Immunogenicity and Safety of a Third Dose, and Immune Persistence Of
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

What Do We Know About India's Covaxin Vaccine?
FEATURE Tamil Nadu, India COVID-19 VACCINES [email protected] BMJ: first published as 10.1136/bmj.n997 on 20 April 2021. Downloaded from Cite this as: BMJ 2021;373:n997 http://dx.doi.org/10.1136/bmj.n997 What do we know about India’s Covaxin vaccine? Published: 20 April 2021 India has rapidly approved and rolled out Covaxin, its own covid-19 vaccine. Kamala Thiagarajan examines what we know so far. Kamala Thiagarajan freelance journalist Who developed Covaxin? cheapest purchased by any country in the world at 206 rupees per shot for the 5.5 million doses the Covaxin was developed by Indian pharmaceutical government currently has on order. The government company Bharat Biotech in collaboration with the has capped the price of the vaccine sold in the private Indian Council of Medical Research, a government market, with private hospitals able to charge up to funded biomedical research institute, and its 250 rupees.13 subsidiary the National Institute of Virology. Covaxin does not require storage at sub-zero Bharat Biotech has brought to market 16 original temperatures, which would be hard to maintain in vaccines, including for rotavirus, hepatitis B, Zika India’s climate and with the frequent power cuts in virus, and chikungunya.1 The company reportedly rural areas. Covaxin is available in multi-dose vials spent $60-$70m (£43-£50m; €50-€58m) developing and is stable at the 2-8°C that ordinary refrigeration Covaxin.2 can achieve. How does Covaxin work? Bharat Biotech says it has a stockpile of 20 million The vaccine is similar to CoronaVac (the Chinese doses of Covaxin for India and is in the process of vaccine developed by Sinovac)3 in that it uses a manufacturing 700 million doses at its four facilities complete infective SARS-CoV-2 viral particle in two cities by the end of the year. -

Scientific Committee on Vaccine Preventable Diseases and Scientific Committee on Emerging and Zoonotic Diseases
Scientific Committee on Emerging and Zoonotic Disease and Scientific Committee on Vaccine Preventable Diseases Consensus Interim Recommendations on the Use of COVID-19 Vaccines in Hong Kong (As of Jan 7, 2021) Introduction The ongoing COVID-19 pandemic causes a significant disease burden worldwide. In Hong Kong, cases and outbreaks continue to be reported. To reduce the impacts of COVID-19 on health and society, vaccines against COVID-19 is considered an important public health tool for containing the pandemic in the medium and long term. On 7 January 2021, the Scientific Committee on Emerging and Zoonotic Diseases (SCEZD), the Scientific Committee on Vaccine Preventable Diseases (SCVPD), and the Expert Advisory Panel to Chief Executive (EAP) reviewed the latest scientific evidence on the epidemiology and clinical features of COVID-19, published data on the COVID- 19 vaccines be procured by the Hong Kong SAR Government, local data as well as overseas recommendations/practices, and provides recommendations on the population groups and circumstances for the use of COVID-19 vaccines in Hong Kong. COVID-19 Vaccines 2. At the meeting held on 13 August 2020, the joint SCEZD and SCVPD together with the EAP reviewed the then scientific development of COVID 19 vaccines and prioritization of target groups for COVID 19 vaccines in Hong Kong. The meeting recommended that vaccine procurement would be aimed at the whole Hong Kong population in the long run. In anticipation of a limited supply at the early stage when vaccines are available, a phased approach has to be taken with certain priority groups of the local population identified to be vaccinated first, in order to reduce morbidity and mortality and maintain essential services. -

