Association Between HOTAIR Lncrna Polymorphisms and Coronary Artery Disease Susceptibility
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

SUPPLEMENTARY MATERIAL Bone Morphogenetic Protein 4 Promotes
www.intjdevbiol.com doi: 10.1387/ijdb.160040mk SUPPLEMENTARY MATERIAL corresponding to: Bone morphogenetic protein 4 promotes craniofacial neural crest induction from human pluripotent stem cells SUMIYO MIMURA, MIKA SUGA, KAORI OKADA, MASAKI KINEHARA, HIROKI NIKAWA and MIHO K. FURUE* *Address correspondence to: Miho Kusuda Furue. Laboratory of Stem Cell Cultures, National Institutes of Biomedical Innovation, Health and Nutrition, 7-6-8, Saito-Asagi, Ibaraki, Osaka 567-0085, Japan. Tel: 81-72-641-9819. Fax: 81-72-641-9812. E-mail: [email protected] Full text for this paper is available at: http://dx.doi.org/10.1387/ijdb.160040mk TABLE S1 PRIMER LIST FOR QRT-PCR Gene forward reverse AP2α AATTTCTCAACCGACAACATT ATCTGTTTTGTAGCCAGGAGC CDX2 CTGGAGCTGGAGAAGGAGTTTC ATTTTAACCTGCCTCTCAGAGAGC DLX1 AGTTTGCAGTTGCAGGCTTT CCCTGCTTCATCAGCTTCTT FOXD3 CAGCGGTTCGGCGGGAGG TGAGTGAGAGGTTGTGGCGGATG GAPDH CAAAGTTGTCATGGATGACC CCATGGAGAAGGCTGGGG MSX1 GGATCAGACTTCGGAGAGTGAACT GCCTTCCCTTTAACCCTCACA NANOG TGAACCTCAGCTACAAACAG TGGTGGTAGGAAGAGTAAAG OCT4 GACAGGGGGAGGGGAGGAGCTAGG CTTCCCTCCAACCAGTTGCCCCAAA PAX3 TTGCAATGGCCTCTCAC AGGGGAGAGCGCGTAATC PAX6 GTCCATCTTTGCTTGGGAAA TAGCCAGGTTGCGAAGAACT p75 TCATCCCTGTCTATTGCTCCA TGTTCTGCTTGCAGCTGTTC SOX9 AATGGAGCAGCGAAATCAAC CAGAGAGATTTAGCACACTGATC SOX10 GACCAGTACCCGCACCTG CGCTTGTCACTTTCGTTCAG Suppl. Fig. S1. Comparison of the gene expression profiles of the ES cells and the cells induced by NC and NC-B condition. Scatter plots compares the normalized expression of every gene on the array (refer to Table S3). The central line -

Appendix 2. Significantly Differentially Regulated Genes in Term Compared with Second Trimester Amniotic Fluid Supernatant
Appendix 2. Significantly Differentially Regulated Genes in Term Compared With Second Trimester Amniotic Fluid Supernatant Fold Change in term vs second trimester Amniotic Affymetrix Duplicate Fluid Probe ID probes Symbol Entrez Gene Name 1019.9 217059_at D MUC7 mucin 7, secreted 424.5 211735_x_at D SFTPC surfactant protein C 416.2 206835_at STATH statherin 363.4 214387_x_at D SFTPC surfactant protein C 295.5 205982_x_at D SFTPC surfactant protein C 288.7 1553454_at RPTN repetin solute carrier family 34 (sodium 251.3 204124_at SLC34A2 phosphate), member 2 238.9 206786_at HTN3 histatin 3 161.5 220191_at GKN1 gastrokine 1 152.7 223678_s_at D SFTPA2 surfactant protein A2 130.9 207430_s_at D MSMB microseminoprotein, beta- 99.0 214199_at SFTPD surfactant protein D major histocompatibility complex, class II, 96.5 210982_s_at D HLA-DRA DR alpha 96.5 221133_s_at D CLDN18 claudin 18 94.4 238222_at GKN2 gastrokine 2 93.7 1557961_s_at D LOC100127983 uncharacterized LOC100127983 93.1 229584_at LRRK2 leucine-rich repeat kinase 2 HOXD cluster antisense RNA 1 (non- 88.6 242042_s_at D HOXD-AS1 protein coding) 86.0 205569_at LAMP3 lysosomal-associated membrane protein 3 85.4 232698_at BPIFB2 BPI fold containing family B, member 2 84.4 205979_at SCGB2A1 secretoglobin, family 2A, member 1 84.3 230469_at RTKN2 rhotekin 2 82.2 204130_at HSD11B2 hydroxysteroid (11-beta) dehydrogenase 2 81.9 222242_s_at KLK5 kallikrein-related peptidase 5 77.0 237281_at AKAP14 A kinase (PRKA) anchor protein 14 76.7 1553602_at MUCL1 mucin-like 1 76.3 216359_at D MUC7 mucin 7, -

