5. Key Post-Emergent Modes of Action
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Evolution of Resistance to Auxinic Herbicides: Historical Perspectives, Mechanisms of Resistance, and Implications for Broadleaf Weed Management in Agronomic Crops J
Weed Science 2011 59:445–457 Evolution of Resistance to Auxinic Herbicides: Historical Perspectives, Mechanisms of Resistance, and Implications for Broadleaf Weed Management in Agronomic Crops J. Mithila, J. Christopher Hall, William G. Johnson, Kevin B. Kelley, and Dean E. Riechers* Auxinic herbicides are widely used for control of broadleaf weeds in cereal crops and turfgrass. These herbicides are structurally similar to the natural plant hormone auxin, and induce several of the same physiological and biochemical responses at low concentrations. After several decades of research to understand the auxin signal transduction pathway, the receptors for auxin binding and resultant biochemical and physiological responses have recently been discovered in plants. However, the precise mode of action for the auxinic herbicides is not completely understood despite their extensive use in agriculture for over six decades. Auxinic herbicide-resistant weed biotypes offer excellent model species for uncovering the mode of action as well as resistance to these compounds. Compared with other herbicide families, the incidence of resistance to auxinic herbicides is relatively low, with only 29 auxinic herbicide-resistant weed species discovered to date. The relatively low incidence of resistance to auxinic herbicides has been attributed to the presence of rare alleles imparting resistance in natural weed populations, the potential for fitness penalties due to mutations conferring resistance in weeds, and the complex mode of action of auxinic herbicides in sensitive dicot plants. This review discusses recent advances in the auxin signal transduction pathway and its relation to auxinic herbicide mode of action. Furthermore, comprehensive information about the genetics and inheritance of auxinic herbicide resistance and case studies examining mechanisms of resistance in auxinic herbicide-resistant broadleaf weed biotypes are provided. -

2,4-Dichlorophenoxyacetic Acid
2,4-Dichlorophenoxyacetic acid 2,4-Dichlorophenoxyacetic acid IUPAC (2,4-dichlorophenoxy)acetic acid name 2,4-D Other hedonal names trinoxol Identifiers CAS [94-75-7] number SMILES OC(COC1=CC=C(Cl)C=C1Cl)=O ChemSpider 1441 ID Properties Molecular C H Cl O formula 8 6 2 3 Molar mass 221.04 g mol−1 Appearance white to yellow powder Melting point 140.5 °C (413.5 K) Boiling 160 °C (0.4 mm Hg) point Solubility in 900 mg/L (25 °C) water Related compounds Related 2,4,5-T, Dichlorprop compounds Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) 2,4-Dichlorophenoxyacetic acid (2,4-D) is a common systemic herbicide used in the control of broadleaf weeds. It is the most widely used herbicide in the world, and the third most commonly used in North America.[1] 2,4-D is also an important synthetic auxin, often used in laboratories for plant research and as a supplement in plant cell culture media such as MS medium. History 2,4-D was developed during World War II by a British team at Rothamsted Experimental Station, under the leadership of Judah Hirsch Quastel, aiming to increase crop yields for a nation at war.[citation needed] When it was commercially released in 1946, it became the first successful selective herbicide and allowed for greatly enhanced weed control in wheat, maize (corn), rice, and similar cereal grass crop, because it only kills dicots, leaving behind monocots. Mechanism of herbicide action 2,4-D is a synthetic auxin, which is a class of plant growth regulators. -

Common and Chemical Names of Herbicides Approved by the WSSA
Weed Science 2010 58:511–518 Common and Chemical Names of Herbicides Approved by the Weed Science Society of America Below is the complete list of all common and chemical of herbicides as approved by the International Organization names of herbicides approved by the Weed Science Society of for Standardization (ISO). A sponsor may submit a proposal America (WSSA) and updated as of September 1, 2010. for a common name directly to the WSSA Terminology Beginning in 1996, it has been published yearly in the last Committee. issue of Weed Science with Directions for Contributors to A herbicide common name is not synonymous with Weed Science. This list is published in lieu of the selections a commercial formulation of the same herbicide, and in printed previously on the back cover of Weed Science. Only many instances, is not synonymous with the active ingredient common and chemical names included in this complete of a commercial formulation as identified on the product list should be used in WSSA publications. In the absence of label. If the herbicide is a salt or simple ester of a parent a WSSA-approved common name, the industry code number compound, the WSSA common name applies to the parent as compiled by the Chemical Abstracts Service (CAS) with compound only. CAS systematic chemical name or the systematic chemical The chemical name used in this list is that preferred by the name alone may be used. The current approved list is also Chemical Abstracts Service (CAS) according to their system of available at our web site (www.wssa.net). -