The Hepatitis B Vaccine Is the Most Widely Used Vaccine in the World, with Over 1 Billion Doses Given
Hepatitis B Vaccine Protect Yourself and Those You Love What is Hepatitis B? Hepatitis B is the most common serious liver infection in the world. It is caused by the hepatitis B virus (HBV), which attacks liver cells and can lead to liver failure, cirrhosis (scarring), or liver cancer later in life. The virus is transmitted through direct contact with infected blood and bodily fluids, and from an infected woman to her newborn at birth. Is there a safe vaccine for hepatitis B? YES! The good news is that there is a safe and effective vaccine for hepatitis B. The vaccine is a series, typically given as three shots over a six-month period that will provide a lifetime of protection. You cannot get hepatitis B from the vaccine – there is no human blood or live virus in the vaccine. The hepatitis B vaccine is the most widely used vaccine in the world, with over 1 billion doses given. The hepatitis B vaccine is the first "anti-cancer" vaccine because it can help prevent liver cancer! Who should be vaccinated against hepatitis B? The World Health Organization (WHO) and the U.S. Centers for Disease Control and Prevention (CDC) recommend the hepatitis B vaccine for all newborns and children up to 18 years of age, and all high-risk adults. All infants should receive the first dose of the vaccine in the delivery room or in the first 24 hours of life, preferably within 12 hours. (CDC recommends the first dose within 12 hours vs. the WHO recommendation of 24 hours.) The HBV vaccine is recommended to anyone who is at high risk of infection. -

COVID-19 Vaccination Programme: Information for Healthcare Practitioners
COVID-19 vaccination programme Information for healthcare practitioners Republished 6 August 2021 Version 3.10 1 COVID-19 vaccination programme: Information for healthcare practitioners Document information This document was originally published provisionally, ahead of authorisation of any COVID-19 vaccine in the UK, to provide information to those involved in the COVID-19 national vaccination programme before it began in December 2020. Following authorisation for temporary supply by the UK Department of Health and Social Care and the Medicines and Healthcare products Regulatory Agency being given to the COVID-19 Vaccine Pfizer BioNTech on 2 December 2020, the COVID-19 Vaccine AstraZeneca on 30 December 2020 and the COVID-19 Vaccine Moderna on 8 January 2021, this document has been updated to provide specific information about the storage and preparation of these vaccines. Information about any other COVID-19 vaccines which are given regulatory approval will be added when this occurs. The information in this document was correct at time of publication. As COVID-19 is an evolving disease, much is still being learned about both the disease and the vaccines which have been developed to prevent it. For this reason, some information may change. Updates will be made to this document as new information becomes available. Please use the online version to ensure you are accessing the latest version. 2 COVID-19 vaccination programme: Information for healthcare practitioners Document revision information Version Details Date number 1.0 Document created 27 November 2020 2.0 Vaccine specific information about the COVID-19 mRNA 4 Vaccine BNT162b2 (Pfizer BioNTech) added December 2020 2.1 1. -
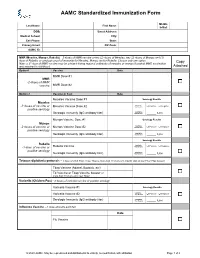
AAMC Standardized Immunization Form
AAMC Standardized Immunization Form Middle Last Name: First Name: Initial: DOB: Street Address: Medical School: City: Cell Phone: State: Primary Email: ZIP Code: AAMC ID: MMR (Measles, Mumps, Rubella) – 2 doses of MMR vaccine or two (2) doses of Measles, two (2) doses of Mumps and (1) dose of Rubella; or serologic proof of immunity for Measles, Mumps and/or Rubella. Choose only one option. Copy Note: a 3rd dose of MMR vaccine may be advised during regional outbreaks of measles or mumps if original MMR vaccination was received in childhood. Attached Option1 Vaccine Date MMR Dose #1 MMR -2 doses of MMR vaccine MMR Dose #2 Option 2 Vaccine or Test Date Measles Vaccine Dose #1 Serology Results Measles Qualitative -2 doses of vaccine or Measles Vaccine Dose #2 Titer Results: Positive Negative positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Mumps Vaccine Dose #1 Serology Results Mumps Qualitative -2 doses of vaccine or Mumps Vaccine Dose #2 Titer Results: Positive Negative positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Serology Results Rubella Qualitative Positive Negative -1 dose of vaccine or Rubella Vaccine Titer Results: positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Tetanus-diphtheria-pertussis – 1 dose of adult Tdap; if last Tdap is more than 10 years old, provide date of last Td or Tdap booster Tdap Vaccine (Adacel, Boostrix, etc) Td Vaccine or Tdap Vaccine booster (if more than 10 years since last Tdap) Varicella (Chicken Pox) - 2 doses of varicella vaccine or positive serology Varicella Vaccine #1 Serology Results Qualitative Varicella Vaccine #2 Titer Results: Positive Negative Serologic Immunity (IgG antibody titer) Quantitative Titer Results: _____ IU/ml Influenza Vaccine --1 dose annually each fall Date Flu Vaccine © 2020 AAMC. -

