Introduction
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Electrolytic Cells
CHEMISTRY LEVEL 4C (CHM415115) ELECTROLYTIC CELLS THEORY SUMMARY & REVISION QUESTIONS (CRITERION 5) ©JAK DENNY Tasmanian TCE Chemistry Revision Guides by Jak Denny are licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. INDEX: PAGES • INTRODUCTORY THEORY 3 • COMPARING ENERGY CONVERSIONS 4 • APPLICATIONS OF ELECTROLYSIS 4 • ELECTROPLATING 5 • COMPARING CELLS 6 • THE ELECTROCHEMICAL SERIES 7 • PREDICTING ELECTROLYSIS PRODUCTS 8-9 • ELECTROLYSIS PREDICTION FLOWCHART 1 0 • ELECTROLYSIS PREDICTION EXAMPLES 11 • ELECTROLYSIS PREDICTION QUESTIONS 12 • INDUSTRIAL ELECTROLYTIC PROCESSES 13 • IMPORTANT ELECTRICAL THEORY 14 • FARADAY’S ELECTROLYSIS LAWS 15 • QUANTITATIVE ELECTROLYSIS 16 • CELLS IN SERIES 17 • ELECTROLYSIS QUESTIONS 18-20 • ELECTROLYSIS TEST QUESTIONS 21-22 • TEST ANSWERS 23 2 ©JAK CHEMISTRY LEVEL 4C (CHM415115) ELECTROLYTIC CELLS (CRITERION 5) INTRODUCTION: In our recent investigation of electrochemical cells, we encountered spontaneous redox reactions that release electrical energy such as takes place in the familiar situations of “batteries”. When a car battery is ‘flat’ and needs to be recharged, a power supply (‘battery charger’) is connected to the flat battery and chemical changes take place and it is subsequently able to operate again as a power supply. The ‘recharging’ process is non-spontaneous and requires an input of energy to occur. When a cell is such that an energy input is required to make a non-spontaneous redox reaction take place, we describe the cell as an ELECTROLYTIC CELL. The redox process occurring in an ELECTROLYTIC CELL is referred to as ELECTROLYSIS. For example, consider the spontaneous redox reaction associated with an electrochemical (fuel) cell; i.e. 2H2(g) + O2(g) → 2H2O(l) + ELECTRICAL ENERGY This chemical reaction RELEASES energy which can be used to power an electric motor, to drive a machine, appliance or car. -

Electrochemical Cells
Electrochemical cells = electronic conductor If two different + surrounding electrolytes are used: electrolyte electrode compartment Galvanic cell: electrochemical cell in which electricity is produced as a result of a spontaneous reaction (e.g., batteries, fuel cells, electric fish!) Electrolytic cell: electrochemical cell in which a non-spontaneous reaction is driven by an external source of current Nils Walter: Chem 260 Reactions at electrodes: Half-reactions Redox reactions: Reactions in which electrons are transferred from one species to another +II -II 00+IV -II → E.g., CuS(s) + O2(g) Cu(s) + SO2(g) reduced oxidized Any redox reactions can be expressed as the difference between two reduction half-reactions in which e- are taken up Reduction of Cu2+: Cu2+(aq) + 2e- → Cu(s) Reduction of Zn2+: Zn2+(aq) + 2e- → Zn(s) Difference: Cu2+(aq) + Zn(s) → Cu(s) + Zn2+(aq) - + - → 2+ More complex: MnO4 (aq) + 8H + 5e Mn (aq) + 4H2O(l) Half-reactions are only a formal way of writing a redox reaction Nils Walter: Chem 260 Carrying the concept further Reduction of Cu2+: Cu2+(aq) + 2e- → Cu(s) In general: redox couple Ox/Red, half-reaction Ox + νe- → Red Any reaction can be expressed in redox half-reactions: + - → 2 H (aq) + 2e H2(g, pf) + - → 2 H (aq) + 2e H2(g, pi) → Expansion of gas: H2(g, pi) H2(g, pf) AgCl(s) + e- → Ag(s) + Cl-(aq) Ag+(aq) + e- → Ag(s) Dissolution of a sparingly soluble salt: AgCl(s) → Ag+(aq) + Cl-(aq) − 1 1 Reaction quotients: Q = a − ≈ [Cl ] Q = ≈ Cl + a + [Ag ] Ag Nils Walter: Chem 260 Reactions at electrodes Galvanic cell: -