Transcription Factor Gene Expression Profiling and Analysis of SOX Gene Family Transcription Factors in Human Limbal Epithelial
Transcription factor gene expression profiling and analysis of SOX gene family transcription factors in human limbal epithelial progenitor cells Der Naturwissenschaftlichen Fakultät der Friedrich-Alexander-Universität Erlangen-Nürnberg zur Erlangung des Doktorgrades Dr. rer. nat. vorgelegt von Dr. med. Johannes Menzel-Severing aus Bonn Als Dissertation genehmigt von der Naturwissenschaftlichen Fakultät der Friedrich-Alexander-Universität Erlangen-Nürnberg Tag der mündlichen Prüfung: 7. Februar 2018 Vorsitzender des Promotionsorgans: Prof. Dr. Georg Kreimer Gutachter: Prof. Dr. Andreas Feigenspan Prof. Dr. Ursula Schlötzer-Schrehardt 1 INDEX 1. ABSTRACTS Page 1.1. Abstract in English 4 1.2. Zusammenfassung auf Deutsch 7 2. INTRODUCTION 2.1. Anatomy and histology of the cornea and the corneal surface 11 2.2. Homeostasis of corneal epithelium and the limbal stem cell paradigm 13 2.3. The limbal stem cell niche 15 2.4. Cell therapeutic strategies in ocular surface disease 17 2.5. Alternative cell sources for transplantation to the corneal surface 18 2.6. Transcription factors in cell differentiation and reprogramming 21 2.7. Transcription factors in limbal epithelial cells 22 2.8. Research question 25 3. MATERIALS AND METHODS 3.1. Human donor corneas 27 3.2. Laser Capture Microdissection (LCM) 28 3.3. RNA amplification and RT2 profiler PCR arrays 29 3.4. Real-time PCR analysis 33 3.5. Immunohistochemistry 34 3.6. Limbal epithelial cell culture 38 3.7. Transcription-factor knockdown/overexpression in vitro 39 3.8. Proliferation assay 40 3.9. Western blot 40 3.10. Statistical analysis 41 2 4. RESULTS 4.1. Quality control of LCM-isolated and amplified RNA 42 4.2. -
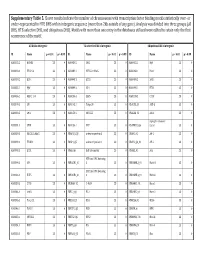
Supplementary Table 5. Clover Results Indicate the Number Of
Supplementary Table 5. Clover results indicate the number of chromosomes with transcription factor binding motifs statistically over‐ or under‐represented in HTE DHS within intergenic sequence (more than 2kb outside of any gene). Analysis was divided into three groups (all DHS, HTE‐selective DHS, and ubiquitous DHS). Motifs with more than one entry in the databases utilized were edited to retain only the first occurrence of the motif. All DHS x Intergenic TEselective DHS x Intergenic Ubiquitous DHS x Intergenic ID Name p < 0.01 p > 0.99 ID Name p < 0.01 p > 0.99 ID Name p < 0.01 p > 0.99 MA0002.2 RUNX1 23 0 MA0080.2 SPI1 23 0 MA0055.1 Myf 23 0 MA0003.1 TFAP2A 23 0 MA0089.1 NFE2L1::MafG 23 0 MA0068.1 Pax4 23 0 MA0039.2 Klf4 23 0 MA0098.1 ETS1 23 0 MA0080.2 SPI1 23 0 MA0055.1 Myf 23 0 MA0099.2 AP1 23 0 MA0098.1 ETS1 23 0 MA0056.1 MZF1_1‐4 23 0 MA0136.1 ELF5 23 0 MA0139.1 CTCF 23 0 MA0079.2 SP1 23 0 MA0145.1 Tcfcp2l1 23 0 V$ALX3_01 ALX‐3 23 0 MA0080.2 SPI1 23 0 MA0150.1 NFE2L2 23 0 V$ALX4_02 Alx‐4 23 0 myocyte enhancer MA0081.1 SPIB 23 0 MA0156.1 FEV 23 0 V$AMEF2_Q6 factor 23 0 MA0089.1 NFE2L1::MafG 23 0 V$AP1FJ_Q2 activator protein 1 23 0 V$AP1_01 AP‐1 23 0 MA0090.1 TEAD1 23 0 V$AP4_Q5 activator protein 4 23 0 V$AP2_Q6_01 AP‐2 23 0 MA0098.1 ETS1 23 0 V$AR_Q6 half‐site matrix 23 0 V$ARX_01 Arx 23 0 BTB and CNC homolog MA0099.2 AP1 23 0 V$BACH1_01 1 23 0 V$BARHL1_01 Barhl‐1 23 0 BTB and CNC homolog MA0136.1 ELF5 23 0 V$BACH2_01 2 23 0 V$BARHL2_01 Barhl2 23 0 MA0139.1 CTCF 23 0 V$CMAF_02 C‐MAF 23 0 V$BARX1_01 Barx1 23 0 MA0144.1 Stat3 23 0 -