Hepatitis B Vaccine – Frequently Asked Questions (Information from the CDC)
AAMC Standardized Immunization Form 2020 Hepatitis B Vaccine – Frequently Asked Questions (Information from the CDC) 1. What are the hepatitis B vaccines licensed for use in the United States? Three single-antigen vaccines and two combination vaccines are currently licensed in the United States. Single-antigen hepatitis B vaccines: • ENGERIX-B® • RECOMBIVAX HB® • HEPLISAV-B™ Combination vaccines: • PEDIARIX®: Combined hepatitis B, diphtheria, tetanus, acellular pertussis (DTaP), and inactivated poliovirus (IPV) vaccine. Cannot be administered before age 6 weeks or after age 7 years. • TWINRIX®: Combined Hepatitis A and hepatitis B vaccine. Recommended for people aged ≥18 years who are at increased risk for both HAV and HBV infections. 2. What are the recommended schedules for hepatitis B vaccination? The vaccination schedule most often used for children and adults is three doses given at 0, 1, and 6 months. Alternate schedules have been approved for certain vaccines and/or populations. A new formulation, Heplisav-B (HepB-CpG), is approved to be given as two doses one month apart. 3. If there is an interruption between doses of hepatitis B vaccine, does the vaccine series need to be restarted? No. The series does not need to be restarted but the following should be considered: • If the vaccine series was interrupted after the first dose, the second dose should be administered as soon as possible. • The second and third doses should be separated by an interval of at least 8 weeks. • If only the third dose is delayed, it should be administered as soon as possible. 4. Is it harmful to administer an extra dose of hepatitis B vaccine or to repeat the entire vaccine series if documentation of the vaccination history is unavailable or the serology test is negative? No, administering extra doses of single-antigen hepatitis B vaccine is not harmful. -

1 Title: Interim Report of a Phase 2 Randomized Trial of a Plant
medRxiv preprint doi: https://doi.org/10.1101/2021.05.14.21257248; this version posted May 17, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. All rights reserved. No reuse allowed without permission. 1 Title: Interim Report of a Phase 2 Randomized Trial of a Plant-Produced Virus-Like Particle 2 Vaccine for Covid-19 in Healthy Adults Aged 18-64 and Older Adults Aged 65 and Older 3 Authors: Philipe Gobeil1, Stéphane Pillet1, Annie Séguin1, Iohann Boulay1, Asif Mahmood1, 4 Donald C Vinh 2, Nathalie Charland1, Philippe Boutet3, François Roman3, Robbert Van Der 5 Most4, Maria de los Angeles Ceregido Perez3, Brian J Ward1,2†, Nathalie Landry1† 6 Affiliations: 1 Medicago Inc., 1020 route de l’Église office 600, Québec, QC, Canada, G1V 7 3V9; 2 Research Institute of the McGill University Health Centre, 1001 Decarie St, Montreal, 8 QC H4A 3J1; 3 GlaxoSmithKline Biologicals SA (Vaccines), Avenue Fleming 20, 1300 Wavre, 9 Belgium; 4 GlaxoSmithKline Biologicals SA (Vaccines), rue de l’Institut 89, 1330 Rixensart, 10 Belgium; † These individuals are equally credited as senior authors. 11 * Corresponding author: Nathalie Landry, 1020 Route de l’Église, Bureau 600, Québec, Qc, 12 Canada, G1V 3V9; Tel. 418 658 9393; Fax. 418 658 6699; [email protected] 13 Abstract 14 The rapid spread of SARS-CoV-2 globally continues to impact humanity on a global scale with 15 rising morbidity and mortality. Despite the development of multiple effective vaccines, new 16 vaccines continue to be required to supply ongoing demand. -