Herbicide Mode of Action Table High Resistance Risk
Herbicide Mode of Action Table High resistance risk Chemical family Active constituent (first registered trade name) GROUP 1 Inhibition of acetyl co-enzyme A carboxylase (ACC’ase inhibitors) clodinafop (Topik®), cyhalofop (Agixa®*, Barnstorm®), diclofop (Cheetah® Gold* Decision®*, Hoegrass®), Aryloxyphenoxy- fenoxaprop (Cheetah®, Gold*, Wildcat®), fluazifop propionates (FOPs) (Fusilade®), haloxyfop (Verdict®), propaquizafop (Shogun®), quizalofop (Targa®) Cyclohexanediones (DIMs) butroxydim (Factor®*), clethodim (Select®), profoxydim (Aura®), sethoxydim (Cheetah® Gold*, Decision®*), tralkoxydim (Achieve®) Phenylpyrazoles (DENs) pinoxaden (Axial®) GROUP 2 Inhibition of acetolactate synthase (ALS inhibitors), acetohydroxyacid synthase (AHAS) Imidazolinones (IMIs) imazamox (Intervix®*, Raptor®), imazapic (Bobcat I-Maxx®*, Flame®, Midas®*, OnDuty®*), imazapyr (Arsenal Xpress®*, Intervix®*, Lightning®*, Midas®* OnDuty®*), imazethapyr (Lightning®*, Spinnaker®) Pyrimidinyl–thio- bispyribac (Nominee®), pyrithiobac (Staple®) benzoates Sulfonylureas (SUs) azimsulfuron (Gulliver®), bensulfuron (Londax®), chlorsulfuron (Glean®), ethoxysulfuron (Hero®), foramsulfuron (Tribute®), halosulfuron (Sempra®), iodosulfuron (Hussar®), mesosulfuron (Atlantis®), metsulfuron (Ally®, Harmony®* M, Stinger®*, Trounce®*, Ultimate Brushweed®* Herbicide), prosulfuron (Casper®*), rimsulfuron (Titus®), sulfometuron (Oust®, Eucmix Pre Plant®*, Trimac Plus®*), sulfosulfuron (Monza®), thifensulfuron (Harmony®* M), triasulfuron (Logran®, Logran® B-Power®*), tribenuron (Express®), -

Exposure to Herbicides in House Dust and Risk of Childhood Acute Lymphoblastic Leukemia
Journal of Exposure Science and Environmental Epidemiology (2013) 23, 363–370 & 2013 Nature America, Inc. All rights reserved 1559-0631/13 www.nature.com/jes ORIGINAL ARTICLE Exposure to herbicides in house dust and risk of childhood acute lymphoblastic leukemia Catherine Metayer1, Joanne S. Colt2, Patricia A. Buffler1, Helen D. Reed3, Steve Selvin1, Vonda Crouse4 and Mary H. Ward2 We examine the association between exposure to herbicides and childhood acute lymphoblastic leukemia (ALL). Dust samples were collected from homes of 269 ALL cases and 333 healthy controls (o8 years of age at diagnosis/reference date and residing in same home since diagnosis/reference date) in California, using a high-volume surface sampler or household vacuum bags. Amounts of agricultural or professional herbicides (alachlor, metolachlor, bromoxynil, bromoxynil octanoate, pebulate, butylate, prometryn, simazine, ethalfluralin, and pendimethalin) and residential herbicides (cyanazine, trifluralin, 2-methyl-4- chlorophenoxyacetic acid (MCPA), mecoprop, 2,4-dichlorophenoxyacetic acid (2,4-D), chlorthal, and dicamba) were measured. Odds ratios (OR) and 95% confidence intervals (CI) were estimated by logistic regression. Models included the herbicide of interest, age, sex, race/ethnicity, household income, year and season of dust sampling, neighborhood type, and residence type. The risk of childhood ALL was associated with dust levels of chlorthal; compared to homes with no detections, ORs for the first, second, and third tertiles were 1.49 (95% CI: 0.82–2.72), 1.49 (95% CI: 0.83–2.67), and 1.57 (95% CI: 0.90–2.73), respectively (P-value for linear trend ¼ 0.05). The magnitude of this association appeared to be higher in the presence of alachlor. -

