap booster Tdap Vaccine (Adacel, Boostrix, etc) Td Vaccine or Tdap Vaccine booster (if more than 10 years since last Tdap) Varicella (Chicken Pox) - 2 doses of varicella vaccine or positive serology Varicella Vaccine #1 Serology Results Qualitative Varicella Vaccine #2 Titer Results: Positive Negative Serologic Immunity (IgG antibody titer) Quantitative Titer Results: _____ IU/ml Influenza Vaccine --1 dose annually each fall Date Flu Vaccine © 2020 AAMC. -

Hepatitis B Vaccine – Frequently Asked Questions (Information from the CDC)
AAMC Standardized Immunization Form 2020 Hepatitis B Vaccine – Frequently Asked Questions (Information from the CDC) 1. What are the hepatitis B vaccines licensed for use in the United States? Three single-antigen vaccines and two combination vaccines are currently licensed in the United States. Single-antigen hepatitis B vaccines: • ENGERIX-B® • RECOMBIVAX HB® • HEPLISAV-B™ Combination vaccines: • PEDIARIX®: Combined hepatitis B, diphtheria, tetanus, acellular pertussis (DTaP), and inactivated poliovirus (IPV) vaccine. Cannot be administered before age 6 weeks or after age 7 years. • TWINRIX®: Combined Hepatitis A and hepatitis B vaccine. Recommended for people aged ≥18 years who are at increased risk for both HAV and HBV infections. 2. What are the recommended schedules for hepatitis B vaccination? The vaccination schedule most often used for children and adults is three doses given at 0, 1, and 6 months. Alternate schedules have been approved for certain vaccines and/or populations. A new formulation, Heplisav-B (HepB-CpG), is approved to be given as two doses one month apart. 3. If there is an interruption between doses of hepatitis B vaccine, does the vaccine series need to be restarted? No. The series does not need to be restarted but the following should be considered: • If the vaccine series was interrupted after the first dose, the second dose should be administered as soon as possible. • The second and third doses should be separated by an interval of at least 8 weeks. • If only the third dose is delayed, it should be administered as soon as possible. 4. Is it harmful to administer an extra dose of hepatitis B vaccine or to repeat the entire vaccine series if documentation of the vaccination history is unavailable or the serology test is negative? No, administering extra doses of single-antigen hepatitis B vaccine is not harmful. -

COVID-19 Vaccine Hesitancy 3
Article 1 Lack of trust, conspiracy beliefs and social media use predict 2 COVID-19 vaccine hesitancy 3 Will Jennings1, Gerry Stoker1, Hannah Bunting1, Viktor Orri Valgarðsson1, Jen Gaskell1, Daniel Devine2, Lawrence 4 McKay1 and Melinda C. Mills3*. 5 1 University of Southampton, UK. 6 2 St Hilda’s College, University of Oxford, UK. 7 3 Leverhulme Centre for Demographic Science & Nuffield College, University of Oxford, UK. 8 * Correspondence: MM [email protected]. 9 Abstract: As COVID-19 vaccines are rolled out across the world, there are growing concerns about 10 the role that trust, belief in conspiracy theories and spread of misinformation through social media 11 impact vaccine hesitancy. We use a nationally representative survey of 1,476 adults in the UK be- 12 tween December 12 to 18, 2020 and five focus groups conducted in the same period. Trust is a core 13 predictor, with distrust in vaccines in general and mistrust in government raising vaccine hesi- 14 tancy. Trust in health institutions and experts and perceived personal threat are vital, with focus 15 groups revealing that COVID-19 vaccine hesitancy is driven by a misunderstanding of herd im- 16 munity as providing protection, fear of rapid vaccine development and side effects and belief the 17 virus is man-made and used for population control. Particularly those who obtain information 18 from relatively unregulated social media sources such as YouTube that have recommendations 19 tailored by watch history and hold general conspiratorial beliefs are less willing to be vaccinated. 20 Since an increasing number of individuals use social media for gathering health information, in- 21 terventions require action from governments, health officials and social media companies. -

