United States Patent Office
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Midterm 2 Examination Key
GLY4200C Name 90 points October 26, 2011 16 took exam - Numbers to the left of the question number in red are the number of incorrect responses. Instructor comments are in blue. Florida Atlantic University MINERALOGY -- MIDTERM 2 EXAMINATION KEY True-False - Print the letter T or F in the blank to indicate if each of the following statements is true or false. Illegible answers are wrong. (1 point each) 0 T 1. All mathematical groups, including crystallographic point and space groups, must contain an identity operation. 2 F 2. All pyramidal forms are closed. 11 F 3. As nuclei grow, their surface area/volume ratio increases. 1 F 4. Growth twins are always penetration twins. 2 T 5. Low-spin Fe3+ is smaller than high-spin Fe3+. 0 T 6.As pressure increases, minerals transform to denser structures, with atoms packed more closely together 1 T 7. Although silicon normally has CN = IV, in the lower mantle the pressures are so great that the CN of silicon is VI. 2 F 8. If two ions have the same charge, the larger ion will enter a crystal structure preferentially. 2 T 9. Crystal classes are defined by the thirty-two possible point groups. 11 F 10. Possible symmetry operations include rotation axes, mirror planes, and an inversion center. What is wrong with this statement? 0 T 11. Faces composed of all anions or all cations are very high energy. They attract ions of opposite sign, grow rapidly, and may grow themselves out of existence. 1 T 12. The calculation of the zone axis is done using the same method as a vector cross- product. -

Economic Diversification Plan for East Kern County
FEBRUARY 2017 ECONOMIC DIVERSIFICATION PLAN EAST KERN COUNTY, CA KERN COUNTY ACKNOWLEDGEMENTS TIP Strategies would like to thank the many individuals who participated in the creation of this Economic Diversification Plan. We are especially grateful to the members of the East Kern Economic Alliance who contributed their time and input throughout this planning process. Their expertise helped us gain a deep understanding of the region’s range of assets and opportunities. We are also thankful for the US Department of Defense Office of Economic Adjustment for their visionary investments and support which helped make this project a reality. We also want to thank the leadership and staff of Kern County and the Kern EDC for their essential support, guidance, and feedback during the development of this plan. EAST KERN ECONOMIC ALLIANCE KEY STAKEHOLDERS Richard Chapman, President & CEO Jennifer Wood, Mayor Gary Parsons, Economic Kern Economic Development California City Development Manager Corporation City of Ridgecrest Karina Drees, CEO and General Bill Deaver, Michelle Vance, Economic Manager Edwards Community Alliance Development Coordinator Mojave Air & Spaceport City of Tehachapi Kimberly Maevers, President Kelly Bearden, Director Ryan Rush, Field Representative Greater Antelope Valley Economic CSU-Bakersfield SBDC Kern County Board of Supervisors, Alliance 2nd District KERN COUNTY PROJECT TEAM Supervisor Zach Scrivner, Chairman of the Kern County Board of Supervisors & 2nd District Supervisor Teresa Hitchcock, Assistant County Administrative -

THE MAIN-GROUP ELEMENTS Atomic Properties
THE MAIN-GROUP ELEMENTS Atomic Properties Properties of elements is based valence-shell electron configurations. Five atomic properties: 1. atomic radius 2. ionization energy 3. electron affinity 4. electronegativity 5. polarizability Atomic Size Adding more valence levels increases the distance from the electrons to the nucleus, it also decrease the effect the nucleus has on the outer electrons. decreases across a period Effective Nuclear Force increases down a group Valence levels Ionization Energy Ionization energy: the energy required to remove the most loosely held electron from an atom in the gaseous state. Size of Atom Ionization Energy Smaller size Higher ionization energy Electron Affinity The electron affinity is a release of energy to form an anion. Ionization Energy Electron Affinity Atoms with higher effective nuclear charges release more energy. Electronegativity Electronegativity is the tendency of an atom to attract electrons to itself. Ionization Energy Electronegativity Atoms with a greater effective nuclear charge have a stronger pull on bonding electrons. Large differences in electronegativity typically form ionic bonds and small differences from covalent bonds. Polarizability Polarizability is the ease an atoms electron cloud can be distorted. Size of Atom Polarizability Electron-rich, heavier atoms are easily polarized. High polarizing power atom-are small sized, highly charged atoms. Bonding Trends Elements in period 2 strictly follow the octet rule; carbon forms four bonds. Elements in Period 3 and higher periods exceed the octet. They reach higher oxidation states because they can access empty d-orbitals. Also, larger atoms can simply make more bonds to more neighbors . Group 1A: Alkali Metals The chemical properties of alkali metals are striking similar. -