Towards the Hydrogen Economy—A Review of the Parameters That Influence the Efficiency of Alkaline Water Electrolyzers
energies Review Towards the Hydrogen Economy—A Review of the Parameters That Influence the Efficiency of Alkaline Water Electrolyzers Ana L. Santos 1,2, Maria-João Cebola 3,4,5 and Diogo M. F. Santos 2,* 1 TecnoVeritas—Serviços de Engenharia e Sistemas Tecnológicos, Lda, 2640-486 Mafra, Portugal; [email protected] 2 Center of Physics and Engineering of Advanced Materials (CeFEMA), Instituto Superior Técnico, Universidade de Lisboa, 1049-001 Lisbon, Portugal 3 CBIOS—Center for Research in Biosciences & Health Technologies, Universidade Lusófona de Humanidades e Tecnologias, Campo Grande 376, 1749-024 Lisbon, Portugal; [email protected] 4 CERENA—Centre for Natural Resources and the Environment, Instituto Superior Técnico, Universidade de Lisboa, Av. Rovisco Pais, 1049-001 Lisbon, Portugal 5 Escola Superior Náutica Infante D. Henrique, 2770-058 Paço de Arcos, Portugal * Correspondence: [email protected] Abstract: Environmental issues make the quest for better and cleaner energy sources a priority. Worldwide, researchers and companies are continuously working on this matter, taking one of two approaches: either finding new energy sources or improving the efficiency of existing ones. Hydrogen is a well-known energy carrier due to its high energy content, but a somewhat elusive one for being a gas with low molecular weight. This review examines the current electrolysis processes for obtaining hydrogen, with an emphasis on alkaline water electrolysis. This process is far from being new, but research shows that there is still plenty of room for improvement. The efficiency of an electrolyzer mainly relates to the overpotential and resistances in the cell. This work shows that the path to better Citation: Santos, A.L.; Cebola, M.-J.; electrolyzer efficiency is through the optimization of the cell components and operating conditions. -

Electrode Reactions and Kinetics
CHEM465/865, 2004-3, Lecture 14-16, 20 th Oct., 2004 Electrode Reactions and Kinetics From equilibrium to non-equilibrium : We have a cell, two individual electrode compartments, given composition, molar reaction Gibbs free ∆∆∆ energy as the driving force, rG , determining EMF Ecell (open circuit potential) between anode and cathode! In other words: everything is ready to let the current flow! – Make the external connection through wires. Current is NOT determined by equilibrium thermodynamics (i.e. by the composition of the reactant mixture, i.e. the reaction quotient Q), but it is controlled by resistances, activation barriers, etc Equilibrium is a limiting case – any kinetic model must give the correct equilibrium expressions). All the involved phenomena are generically termed ELECTROCHEMICAL KINETICS ! This involves: Bulk diffusion Ion migration (Ohmic resistance) Adsorption Charge transfer Desorption Let’s focus on charge transfer ! Also called: Faradaic process . The only reaction directly affected by potential! An electrode reaction differs from ordinary chemical reactions in that at least one partial reaction must be a charge transfer reaction – against potential-controlled activation energy, from one phase to another, across the electrical double layer. The reaction rate depends on the distributions of species (concentrations and pressures), temperature and electrode potential E. Assumption used in the following: the electrode material itself (metal) is inert, i.e. a catalyst. It is not undergoing any chemical transformation. It is only a container of electrons. The general question on which we will focus in the following is: How does reaction rate depend on potential? The electrode potential E of an electrode through which a current flows differs from the equilibrium potential Eeq established when no current flows. -

3 PRACTICAL APPLICATION BATTERIES and ELECTROLYSIS Dr
ELECTROCHEMISTRY – 3 PRACTICAL APPLICATION BATTERIES AND ELECTROLYSIS Dr. Sapna Gupta ELECTROCHEMICAL CELLS An electrochemical cell is a system consisting of electrodes that dip into an electrolyte and in which a chemical reaction either uses or generates an electric current. A voltaic or galvanic cell is an electrochemical cell in which a spontaneous reaction generates an electric current. An electrolytic cell is an electrochemical cell in which an electric current drives an otherwise nonspontaneous reaction. Dr. Sapna Gupta/Electrochemistry - Applications 2 GALVANIC CELLS • Galvanic cell - the experimental apparatus for generating electricity through the use of a spontaneous reaction • Electrodes • Anode (oxidation) • Cathode (reduction) • Half-cell - combination of container, electrode and solution • Salt bridge - conducting medium through which the cations and anions can move from one half-cell to the other. • Ion migration • Cations – migrate toward the cathode • Anions – migrate toward the anode • Cell potential (Ecell) – difference in electrical potential between the anode and cathode • Concentration dependent • Temperature dependent • Determined by nature of reactants Dr. Sapna Gupta/Electrochemistry - Applications 3 BATTERIES • A battery is a galvanic cell, or a series of cells connected that can be used to deliver a self-contained source of direct electric current. • Dry Cells and Alkaline Batteries • no fluid components • Zn container in contact with MnO2 and an electrolyte Dr. Sapna Gupta/Electrochemistry - Applications 4 ALKALINE CELL • Common watch batteries − − Anode: Zn(s) + 2OH (aq) Zn(OH)2(s) + 2e − − Cathode: 2MnO2(s) + H2O(l) + 2e Mn2O3(s) + 2OH (aq) This cell performs better under current drain and in cold weather. It isn’t truly “dry” but rather uses an aqueous paste. -