R Graphics Output
Dexamethasone sodium phosphate ( 0.339 ) Melengestrol acetate ( 0.282 ) 17beta−Trenbolone ( 0.252 ) 17alpha−Estradiol ( 0.24 ) 17alpha−Hydroxyprogesterone ( 0.238 ) Triamcinolone ( 0.233 ) Zearalenone ( 0.216 ) CP−634384 ( 0.21 ) 17alpha−Ethinylestradiol ( 0.203 ) Raloxifene hydrochloride ( 0.203 ) Volinanserin ( 0.2 ) Tiratricol ( 0.197 ) trans−Retinoic acid ( 0.192 ) Chlorpromazine hydrochloride ( 0.191 ) PharmaGSID_47315 ( 0.185 ) Apigenin ( 0.183 ) Diethylstilbestrol ( 0.178 ) 4−Dodecylphenol ( 0.161 ) 2,2',6,6'−Tetrachlorobisphenol A ( 0.156 ) o,p'−DDD ( 0.155 ) Progesterone ( 0.152 ) 4−Hydroxytamoxifen ( 0.151 ) SSR150106 ( 0.149 ) Equilin ( 0.3 ) 3,5,3'−Triiodothyronine ( 0.256 ) 17−Methyltestosterone ( 0.242 ) 17beta−Estradiol ( 0.24 ) 5alpha−Dihydrotestosterone ( 0.235 ) Mifepristone ( 0.218 ) Norethindrone ( 0.214 ) Spironolactone ( 0.204 ) Farglitazar ( 0.203 ) Testosterone propionate ( 0.202 ) meso−Hexestrol ( 0.199 ) Mestranol ( 0.196 ) Estriol ( 0.191 ) 2,2',4,4'−Tetrahydroxybenzophenone ( 0.185 ) 3,3,5,5−Tetraiodothyroacetic acid ( 0.183 ) Norgestrel ( 0.181 ) Cyproterone acetate ( 0.164 ) GSK232420A ( 0.161 ) N−Dodecanoyl−N−methylglycine ( 0.155 ) Pentachloroanisole ( 0.154 ) HPTE ( 0.151 ) Biochanin A ( 0.15 ) Dehydroepiandrosterone ( 0.149 ) PharmaCode_333941 ( 0.148 ) Prednisone ( 0.146 ) Nordihydroguaiaretic acid ( 0.145 ) p,p'−DDD ( 0.144 ) Diphenhydramine hydrochloride ( 0.142 ) Forskolin ( 0.141 ) Perfluorooctanoic acid ( 0.14 ) Oleyl sarcosine ( 0.139 ) Cyclohexylphenylketone ( 0.138 ) Pirinixic acid ( 0.137 ) -