COVID-19 Vaccines Frequently Asked Questions
Page 1 of 12 COVID-19 Vaccines 2020a Frequently Asked Questions Michigan.gov/Coronavirus The information in this document will change frequently as we learn more about COVID-19 vaccines. There is a lot we are learning as the pandemic and COVID-19 vaccines evolve. The approach in Michigan will adapt as we learn more. September 29, 2021. Quick Links What’s new | Why COVID-19 vaccination is important | Booster and additional doses | What to expect when you get vaccinated | Safety of the vaccine | Vaccine distribution/prioritization | Additional vaccine information | Protecting your privacy | Where can I get more information? What’s new − Pfizer booster doses recommended for some people to boost waning immunity six months after completing the Pfizer vaccine. Why COVID-19 vaccination is important − If you are fully vaccinated, you don’t have to quarantine after being exposed to COVID-19, as long as you don’t have symptoms. This means missing less work, school, sports and other activities. − COVID-19 vaccination is the safest way to build protection. COVID-19 is still a threat, especially to people who are unvaccinated. Some people who get COVID-19 can become severely ill, which could result in hospitalization, and some people have ongoing health problems several weeks or even longer after getting infected. Even people who did not have symptoms when they were infected can have these ongoing health problems. − After you are fully vaccinated for COVID-19, you can resume many activities that you did before the pandemic. CDC recommends that fully vaccinated people wear a mask in public indoor settings if they are in an area of substantial or high transmission. -

Immunogenicity and Safety of a Third Dose, and Immune Persistence Of
medRxiv preprint doi: https://doi.org/10.1101/2021.07.23.21261026; this version posted July 25, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. All rights reserved. No reuse allowed without permission. 1 Immunogenicity and safety of a third dose, and immune persistence of 2 CoronaVac vaccine in healthy adults aged 18-59 years: interim results 3 from a double-blind, randomized, placebo-controlled phase 2 clinical 4 trial 5 6 Hongxing Pan MSc1*, Qianhui Wu MPH2*, Gang Zeng Ph.D.3*, Juan Yang Ph.D.1, Deyu 7 Jiang MSc4, Xiaowei Deng MSc2, Kai Chu MSc1, Wen Zheng BSc2, Fengcai Zhu M.D.5†, 8 Hongjie Yu M.D. Ph.D.2,6,7†, Weidong Yin MBA8† 9 10 Affiliations 11 1. Vaccine Evaluation Institute, Jiangsu Provincial Center for Disease Control and 12 Prevention, Nanjing, China 13 2. School of Public Health, Fudan University, Key Laboratory of Public Health Safety, 14 Ministry of Education, Shanghai, China 15 3. Clinical Research Department, Sinovac Biotech Co., Ltd., Beijing, China 16 4. Covid-19 Vaccine Department, Sinovac Life Sciences Co., Ltd., Beijing, China 17 5. Jiangsu Provincial Center for Disease Control and Prevention, Nanjing, China 18 6. Shanghai Institute of Infectious Disease and Biosecurity, Fudan University, 19 Shanghai, China 20 7. Department of Infectious Diseases, Huashan Hospital, Fudan University, 21 Shanghai, China 22 8. Sinovac Biotech Co., Ltd., Beijing, China NOTE: This preprint reports new research that has not been certified by peer review and should not be used to guide clinical practice. -

The Mississippi Covid-19 Vaccine Confidence Survey: Population Results Report
MAY 2021 THE MISSISSIPPI COVID-19 VACCINE CONFIDENCE SURVEY: POPULATION RESULTS Report A collaborative population-based study The Mississippi Community Engagement Alliance Against COVID-19 Disparities (CEAL) Team The Mississippi State Department of Health: Office of Preventive Health and Health Equity EXECUTIVE SUMMARY Since the Spring of 2020, the Novel 2019 Office of Preventive Health and Health Coronavirus (COVID-19) Pandemic has Equity (OPHHE) disseminated a statewide impacted Mississippians of every race, vaccine confidence survey beginning end ethnicity, age, gender, and income bracket. of December 2020 and collecting data Unfortunately, it has disproportionately until March 2021. The survey is intended to impacted Mississippians of color, the elderly, be representative of Mississippians, with and those living with chronic disease. For intentional efforts invested to reach lower most of the past year, the State has worked income and rural Mississippi populations, as to protect its population through preventive well as the state’s Black, Hispanic (Latino/ measures such as social distancing and Latinx), Asian (including the Vietnamese personal protective equipment. However, population of the Gulf Coast), and Native with the release of COVID-19 Vaccines to American/Choctaw communities. The sur- the public, the population of Mississippi has vey was administered in three languages- the opportunity to embrace a long-term English, Spanish, and Vietnamese- through a solution to COVID-19. That is, if Mississippians mixed-modal survey effort, including: web- are willing to receive the vaccine. To based, paper-based, and verbal-oratory assess Mississippians’ COVID-19 Vaccine administration. All targeted populations confidence, the Mississippi Community were ultimately reached and are represented Engagement Alliance Against COVID-19 in the over 11,000 completed responses Disparities (CEAL) Team and the Mississippi from all 82 of Mississippi’s counties.