Lecture 03 Electrochemical Kinetics
EMA4303/5305 Electrochemical Engineering Lecture 03 Electrochemical Kinetics Dr. Junheng Xing, Prof. Zhe Cheng Mechanical & Materials Engineering Florida International University Electrochemical Kinetics Electrochemical kinetics “Electrochemical kinetics is a field of electrochemistry studying the rate of electrochemical processes. Due to electrochemical phenomena unfolding at the interface between an electrode and an electrolyte, there are accompanying phenomena to electrochemical reactions which contribute to the overall reaction rate.” (Wikipedia) “The main goal of the electrochemical kinetics is to find a relationship between the electrode overpotential and current density due to an applied potential.” (S. N. Lvov) Main contents of this lecture . Overpotential (η) . Charge transfer overpotential − Butler-Volmer equation − Tafel equation . Mass transfer overpotential EMA 5305 Electrochemical Engineering Zhe Cheng (2017) 3 Electrochemical Kinetics 2 Cell Potential in Different Modes Electrolytic cell (EC) Galvanic cell (GC) 푬푬푸 = 푰 (푹풆풙풕 + 푹풊풏풕) 푬푨풑풑풍풊풆풅 = 푬푬푸 + 푰푹풊풏풕 푬풆풙풕 = 푬푬푸 − 푰푹풊풏풕= IRext EMA 5305 Electrochemical Engineering Zhe Cheng (2017) 3 Electrochemical Kinetics 3 Cell Potential-Current Dependence . 푹풊풏풕 represents the total internal resistance of the cell, which is usually not zero, in most cases. Therefore, the difference between cell equilibrium potential and apparent potential is proportional to the current passing through the cell. Cell potential-current dependences EMA 5305 Electrochemical Engineering Zhe Cheng (2017) 3 Electrochemical Kinetics 4 Overpotential (η) Overpotential for a single electrode . The difference between the actual potential (measured) for an electrode, E, and the equilibrium potential for that electrode, 푬푬푸, is called the overpotential of that electrode, 휼 = 푬 −푬푬푸, which could be either positive or negative, depending on the direction of the half (cell) reaction. -

Solar-Driven, Highly Sustained Splitting of Seawater Into Hydrogen and Oxygen Fuels
Solar-driven, highly sustained splitting of seawater into hydrogen and oxygen fuels Yun Kuanga,b,c,1, Michael J. Kenneya,1, Yongtao Menga,d,1, Wei-Hsuan Hunga,e, Yijin Liuf, Jianan Erick Huanga, Rohit Prasannag, Pengsong Lib,c, Yaping Lib,c, Lei Wangh,i, Meng-Chang Lind, Michael D. McGeheeg,j, Xiaoming Sunb,c,d,2, and Hongjie Daia,2 aDepartment of Chemistry, Stanford University, Stanford, CA 94305; bState Key Laboratory of Chemical Resource Engineering, Beijing University of Chemical Technology, Beijing 100029, China; cBeijing Advanced Innovation Center for Soft Matter Science and Engineering, Beijing University of Chemical Technology, Beijing 100029, China; dCollege of Electrical Engineering and Automation, Shandong University of Science and Technology, Qingdao 266590, China; eDepartment of Materials Science and Engineering, Feng Chia University, Taichung 40724, Taiwan; fStanford Synchrotron Radiation Light Source, SLAC National Accelerator Laboratory, Menlo Park, CA 94025; gDepartment of Materials Science and Engineering, Stanford University, Stanford, CA 94305; hCenter for Electron Microscopy, Institute for New Energy Materials, Tianjin University of Technology, Tianjin 300384, China; iTianjin Key Laboratory of Advanced Functional Porous Materials, School of Materials, Tianjin University of Technology, Tianjin 300384, China; and jDepartment of Chemical Engineering, University of Colorado Boulder, Boulder, CO 80309 Contributed by Hongjie Dai, February 5, 2019 (sent for review January 14, 2019; reviewed by Xinliang Feng and Ali Javey) Electrolysis of water to generate hydrogen fuel is an attractive potential ∼490 mV higher than that of OER, which demands renewable energy storage technology. However, grid-scale fresh- highly active OER electrocatalysts capable of high-current (∼1 2 water electrolysis would put a heavy strain on vital water re- A/cm ) operations for high-rate H2/O2 production at over- sources. -