Aberrant HOXC Expression Accompanies the Malignant Phenotype in Human Prostate1
[CANCER RESEARCH 63, 5879–5888, September 15, 2003] Aberrant HOXC Expression Accompanies the Malignant Phenotype in Human Prostate1 Gary J. Miller,2 Heidi L. Miller, Adrie van Bokhoven, James R. Lambert, Priya N. Werahera, Osvaldo Schirripa,3 M. Scott Lucia, and Steven K. Nordeen4 Department of Pathology, University of Colorado Health Sciences Center, Denver, Colorado 80262 ABSTRACT breast (13, 14), and renal (15) carcinomas; melanomas (16); and squamous carcinomas of the skin (17). Because the genes implicated Dysregulation of HOX gene expression has been implicated as a factor show little consensus, the dysregulation may be a tissue-specific in malignancies for a number of years. However, no consensus has perturbation of the existing HOX expression pattern rather than a emerged regarding specific causative genes. Using a degenerate reverse transcription-PCR technique, we show up-regulation of genes from the single causative gene. Tissue-specific expression patterns have been HOXC cluster in malignant prostate cell lines and lymph node metastases. reported in kidney and colon, by Northern blot analysis (12, 15). When relative expression levels of the four HOX clusters were examined, Primary tumors in both kidney and colon showed variations in spe- lymph node metastases and cell lines derived from lymph node metastases cific HOX gene expression from the corresponding normal tissue, but exhibited very similar patterns, patterns distinct from those in benign cells overall expression patterns for individual tumors were not reported. or malignant cell lines derived from other tumor sites. Specific reverse Only primary kidney tumors were examined (15), but liver metastases transcription-PCR for HOXC4, HOXC5, HOXC6, and HOXC8 confirmed from colon tumors reportedly displayed expression of specific HOX overexpression of these genes in malignant cell lines and lymph node genes similar to that seen in either primary colon tumors or normal metastases. -