INDEX to PESTICIDE TYPES and FAMILIES and PART 180 TOLERANCE INFORMATION of PESTICIDE CHEMICALS in FOOD and FEED COMMODITIES
US Environmental Protection Agency Office of Pesticide Programs INDEX to PESTICIDE TYPES and FAMILIES and PART 180 TOLERANCE INFORMATION of PESTICIDE CHEMICALS in FOOD and FEED COMMODITIES Note: Pesticide tolerance information is updated in the Code of Federal Regulations on a weekly basis. EPA plans to update these indexes biannually. These indexes are current as of the date indicated in the pdf file. For the latest information on pesticide tolerances, please check the electronic Code of Federal Regulations (eCFR) at http://www.access.gpo.gov/nara/cfr/waisidx_07/40cfrv23_07.html 1 40 CFR Type Family Common name CAS Number PC code 180.163 Acaricide bridged diphenyl Dicofol (1,1-Bis(chlorophenyl)-2,2,2-trichloroethanol) 115-32-2 10501 180.198 Acaricide phosphonate Trichlorfon 52-68-6 57901 180.259 Acaricide sulfite ester Propargite 2312-35-8 97601 180.446 Acaricide tetrazine Clofentezine 74115-24-5 125501 180.448 Acaricide thiazolidine Hexythiazox 78587-05-0 128849 180.517 Acaricide phenylpyrazole Fipronil 120068-37-3 129121 180.566 Acaricide pyrazole Fenpyroximate 134098-61-6 129131 180.572 Acaricide carbazate Bifenazate 149877-41-8 586 180.593 Acaricide unclassified Etoxazole 153233-91-1 107091 180.599 Acaricide unclassified Acequinocyl 57960-19-7 6329 180.341 Acaricide, fungicide dinitrophenol Dinocap (2, 4-Dinitro-6-octylphenyl crotonate and 2,6-dinitro-4- 39300-45-3 36001 octylphenyl crotonate} 180.111 Acaricide, insecticide organophosphorus Malathion 121-75-5 57701 180.182 Acaricide, insecticide cyclodiene Endosulfan 115-29-7 79401 -
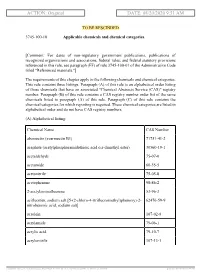
ACTION: Original DATE: 08/20/2020 9:51 AM
ACTION: Original DATE: 08/20/2020 9:51 AM TO BE RESCINDED 3745-100-10 Applicable chemicals and chemical categories. [Comment: For dates of non-regulatory government publications, publications of recognized organizations and associations, federal rules, and federal statutory provisions referenced in this rule, see paragraph (FF) of rule 3745-100-01 of the Administrative Code titled "Referenced materials."] The requirements of this chapter apply to the following chemicals and chemical categories. This rule contains three listings. Paragraph (A) of this rule is an alphabetical order listing of those chemicals that have an associated "Chemical Abstracts Service (CAS)" registry number. Paragraph (B) of this rule contains a CAS registry number order list of the same chemicals listed in paragraph (A) of this rule. Paragraph (C) of this rule contains the chemical categories for which reporting is required. These chemical categories are listed in alphabetical order and do not have CAS registry numbers. (A) Alphabetical listing: Chemical Name CAS Number abamectin (avermectin B1) 71751-41-2 acephate (acetylphosphoramidothioic acid o,s-dimethyl ester) 30560-19-1 acetaldehyde 75-07-0 acetamide 60-35-5 acetonitrile 75-05-8 acetophenone 98-86-2 2-acetylaminofluorene 53-96-3 acifluorfen, sodium salt [5-(2-chloro-4-(trifluoromethyl)phenoxy)-2- 62476-59-9 nitrobenzoic acid, sodium salt] acrolein 107-02-8 acrylamide 79-06-1 acrylic acid 79-10-7 acrylonitrile 107-13-1 [ stylesheet: rule.xsl 2.14, authoring tool: RAS XMetaL R2_0F1, (dv: 0, p: 185720, pa: -

Please Use Caution When Applying Herbicides Near Wine Grapes
Please Use Caution When Applying Herbicides Near Wine Grapes Phenoxy herbicides are very damaging to grapevines Grapevines are extremely sensitive to the application of certain herbicides commonly used by farmers and homeowners, especially phenoxy herbicides. Phenoxy herbicides include 2,4-D, MCPA, Crossbow, Banvel, Garlon, Weed-B-Gone, and Brush Killer, among others. The active ingredient of phenoxy-type herbicides may be listed on the label in “weed and feed” and brush control products for use in home landscaping as 2,4-dichlorophenoxy- acetic acid (2,4-D), 2-methyl-4-chlorophenoxyacetic acid, triclopyr, or dicamba. Sensitivity to phenoxy herbicides exists throughout the grapevine's growing season (mid-March through October). Grapevines are most vulnerable from the early growing season through the bloom and fruit set period (mid-March through June). Phenoxy herbicides do not require a pesticide license for purchase in Oregon and are readily available from home improvement stores, garden centers, retail nurseries, etc. This family of herbicides is very effective and economical for controlling broadleaf weeds. These herbicides are commonly used on a variety of sites such as lawns, golf courses, rights-of-way and agricultural fields and by homeowners. Two forms of spray drift can damage grapevines Drift of spray droplets: Small particles can move with the wind, land on grapes, and be absorbed into the grapevines through the cuticle on the leaf. The smaller the droplet, the further it will travel. Vapor drift: Volatile herbicides may produce vapors that are carried several miles from the target area. Herbicide particles or vapors may be moved from the application site by wind, shifting air currents, climatic inversions or using high pressures when spraying. -