Origin of the Kramer Borax Deposit, Boron, CA
A 50 year retrospective 1 OUTLINE 1. A brief history of borax 2. Kramer borax deposit a) Setting and Discovery b) Mineralogy of sedimentary borates c) Stratigraphy and Lithology d) Petrography and implications for geologic setting e) Solubility studies and modeling lake characteristics f) Comparable modern analogues 3. New evidence a) Turkish and Argentinian deposits b) Boron isotopic studies 4. Broader questions – Source water controls (thermal springs), B-As-Sb association, igneous-metamorphic controls on boron in thermal waters 2 Why give this talk? 1. Old (but rusty) material to me, new to most of you 2. Desire to see if ideas have changed in the past 50+ years. 3. Citation of my work even today suggests I did something right. 4. Wish to compare Kramer work with evidence from newer borate deposits in Turkey and South America 5. A wish to evaluate these ideas in light of new evidence using tools that weren’t available in 1964 6. A chance to ponder broader questions about boron’s geochemical cycle. 7. Work done so long ago that if you ask penetrating questions I can always plead a “senior moment” 3 What was unique about my research on the Kramer deposits? • Used a combination of geological tools (Field AND lab work – rare in 1964) • Stratigraphy, Petrography, and XRD based mineralogy • Experimental solubility studies of effects of other salts on Na-borate solubilities • Field studies of other possible borate environments (Borax Lake, Teels and Columbus Marsh, NV, Death Valley, Searles Lake) • Benefits of discussions with an all-star support team with similar interests (Mary Clark, Blair Jones, G.I. -

Temperature and Relative Humidity (RH) Using Saturated Salt Solutions
THERMODYNAMICS OF 1:2 SODIUM BORATE MINERALS By LAURA SUZANNE RUHL A THESIS PRESENTED TO THE GRADUATE SCHOOL OF THE UNIVERSITY OF FLORIDA IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF MASTER OF SCIENCE UNIVERSITY OF FLORIDA 2008 1 © 2008 Laura Suzanne Ruhl 2 To my family, who have kept my inquisitive spirit and thirst for knowledge alive, always encouraging me, and giving me strength. 3 ACKNOWLEDGMENTS First and foremost, I thank the chair of my committee, Phil Neuhoff, for all of his time and all of the knowledge he has bestowed upon me throughout my time as his student. I would also like to thank my committee members, Ellen Martin and Guerry McClellan, for their time and support in my academic endeavors. I thank Jie Wang and Gökce Atalan for their help in the lab, for many scientific discussions and support. I also thank Derrick Newkirk for his unending encouragement and support. Major thanks go to my parents and siblings for always being there to help in any endeavor and their never-ending support and love. Lastly, I appreciate all of the never-ending encouragement from my friends and family. The National Science Foundation funded this project. 4 TABLE OF CONTENTS page ACKNOWLEDGMENTS ...............................................................................................................4 LIST OF TABLES...........................................................................................................................6 LIST OF FIGURES .........................................................................................................................7 -

Kernite, a New Sodium Borate ::
THE AMERICAN MINERA LOGIST KERNITE, A NEW SODIUM BORATE :: .:. War.rBuan T. ScnRr,rrin, U. S. GeologicalSaraey, Washingtoh D. C. The new mineral kernite, Na2B4OT'4HsO,was receivedthrough Hoyt S. Gale of Los Angeles, California, and is reported to occur in quantity in the southeast corner of Kern County, California, in the -The Kratner boron district. name kernite is proposed after the county name. The exact locality is about 4 miles north of Rich, a station on the Santa Fe R. R. and about 25 miles directly east of Mojave, being in Sec.22, T. 11 N., R. 8W. The mineral was discovered in exploratory borings at a depth of several hundred feet. Ulexite and possibly colemanite are associated rninerals. The geology of this field has been described by Galel and Noble.z The writer has a cleavage piece of kernite 2 by 2-l/2 by 6 inches. Apparently single crystals are of large size though no terminated crystals have so far been seen. The mineral is colorless to white, transparent, vitreous to pearly in luster, and greatly resembles some specimens of massive cleavable selenite. Ortho- rhombic. Perfect prismatic cleavage, rtuAm:71" 08'. Readily breaksinto long thin fibersandlaths. H:about 3. Sp. Gr.: 1.953. Optically probably negative, although 2V is nearly 90o and sign uncertain. Elongation (c axis) positive. Axial plane is parallel to elongation. a:I.454, 0:I.472, t:1.488. Before the blowpipe it swells considerably and finally fuses to a clear glass. Very slowly soluble in cold water, the clear cleavage pieces first be- coming opaque white due to abundant etch figures. -