1 Novel Expression Signatures Identified by Transcriptional Analysis
ARD Online First, published on October 7, 2009 as 10.1136/ard.2009.108043 Ann Rheum Dis: first published as 10.1136/ard.2009.108043 on 7 October 2009. Downloaded from Novel expression signatures identified by transcriptional analysis of separated leukocyte subsets in SLE and vasculitis 1Paul A Lyons, 1Eoin F McKinney, 1Tim F Rayner, 1Alexander Hatton, 1Hayley B Woffendin, 1Maria Koukoulaki, 2Thomas C Freeman, 1David RW Jayne, 1Afzal N Chaudhry, and 1Kenneth GC Smith. 1Cambridge Institute for Medical Research and Department of Medicine, Addenbrooke’s Hospital, Hills Road, Cambridge, CB2 0XY, UK 2Roslin Institute, University of Edinburgh, Roslin, Midlothian, EH25 9PS, UK Correspondence should be addressed to Dr Paul Lyons or Prof Kenneth Smith, Department of Medicine, Cambridge Institute for Medical Research, Addenbrooke’s Hospital, Hills Road, Cambridge, CB2 0XY, UK. Telephone: +44 1223 762642, Fax: +44 1223 762640, E-mail: [email protected] or [email protected] Key words: Gene expression, autoimmune disease, SLE, vasculitis Word count: 2,906 The Corresponding Author has the right to grant on behalf of all authors and does grant on behalf of all authors, an exclusive licence (or non-exclusive for government employees) on a worldwide basis to the BMJ Publishing Group Ltd and its Licensees to permit this article (if accepted) to be published in Annals of the Rheumatic Diseases and any other BMJPGL products to exploit all subsidiary rights, as set out in their licence (http://ard.bmj.com/ifora/licence.pdf). http://ard.bmj.com/ on September 29, 2021 by guest. Protected copyright. 1 Copyright Article author (or their employer) 2009. -

Reshuffling Genomic Landscapes to Study the Regulatory Evolution of Hox Gene Clusters
Reshuffling genomic landscapes to study the regulatory evolution of Hox gene clusters Patrick Tschoppa, Nadine Fraudeaua, Frédérique Bénab, and Denis Duboulea,c,1 aNational Research Centre Frontiers in Genetics, Department of Genetics and Evolution, University of Geneva, Sciences III, 1211 Geneva 4, Switzerland; bService of Genetic Medicine, Centre Médical Universitaire, 1211 Geneva 4, Switzerland; and cSchool of Life Sciences, Ecole Polytechnique Fédérale, Lausanne, 1015 Lausanne, Switzerland Edited by Matthew P. Scott, Stanford University/Howard Hughes Medical Institute, Stanford, CA, and approved May 23, 2011 (received for review February 22, 2011) The emergence of Vertebrata was accompanied by two rounds of this genomic context. As a result, these latter genes show similar whole-genome duplications. This enabled paralogous genes to expression in the developing hands and feet by means of a by- acquire novel functions with high evolutionary potential, a process stander effect, with only minor functional relevance for these suggested to occur mostly by changes in gene regulation, rather structures (16, 18). than in protein sequences. In the case of Hox gene clusters, such Recently, we reported that these enhancer sequences located duplications favored the appearance of distinct global regulations. centromeric to HoxD and driving these distal limb expression To assess the impact of such “regulatory evolution” upon neo- patterns, are absolutely necessary, as shown by an inversion tak- functionalization, we developed PANTHERE (PAN-genomic Trans- ing these elements away from an otherwise intact HoxD cluster. location for Heterologous Enhancer RE-shuffling) to bring the en- This inverted allele phenocopied the digit defects observed after tire megabase-scale HoxD regulatory landscape in front of the a deletion of all posterior Hoxd genes (19). -

BMC Biology Biomed Central
BMC Biology BioMed Central Research article Open Access Classification and nomenclature of all human homeobox genes PeterWHHolland*†1, H Anne F Booth†1 and Elspeth A Bruford2 Address: 1Department of Zoology, University of Oxford, South Parks Road, Oxford, OX1 3PS, UK and 2HUGO Gene Nomenclature Committee, European Bioinformatics Institute (EMBL-EBI), Wellcome Trust Genome Campus, Hinxton, Cambridgeshire, CB10 1SA, UK Email: Peter WH Holland* - [email protected]; H Anne F Booth - [email protected]; Elspeth A Bruford - [email protected] * Corresponding author †Equal contributors Published: 26 October 2007 Received: 30 March 2007 Accepted: 26 October 2007 BMC Biology 2007, 5:47 doi:10.1186/1741-7007-5-47 This article is available from: http://www.biomedcentral.com/1741-7007/5/47 © 2007 Holland et al; licensee BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Abstract Background: The homeobox genes are a large and diverse group of genes, many of which play important roles in the embryonic development of animals. Increasingly, homeobox genes are being compared between genomes in an attempt to understand the evolution of animal development. Despite their importance, the full diversity of human homeobox genes has not previously been described. Results: We have identified all homeobox genes and pseudogenes in the euchromatic regions of the human genome, finding many unannotated, incorrectly annotated, unnamed, misnamed or misclassified genes and pseudogenes. -