Butafenacil – a New Complimentary Premix Partner for Triasulfuron Or Glyphosate for the Enhanced Knockdown and Residual Contro
Thirteenth Australian Weeds Conference Butafenacil – a new complimentary premix partner for triasulfuron or glyphosate for the enhanced knockdown and residual control of weeds in broadacre cropping situations Brett A. Davies Syngenta Crop Protection Pty. Ltd., 140–150 Bungaree Road, PO Box 249, Pendle Hill, New South Wales 2145, Australia Summary Butafenacil is a new pyrimidindione her- symptoms, however, are already observed within a few bicide released in Australia during the 2002 season hours after treatment. as a complementary component of two new products Trial work with butafenacil has been ongoing in – Logran® B-Power™ (a premix of triasulfuron and Australia since the mid 1990s. Over 100 trials have butafenacil), for the pre-emergent control of grass been conducted to investigate the complimentary activ- and broadleaf weeds in wheat, and Touchdown® B- ity of butafenacil in mixtures with either glyphosate or Power™ (a premix of glyphosate and butafenacil), for triasulfuron for pre-plant weed knockdown and in-crop the knockdown control of grass and broadleaf weeds residual weed. prior to the sowing of cereals crops. This paper provides a summary of the spectrum Effi cacy trials conducted throughout the major of activity on weeds of both Touchdown B-Power (a grain growing regions of Australia since the late premix of 5 g a.i. kg-1 of butafenacil, and 225 g a.i. 1990s have highlighted the enhanced knockdown kg-1 of glyphosate present as the isopropylamine salt control of Touchdown B-Power at rates ranging from as a suspension concentrate formulation) and Logran 184–636 g a.i. ha-1 of a wide range of both grass and B-Power (a premix of 200 g a.i. -

Appendix a Analysis of Products with Two Or More Active Ingredients
APPENDIX A ANALYSIS OF PRODUCTS WITH TWO OR MORE ACTIVE INGREDIENTS The Agency does not routinely include, in its risk assessments, an evaluation of mixtures of active ingredients, either those mixtures of multiple active ingredients in product formulations or those in the applicator’s tank. In the case of the product formulations of active ingredients (that is, a registered product containing more than one active ingredient), each active ingredient is subject to an individual risk assessment for regulatory decision regarding the active ingredient on a particular use site. If effects data are available for a formulated product containing more than one active ingredient, they may be used qualitatively or quantitatively1 2. There are no product LD50 values, with associated 95% Confidence Intervals (CIs) available for glyphosate. As discussed in USEPA (2000) a quantitative component-based evaluation of mixture toxicity requires data of appropriate quality for each component of a mixture. In this mixture evaluation an LD50 with associated 95% CI is needed for the formulated product. The same quality of data is also required for each component of the mixture. Given that the formulated products for glyphosate do not have LD50 data available it is not possible to undertake a quantitative or qualitative analysis for potential interactive effects. However, because the active ingredients are not expected to have similar mechanisms of action, metabolites, or toxicokinetic behavior, it is reasonable to conclude that an assumption of dose-addition would be inappropriate. Consequently, an assessment based on the toxicity of glyphosate is the only reasonable approach that employs the available data to address the potential acute risks of the formulated products. -

AP-42, CH 9.2.2: Pesticide Application
9.2.2PesticideApplication 9.2.2.1General1-2 Pesticidesaresubstancesormixturesusedtocontrolplantandanimallifeforthepurposesof increasingandimprovingagriculturalproduction,protectingpublichealthfrompest-bornediseaseand discomfort,reducingpropertydamagecausedbypests,andimprovingtheaestheticqualityofoutdoor orindoorsurroundings.Pesticidesareusedwidelyinagriculture,byhomeowners,byindustry,andby governmentagencies.Thelargestusageofchemicalswithpesticidalactivity,byweightof"active ingredient"(AI),isinagriculture.Agriculturalpesticidesareusedforcost-effectivecontrolofweeds, insects,mites,fungi,nematodes,andotherthreatstotheyield,quality,orsafetyoffood.Theannual U.S.usageofpesticideAIs(i.e.,insecticides,herbicides,andfungicides)isover800millionpounds. AiremissionsfrompesticideusearisebecauseofthevolatilenatureofmanyAIs,solvents, andotheradditivesusedinformulations,andofthedustynatureofsomeformulations.Mostmodern pesticidesareorganiccompounds.EmissionscanresultdirectlyduringapplicationorastheAIor solventvolatilizesovertimefromsoilandvegetation.Thisdiscussionwillfocusonemissionfactors forvolatilization.Thereareinsufficientdataavailableonparticulateemissionstopermitemission factordevelopment. 9.2.2.2ProcessDescription3-6 ApplicationMethods- Pesticideapplicationmethodsvaryaccordingtothetargetpestandtothecroporothervalue tobeprotected.Insomecases,thepesticideisapplieddirectlytothepest,andinotherstothehost plant.Instillothers,itisusedonthesoilorinanenclosedairspace.Pesticidemanufacturershave developedvariousformulationsofAIstomeetboththepestcontrolneedsandthepreferred