COVID-19 Vaccines Frequently Asked Questions
Page 1 of 12 COVID-19 Vaccines 2020a Frequently Asked Questions Michigan.gov/Coronavirus The information in this document will change frequently as we learn more about COVID-19 vaccines. There is a lot we are learning as the pandemic and COVID-19 vaccines evolve. The approach in Michigan will adapt as we learn more. September 29, 2021. Quick Links What’s new | Why COVID-19 vaccination is important | Booster and additional doses | What to expect when you get vaccinated | Safety of the vaccine | Vaccine distribution/prioritization | Additional vaccine information | Protecting your privacy | Where can I get more information? What’s new − Pfizer booster doses recommended for some people to boost waning immunity six months after completing the Pfizer vaccine. Why COVID-19 vaccination is important − If you are fully vaccinated, you don’t have to quarantine after being exposed to COVID-19, as long as you don’t have symptoms. This means missing less work, school, sports and other activities. − COVID-19 vaccination is the safest way to build protection. COVID-19 is still a threat, especially to people who are unvaccinated. Some people who get COVID-19 can become severely ill, which could result in hospitalization, and some people have ongoing health problems several weeks or even longer after getting infected. Even people who did not have symptoms when they were infected can have these ongoing health problems. − After you are fully vaccinated for COVID-19, you can resume many activities that you did before the pandemic. CDC recommends that fully vaccinated people wear a mask in public indoor settings if they are in an area of substantial or high transmission. -

The Mississippi Covid-19 Vaccine Confidence Survey: Population Results Report
MAY 2021 THE MISSISSIPPI COVID-19 VACCINE CONFIDENCE SURVEY: POPULATION RESULTS Report A collaborative population-based study The Mississippi Community Engagement Alliance Against COVID-19 Disparities (CEAL) Team The Mississippi State Department of Health: Office of Preventive Health and Health Equity EXECUTIVE SUMMARY Since the Spring of 2020, the Novel 2019 Office of Preventive Health and Health Coronavirus (COVID-19) Pandemic has Equity (OPHHE) disseminated a statewide impacted Mississippians of every race, vaccine confidence survey beginning end ethnicity, age, gender, and income bracket. of December 2020 and collecting data Unfortunately, it has disproportionately until March 2021. The survey is intended to impacted Mississippians of color, the elderly, be representative of Mississippians, with and those living with chronic disease. For intentional efforts invested to reach lower most of the past year, the State has worked income and rural Mississippi populations, as to protect its population through preventive well as the state’s Black, Hispanic (Latino/ measures such as social distancing and Latinx), Asian (including the Vietnamese personal protective equipment. However, population of the Gulf Coast), and Native with the release of COVID-19 Vaccines to American/Choctaw communities. The sur- the public, the population of Mississippi has vey was administered in three languages- the opportunity to embrace a long-term English, Spanish, and Vietnamese- through a solution to COVID-19. That is, if Mississippians mixed-modal survey effort, including: web- are willing to receive the vaccine. To based, paper-based, and verbal-oratory assess Mississippians’ COVID-19 Vaccine administration. All targeted populations confidence, the Mississippi Community were ultimately reached and are represented Engagement Alliance Against COVID-19 in the over 11,000 completed responses Disparities (CEAL) Team and the Mississippi from all 82 of Mississippi’s counties. -

Immunity and How Vaccines Work
Immunity and how vaccines work Dr Brenda Corcoran National Immunisation Office Presentation Outline An understanding of the following principles: • Overview of immunity • Different types of vaccines and vaccine contents • Vaccine failures • Time intervals between vaccine doses • Vaccine overload • Adverse reactions • Herd immunity Immunity Immunity • The ability of the human body to protect itself from infectious disease The immune system • Cells with a protective function in the – bone marrow – thymus – lymphatic system of ducts and nodes – spleen –blood Types of immunity Source: http://en.wikipedia.org/wiki/Immunological_memory Natural (innate) immunity Non-specific mechanisms – Physical barriers • skin and mucous membranes – Chemical barriers • gastric and digestive enzymes – Cellular and protein secretions • phagocytes, macrophages, complement system ** No “memory” of protection exists afterwards ** Passive immunity – adaptive mechanisms Natural • maternal transfer of antibodies to infant via placenta Artificial • administration of pre- formed substance to provide immediate but short-term protection (antitoxin, antibodies) Protection is temporary and wanes with time (usually few months) Active immunity – adaptive mechanisms Natural • following contact with organism Artificial • administration of agent to stimulate immune response (immunisation) Acquired through contact with an micro-organism Protection produced by individual’s own immune system Protection often life-long but may need boosting How vaccines work • Induce active immunity – Immunity and immunologic memory similar to natural infection but without risk of disease • Immunological memory allows – Rapid recognition and response to pathogen – Prevent or modify effect of disease Live attenuated vaccines Weakened viruses /bacteria – Achieved by growing numerous generations in laboratory – Produces long lasting immune response after one or two doses – Stimulates immune system to react as it does to natural infection – Can cause mild form of the disease (e.g. -