Overpotential Analysis of Alkaline and Acidic Alcohol Electrolysers And
1 2 Overpotential analysis of alkaline and acidic alcohol electrolysers 3 and optimized membrane-electrode assemblies 4 5 F. M. Sapountzia*, V. Di Palmab, G. Zafeiropoulosc, H. Penchevd, M.Α. Verheijenb, 6 M. Creatoreb, F. Ublekovd, V. Sinigerskyd, W.M. Arnold Bikc, H.O.A. Fredrikssona, 7 M.N. Tsampasc, J.W. Niemantsverdrieta 8 9 a SynCat@DIFFER, Syngaschem BV, P.O. Box 6336, 5600 HH, Eindhoven, The Netherlands, 10 www.syngaschem.com 11 b Department of Applied Physics, Eindhoven University of Technology, 5600MB, Eindhoven, The Netherlands 12 c DIFFER, Dutch Institute For Fundamental Energy Research, De Zaale 20, 5612 AJ, Eindhoven, The Netherlands 13 d Institute of Polymers, Bulgarian Academy of Sciences, Acad. Georgi Bonchev 103A Str., BG 1113, Sofia, 14 Bulgaria 15 16 *Corresponding author: Foteini Sapountzi, email: [email protected], postal address: Syngaschem BV, P.O. 17 Box 6336, 5600 HH, Eindhoven, The Netherlands 18 19 20 Abstract: Alcohol electrolysis using polymeric membrane electrolytes is a promising route for 21 storing excess renewable energy in hydrogen, alternative to the thermodynamically limited water 22 electrolysis. By properly choosing the ionic agent (i.e. H+ or OH-) and the catalyst support, and 23 by tuning the catalyst structure, we developed membrane-electrode-assemblies which are 24 suitable for cost-effective and efficient alcohol electrolysis. Novel porous electrodes were 25 prepared by Atomic Layer Deposition (ALD) of Pt on a TiO2-Ti web of microfibers and were 26 interfaced to polymeric membranes with either H+ or OH- conductivity. Our results suggest that 27 alcohol electrolysis is more efficient using OH- conducting membranes under appropriate 28 operation conditions (high pH in anolyte solution). -

Electrolytic Production of Aluminium:� Fundamental
r , ELECTROLYTIC PRODUCTION OF ALUMINIUM:� FUNDAMENTAL AND APPLIED STUDIES ON THE EFFECT OF ANODE COMPOSITION ON TEE ANODE-ELECTROLYTE INTERFACIAL CONDITIONS. by :,:.!::_OSBORNE. B.Sc. (Hons.), (Tas.) being a Thesis submitted. in fulfilment of the require ments for the Degree of Master of Science UNIVERSITY OF TASMANIA HOBART'. (May 1968) DECLARATION OF ORIGINALITY All of the experimental w0rk in this thesis, except where directly ascribed to others, has been performed solely by the auth0r. No part 0f the text bas previoµsly been submitted f@r any other degree or diplGma in any University, nor dees it contain, to the best knowledge of the undersigned, any material which has been published by others, except where due reference bas been SQ made. SUMMARY This thesis deals with a study of the wettability of amorphous carbon, of the type empl©yed as anode material in the production of aluminium, by cryolite based solutions. The wettability of carbon has been improved by the addition of inorganic additives 0f the type used in cryolite electrolytes, such as aluminium oxide, calcium flanride and carbonate, and lithium flunride and carbonate. Particular improvements in carbon wettability are noted when the system is contained in an atmosphere of carbon dioxide - carbon monoxide, corresponding to practical production conditions. Furtheremore, when anodes prepared c0ntaining these additives are used in a laboratory aluminium cell, notable reductions in cell voltage are achieved at practical an0de current densities. The improved adhesion of the two phases has also been demonstrated by measurement of the current density and voltage con ditions required to llinduce anode effects in this cell •. -