Gene Regulation Gravitates Toward Either Addition Or Multiplication When Combining the Effects of Two Signals
RESEARCH ARTICLE Gene regulation gravitates toward either addition or multiplication when combining the effects of two signals Eric M Sanford1, Benjamin L Emert1, Allison Cote´ 2,3, Arjun Raj2,3* 1Genomics and Computational Biology Graduate Group, Perelman School of Medicine, University of Pennsylvania, Philadelphia, United States; 2Department of Bioengineering, School of Engineering and Applied Sciences, University of Pennsylvania, Philadelphia, United States; 3Department of Genetics, Perelman School of Medicine, University of Pennsylvania, Philadelphia, United States Abstract Two different cell signals often affect transcription of the same gene. In such cases, it is natural to ask how the combined transcriptional response compares to the individual responses. The most commonly used mechanistic models predict additive or multiplicative combined responses, but a systematic genome-wide evaluation of these predictions is not available. Here, we analyzed the transcriptional response of human MCF-7 cells to retinoic acid and TGF-b, applied individually and in combination. The combined transcriptional responses of induced genes exhibited a range of behaviors, but clearly favored both additive and multiplicative outcomes. We performed paired chromatin accessibility measurements and found that increases in accessibility were largely additive. There was some association between super-additivity of accessibility and multiplicative or super-multiplicative combined transcriptional responses, while sub-additivity of accessibility associated with additive transcriptional responses. Our findings suggest that mechanistic models of combined transcriptional regulation must be able to reproduce a range of behaviors. *For correspondence: [email protected] Introduction Suppose a cell at baseline expresses 100 copies of mRNA of gene X. If you give signal A, the cell Competing interest: See expresses 200 copies of gene X. -

Supplementary File Table S1
Supplementary file Table S1 Grouping of Homeobox genes according to their main known function. Anatomical Structure Morphogenesis EN1, HOXC10, HOXC13, HOXD3, LBX1, SIX2, SIX4 Organ Morphogenesis CDX1, CDX2, HOXA11, HOXA13, ISL1, LHX1, PAX3, PDHX, PITX2, PITX3, PROX1, SIX6 Body Pattern Formation ALX3, EMX2, HHEX, HOXA11, HOXA2, HOXA4, HOXA5, HOXA6, HOXB1, HOXB5, HOXB6, HOXC5, HOXD10, HOXD8, LMX1B, PITX2 Ectoderm Development PROX1, VAX2 Endoderm Development HOXC11 Brain & Nervous System Development Brain Development ALX1, DLX2, EMX2 Nervous System Development: ARX, DLX5, DLX6, HOXD10, LBX1, LHX1, OTP, PAX3, PHOX2A, PHOX2B Skeletal Development: ALX3, ALX4, DLX3, DLX5, DLX6, EN1, HOXA11, HOXA13, HOXA2, HOXB6, HOXD10, HOXD13, MSX2 Muscle Development: BARX2, MKX, SIRT1, SIRT2, SIX1 Other Homeobox Genes Involved In BARX1, CDX4, CUX1, DLX1, EMX1, EN2, Multicellular Organismal HOXA1, HOXA7, HOXA9, HOXB13, HOXB2, Development: HOXB3, HOXB4, HOXB7, HOXB8, HOXB9, HOXC12, HOXC8, HOXC9, HOXD1, HOXD11, HOXD12, HOXD9, ISL2, LBX2, LMX1A, MEIS1, NKX3-1, OTX1, TLX1, VAX1, VSX1, VSX2 Homeobox Genes Involved In Cell ARX, EMX2, HHEX, HLX, HOPX, LBX1, LHX1, Differentiation: LMX1B, MIXL1, OTP, PHOX2A, SIRT1, VSX2 Other Genes: PHTF1, SIRT3, SIRT6, SIRT7, ZHX1, ZHX2 Homeobox genes include two subsets of genes coding for transcription factors involved in multiple functions. The clustered HOX genes are indicated in bold. Supplementary file Figure S2 5’ Spatial collinearity 3’ HOXA Chr. 7p15.3 HOXB Chr. 17q21.3 HOXC Chr. 12q13.3 HOXD Chr. 2q31 13 12 11 10 9 8 7 6 5 4 3 2 1 Paralogous HOX groups Distribution of the 39 human HOX genes in four clusters located in different chromosomal regions*. Blue indicates anterior HOX genes. Yellow, paralogy group 3 Hox genes, green and purple indicatete central HOX genes and Red the posterior HOX genes. -