Diamond Dan's Mineral Names Dictionary
A Dictionary of Mineral Names By Darryl Powell Mineral Names What do they mean? Who created them? What can I learn from them? This mineral diction‐ ary is unique because it is illustrated, both with mineral drawings as well as pictures of people and places after which some minerals are named. The people pictured on this page have all made a con‐ tribution to what is formally called “mineral nomenclature.” Keep reading and you will discover who they are and what they did. In 1995, Diamond Dan Publications pub‐ lished its first full book, “A Mineral Collector’s Guide to Common Mineral Names: Their Ori‐ gins & Meanings.” Now it is twenty years later. What you will discover in this issue and in the March issue is a re‐ vised and improved version of this book. This Mineral Names Dictionary contains mineral names that the average mineral collector will encounter while collecting minerals, attending shows and visiting museum displays. In addition to the most common min‐ eral names, there are some unofficial names which you will still find on labels. Each mineral name has a story to tell or a lesson to teach. If you wanted to take the time, each name could become a topic to study. Armalcolite, for example, could quickly be‐ come a study of a mineral, first discovered on the moon, and brought back to earth by the astronauts Armstrong, Aldrin and Collins (do you see parts of their names in this mineral name?) This could lead you to a study of American astronauts landing on the moon, what it took to get there and what we discovered by landing on the moon. -

The Elements.Pdf
A Periodic Table of the Elements at Los Alamos National Laboratory Los Alamos National Laboratory's Chemistry Division Presents Periodic Table of the Elements A Resource for Elementary, Middle School, and High School Students Click an element for more information: Group** Period 1 18 IA VIIIA 1A 8A 1 2 13 14 15 16 17 2 1 H IIA IIIA IVA VA VIAVIIA He 1.008 2A 3A 4A 5A 6A 7A 4.003 3 4 5 6 7 8 9 10 2 Li Be B C N O F Ne 6.941 9.012 10.81 12.01 14.01 16.00 19.00 20.18 11 12 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 3 Na Mg IIIB IVB VB VIB VIIB ------- VIII IB IIB Al Si P S Cl Ar 22.99 24.31 3B 4B 5B 6B 7B ------- 1B 2B 26.98 28.09 30.97 32.07 35.45 39.95 ------- 8 ------- 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 4 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 39.10 40.08 44.96 47.88 50.94 52.00 54.94 55.85 58.47 58.69 63.55 65.39 69.72 72.59 74.92 78.96 79.90 83.80 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 5 Rb Sr Y Zr NbMo Tc Ru Rh PdAgCd In Sn Sb Te I Xe 85.47 87.62 88.91 91.22 92.91 95.94 (98) 101.1 102.9 106.4 107.9 112.4 114.8 118.7 121.8 127.6 126.9 131.3 55 56 57 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86 6 Cs Ba La* Hf Ta W Re Os Ir Pt AuHg Tl Pb Bi Po At Rn 132.9 137.3 138.9 178.5 180.9 183.9 186.2 190.2 190.2 195.1 197.0 200.5 204.4 207.2 209.0 (210) (210) (222) 87 88 89 104 105 106 107 108 109 110 111 112 114 116 118 7 Fr Ra Ac~RfDb Sg Bh Hs Mt --- --- --- --- --- --- (223) (226) (227) (257) (260) (263) (262) (265) (266) () () () () () () http://pearl1.lanl.gov/periodic/ (1 of 3) [5/17/2001 4:06:20 PM] A Periodic Table of the Elements at Los Alamos National Laboratory 58 59 60 61 62 63 64 65 66 67 68 69 70 71 Lanthanide Series* Ce Pr NdPmSm Eu Gd TbDyHo Er TmYbLu 140.1 140.9 144.2 (147) 150.4 152.0 157.3 158.9 162.5 164.9 167.3 168.9 173.0 175.0 90 91 92 93 94 95 96 97 98 99 100 101 102 103 Actinide Series~ Th Pa U Np Pu AmCmBk Cf Es FmMdNo Lr 232.0 (231) (238) (237) (242) (243) (247) (247) (249) (254) (253) (256) (254) (257) ** Groups are noted by 3 notation conventions. -