Periodic Update on AEFI
CONSOLIDATED REGIONAL AND GLOBAL INFORMATION ON ADVERSE EVENTS FOLLOWING IMMUNIZATION (AEFI) AGAINST COVID-19 AND OTHER UPDATES Thirteenth report WASHINGTON, DC Updated: 10 May 2021 1 OFFICIAL REPORTS ON PHARMACOVIGILANCE PROGRAMS CANADA • As of 30 April 2021, 13,420,198 doses of the COVID-19 vaccines of Pfizer-BioNTech, Moderna, Oxford- AstraZeneca, and the vaccine Covishield (AstraZeneca manufactured by the Serum Institute of India), had been administered. • A total of 4,548 individual reports of one or more adverse events (0.034% of doses administered) were received. Of these, 748 were considered serious events (0.006% of doses administered), with anaphylaxis being the one most frequently reported. • Of total reports, there were 2,072 non-serious and 520 serious events associated with the Pfizer-BioNTech vaccine. For the Moderna vaccine, 1,457 non-serious and 103 serious events were reported; for Covishield, there were 201 non-serious and 48 serious events, and for Oxford-AstraZeneca, 65 non-serious events and 62 serious events. • A total of 13,596 adverse events following immunization (AEFI) were reported, with 4,548 reports with one or more events. The most frequently reported non-serious adverse events were injection-site reactions, paresthesia, itching, hives, headache, hypoesthesia, and nausea. There were 61 reports of anaphylaxis. • Although 55% of vaccine doses were administered to women as of 30 April, and 45% to men, most of the reported adverse events were in women (84.3% of total doses). At the same time, 43.0% of total events were reported for people between the ages of 18 and 49, who account for 24% of people vaccinated. -

COVID-19: China Medical Supply Chains and Broader Trade Issues
COVID-19: China Medical Supply Chains and Broader Trade Issues Updated December 23, 2020 Congressional Research Service https://crsreports.congress.gov R46304 SUMMARY R46304 COVID-19: China Medical Supply Chains and December 23, 2020 Broader Trade Issues Karen M. Sutter, The outbreak of Coronavirus Disease 2019 (COVID-19), first in China, and then Coordinator globally, including in the United States, has drawn attention to the ways in which the Specialist in Asian Trade U.S. economy depends on manufacturing and supply chains based in China. This report and Finance aims to assess current developments and identify immediate and longer range China trade issues for Congress. Andres B. Schwarzenberg Analyst in International An area of particular concern to Congress has been U.S. shortages in medical supplies— Trade and Finance including personal protective equipment (PPE) and pharmaceuticals—as the United States stepped up efforts to contain the COVID-19 pandemic with limited domestic Michael D. Sutherland stockpiles and insufficient U.S. industrial capacity. Because of China’s role as a global Analyst in International supplier of PPE, medical devices, antibiotics, and active pharmaceutical ingredients, Trade and Finance reduced exports from China led to shortages of critical medical supplies in the United States. Exacerbating the situation, in early February 2020, the Chinese government nationalized control of the production and distribution of medical supplies in China— directing all production for domestic use—and directed the bureaucracy and Chinese industry to secure supplies from the global market. Once past the initial peak of its COVID-19 outbreak, the Chinese government appears to have prioritized certain countries and selectively released some medical supplies for overseas delivery.