The Enigmatic HOX Genes: Can We Crack Their Code?
cancers Review The Enigmatic HOX Genes: Can We Crack Their Code? Zhifei Luo, Suhn K. Rhie and Peggy J. Farnham * Department of Biochemistry and Molecular Medicine and the Norris Comprehensive Cancer Center, Keck School of Medicine, University of Southern California, Los Angeles, CA 90089, USA; [email protected] (Z.L.); [email protected] (S.K.R.) * Correspondence: [email protected]; Tel.: +323-442-8015 Received: 2 February 2019; Accepted: 1 March 2019; Published: 7 March 2019 Abstract: Homeobox genes (HOX) are a large family of transcription factors that direct the formation of many body structures during early embryonic development. There are 39 genes in the subgroup of homeobox genes that constitute the human HOX gene family. Correct embryonic development of flies and vertebrates is, in part, mediated by the unique and highly regulated expression pattern of the HOX genes. Disruptions in these fine-tuned regulatory mechanisms can lead to developmental problems and to human diseases such as cancer. Unfortunately, the molecular mechanisms of action of the HOX family of transcription factors are severely under-studied, likely due to idiosyncratic details of their structure, expression, and function. We suggest that a concerted and collaborative effort to identify interacting protein partners, produce genome-wide binding profiles, and develop HOX network inhibitors in a variety of human cell types will lead to a deeper understanding of human development and disease. Within, we review the technological challenges and possible approaches needed to achieve this goal. Keywords: HOX; ChIP-seq; cancer biomarkers; targeted therapy 1. Introduction The homeobox gene family is the second largest family of transcription factors encoded in the human genome and consists of an estimated 257 genes [1], each of which contains a 183-nucleotide sequence that encodes a 61-amino acid homeodomain that forms a helix-turn-helix structure. -

The Changing Chromatome As a Driver of Disease: a Panoramic View from Different Methodologies
The changing chromatome as a driver of disease: A panoramic view from different methodologies Isabel Espejo1, Luciano Di Croce,1,2,3 and Sergi Aranda1 1. Centre for Genomic Regulation (CRG), Barcelona Institute of Science and Technology, Dr. Aiguader 88, Barcelona 08003, Spain 2. Universitat Pompeu Fabra (UPF), Barcelona, Spain 3. ICREA, Pg. Lluis Companys 23, Barcelona 08010, Spain *Corresponding authors: Luciano Di Croce ([email protected]) Sergi Aranda ([email protected]) 1 GRAPHICAL ABSTRACT Chromatin-bound proteins regulate gene expression, replicate and repair DNA, and transmit epigenetic information. Several human diseases are highly influenced by alterations in the chromatin- bound proteome. Thus, biochemical approaches for the systematic characterization of the chromatome could contribute to identifying new regulators of cellular functionality, including those that are relevant to human disorders. 2 SUMMARY Chromatin-bound proteins underlie several fundamental cellular functions, such as control of gene expression and the faithful transmission of genetic and epigenetic information. Components of the chromatin proteome (the “chromatome”) are essential in human life, and mutations in chromatin-bound proteins are frequently drivers of human diseases, such as cancer. Proteomic characterization of chromatin and de novo identification of chromatin interactors could thus reveal important and perhaps unexpected players implicated in human physiology and disease. Recently, intensive research efforts have focused on developing strategies to characterize the chromatome composition. In this review, we provide an overview of the dynamic composition of the chromatome, highlight the importance of its alterations as a driving force in human disease (and particularly in cancer), and discuss the different approaches to systematically characterize the chromatin-bound proteome in a global manner.