Download PDF About Minerals Sorted by Mineral Group
MINERALS SORTED BY MINERAL GROUP Most minerals are chemically classified as native elements, sulfides, sulfates, oxides, silicates, carbonates, phosphates, halides, nitrates, tungstates, molybdates, arsenates, or vanadates. More information on and photographs of these minerals in Kentucky is available in the book “Rocks and Minerals of Kentucky” (Anderson, 1994). NATIVE ELEMENTS (DIAMOND, SULFUR, GOLD) Native elements are minerals composed of only one element, such as copper, sulfur, gold, silver, and diamond. They are not common in Kentucky, but are mentioned because of their appeal to collectors. DIAMOND Crystal system: isometric. Cleavage: perfect octahedral. Color: colorless, pale shades of yellow, orange, or blue. Hardness: 10. Specific gravity: 3.5. Uses: jewelry, saws, polishing equipment. Diamond, the hardest of any naturally formed mineral, is also highly refractive, causing light to be split into a spectrum of colors commonly called play of colors. Because of its high specific gravity, it is easily concentrated in alluvial gravels, where it can be mined. This is one of the main mining methods used in South Africa, where most of the world's diamonds originate. The source rock of diamonds is the igneous rock kimberlite, also referred to as diamond pipe. A nongem variety of diamond is called bort. Kentucky has kimberlites in Elliott County in eastern Kentucky and Crittenden and Livingston Counties in western Kentucky, but no diamonds have ever been discovered in or authenticated from these rocks. A diamond was found in Adair County, but it was determined to have been brought in from somewhere else. SULFUR Crystal system: orthorhombic. Fracture: uneven. Color: yellow. Hardness 1 to 2. -

Investigation of Kinetic Parameters of Dehydrated Ulexite Mineral in Boric Acide Solution
Araştırma Makalesi / Research Article Iğdır Üni. Fen Bilimleri Enst. Der. / Iğdır Univ. J. Inst. Sci. & Tech. 8(2): 149-157, 2018 Investigation of Kinetic Parameters of Dehydrated Ulexite Mineral in Boric Acide Solution Mehmet Harbi ÇALIMLI1, Mehmet TUNÇ2 ABSTRACT: Ulexite used in the production of boron compounds is an important mineral. In this work, the kinetic parameters of dehydrated ulexite mineral and orginal ulexite in boric acid solution were investigated. According to the DTA diagram, it was seen that the mineral did not change the structure until around 170 oC. It was observed that the dissolution kinetics increased to this temperature. In the SEM images, it was observed that the number of pores in the mineral was increased until 170 oC and afterwards number of pores was decreased. The kinetic parameters of the original ulexite were evaluated. Study indicates that the dissolution kinetics model in the boric acide was found as below; -1 -1,013 0,4579 -1 -0650 0,0545 -2649,12 -1 1-(1-X) = [2.02(Rp) [CA]0 (S.L ) (M.S) e T ].t Keywords: Boric acide, dissolution rate, ulexite, kinetic parameters Iğdır Üniversitesi Fen Bilimleri Enstitüsü Dergisi Iğdır Iğdır University Journal of the Institute of Science and Technology Iğdır University Journal of the Institute Science and Technology Hidrate Edimiş Uleksit Mineralinin Borik Asit Çözeltisindeki Kinetik Parametrelerinin İncelenmesi ÖZET: Bor bileşiklerinin üretiminde kullanılan Uleksit çok önemli bir bor mineralidir. Bu çalışmada hidrate edilmiş uleksit minerali ve orginal mineralinin borik asit çözeltisindeki kinetik parametreleri incelenmiştir. DTA diyagramına göre 170 oC’ye kadar mineral yapısında değişim olmadığı görülmüştür. -
Chapter 3 Molecular Shape and Structure
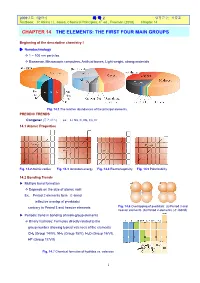
2009년도 제2학기 화 학 2 담당교수: 신국조 Textbook: P. Atkins / L. Jones, Chemical Principles, 4th ed., Freeman (2008) Chapter 14 CHAPTER 14 THE ELEMENTS: THE FIRST FOUR MAIN GROUPS Beginning of the descriptive chemistry ! ▶ Nanotechnology Æ 1 ~ 100 nm particles Æ Biosensor, Microscopic computers, Artificial bones, Light weight, strong materials Fig. 14.1 The relative abundances of the principal elements. PREODIC TRENDS Congener (동족원소) ex. Li, Na, K, Rb, Cs, Fr 14.1 Atomic Properties Fig. 14.2 Atomic radius Fig. 14.3 Ionization energy Fig. 14.4 Electronegativity Fig. 14.5 Polarizability 14.2 Bonding Trends ▶ Multiple bond formation Æ Depends on the size of atomic radii Ex. Period 2 elements form π -bond (effective overlap of p-orbitals) contrary to Period 3 and heavier elements Fig. 14.6 Overlapping of p-orbitals: (a) Period 3 and heavier elements. (b) Period 2 elements (π -bond) ▶ Periodic trend in bonding of main-group elements Æ Binary hydrides: Formulas directly related to the group number showing typical valences of the elements CH4 (Group 14/IV), NH3 (Group 15/V), H2O (Group 16/VI), HF (Group 17/VII) Fig. 14.7 Chemical formulas of hydrides vs. valences 1 2009년도 제2학기 화 학 2 담당교수: 신국조 Textbook: P. Atkins / L. Jones, Chemical Principles, 4th ed., Freeman (2008) Chapter 14 ▶ Binary hydrides Saline hydrides (이온성 수소화물): s-block elements, white, high m.p., crystals, portable fuel, H– ∆ 2 K(sg) +⎯H2 ( ) ⎯→2 KH(s) Metallic hydrides: d-block elements, black, conducting powder, portable fuel ∆ 2 Cu(sg) +⎯H2 ( ) ⎯→2 CuH(s) Molecular hydrides: non-metal elements, low melting volatile Brønsted acid NH3, HX, hydrocarbons Fig. -
Kernite from Tincalayu, Salta, Argentina
American Mineralogist, Volume 58, pages 308'313, 1973 Kernitefrom Tincalayu,Salta, Argentina C. S. Hunrsut, Jn. D epartment ol Geological Sciences, Hansmd (Jniuersity, Cambridge, Massochusetts02138r, L. F. AnrsrAn^crN ObseraatorioNacional De Fisica Cosmica, San Miguel, Pcia. B. A., Argentina AND R. C. Enu U.S. Geological Suroey, Menlo Park, Calilornia 940252 Abshact The Tincalayu mine (Salta, Argentina) is essentially a monomineralic deposit of borax partly transformed to kernite with minor amounts of several rare borates. The deposit is interpreted as an old playa accumulation, buried, metamorphosed and deformed by folding and faulting. It is intercalated with clastic continental sediments of Pliocene or post-Pliocene age. The principal explored lens is more than 30 m thick and approximately 100 m in diameter. Kernite crystals are clear, transparent and well developed; many exceed 30 cm in length and l0 crn across; no terminal faces were observed. X-ray powder data are given with the orienta- - - -+ tion: c > a, p 2 9O'.The spacegroup is Ph/c; a 7.016 ! 0.001A, b 9.152 0.0024; - - c = 15.678 -{- 0.002A. p - 108"52.8' !. o.7'; cell volume 952.6 -r 0.2A3; Z 4. a:b:c 0.7666:l:1.7131. Cleavage:{100} and {00I} perfect, {TOZ} poor. Optically biaxial (-);a: 1.545,P-1.473,.y-1.488-F0.001(Nalight);2V-80",r)o(slight);XAc-70'30', Z - b.Density (calc) 1.905 + 0.001 g/cma, specificgravity (meas) 1.906 = 0.003. A chemical analysisby wet methods gives wt percent Na,O 23.1, B,O, 51'0, and H'O 26.1 (total 100.2).