Understanding Anode Overpotential
UNDERSTANDING ANODE OVERPOTENTIAL Rebecca Jayne Thorne1, Camilla Sommerseth1, Ann Mari Svensson1, Espen Sandnes2, Lorentz Petter Lossius2, Hogne Linga2, and Arne Petter Ratvik1 1Dept. of Materials Science and Engineering, Norwegian University of Science and Technology, NO-7491 Trondheim, Norway 2Hydro Aluminium, Årdal, Norway Keywords: Anode, carbon, electrochemistry, characterisation, overpotential. Abstract have not linked these coke properties to overpotential directly [2, 9-11]. It is likely that coke and anode physical properties will In aluminium electrolysis cells the anodic process is associated have an effect on overpotential, as the reactivity of carbons, both with a substantial overpotential. Industrial carbon anodes are electrochemical reactions and oxidation reactions, is generally produced from a blend of coke materials, but the effect of coke known to be related to microstructural parameters [12, 13]. In type on anodic overpotential has not been well studied. In this addition, the formation and release of CO2 bubbles also relies on work, lab-scale anodes were fabricated from single cokes and the surface microstructure [14]. electrochemical methods were subsequently used to determine the overpotential trend of the anode materials. Attempts were then In a previous study that served as preliminary work on a new made to explain these trends in terms of both the physical and project, the total overpotential of a range of carbon anodes chemical characteristics of the baked anodes themselves and their varying only in coke type was measured [15]. These anodes were raw materials. Routine coke and anode characterisation methods labeled 1 to 4, and a distinct overpotential trend was found with were used to measure properties such as impurity concentrations anode 4 having a lower overpotential than anode 1 by -2 and reactivity (to air and CO2), while non-routine characterisation approximately 200 mV at 1 A cm . -
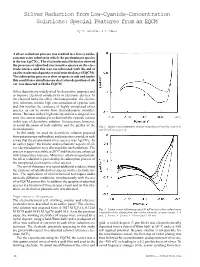
Silver Reduction from Low-Cyanide-Concentration Solutions: Special Features from an EQCM
Silver Reduction from Low-Cyanide-Concentration Solutions: Special Features from an EQCM By H. Sanchez & Y. Meas A silver reduction process was studied in a low-cyanide- concentration solution in which the predominant species - is the ion Ag(CN)2 . The electrochemical behavior showed the presence of adsorbed electroactive species on the elec- trode surface and this was corroborated with the aid of an electrochemical quartz crystal microbalance (EQCM). The adsorption process is slow at open circuit and under this condition a simultaneous electroless deposition of sil- ver was detected with the EQCM. Silver deposits are widely used for decorative purposes and to improve electrical conductivity in electronic devices. In the classical baths for silver electrodeposition, the electro- lytic solutions contain high concentrations of cyanide ions and this implies the existence of highly complexed silver species, as can be shown from thermodynamic consider- ations.1 Because of their high toxicity and for ecological rea- sons, the current tendency is to diminish the cyanide content in this type of electrolytic solution. It is necessary, however, to avoid alteration of bath stability and the quality of the Fig. 1—Single scan voltammetry of silver reduction at 10 mV/sec (curve 1) electrodeposits. and 20 mV/sec (curve 2). In this study, we used an electrolytic solution prepared from potassium pyrophosphate and potassium cyanide in such - a way that the predominant silver species was Ag(CN)2 . In an earlier paper,1 the kinetic and mechanistic aspects of sil- ver electroreduction were discussed for such solutions. The process is quasi-reversible at 20 °C and kinetics are enhanced with temperature increase. -

Electrolysis of Salt Water
ELECTROLYSIS OF SALT WATER Unit: Salinity Patterns & the Water Cycle l Grade Level: High school l Time Required: Two 45 min. periods l Content Standard: NSES Physical Science, properties and changes of properties in matter; atoms have measurable properties such as electrical charge. l Ocean Literacy Principle 1e: Most of of Earth's water (97%) is in the ocean. Seawater has unique properties: it is saline, its freezing point is slightly lower than fresh water, its density is slightly higher, its electrical conductivity is much higher, and it is slightly basic. Big Idea: Water is comprised of two elements – hydrogen (H) and oxygen (O). Distilled water is pure and free of salts; thus it is a very poor conductor of electricity. By adding ordinary table salt (NaCl) to distilled water, it becomes an electrolyte solution, able to conduct electricity. Key Concepts o Ionic compounds such as salt water, conduct electricity when they dissolve in water. o Ionic compounds consist of two or more ions that are held together by electrical attraction. One of the ions has a positive charge (called a "cation") and the other has a negative charge ("anion"). o Molecular compounds, such as water, are made of individual molecules that are bound together by shared electrons (i.e., covalent bonds). o Essential Questions o What happens to salt when it is dissolved in water? o What are electrolytes? o How can we determine the volume of dissolved ions in a water sample? o How are atoms held together in an element? Knowledge and Skills o Conduct an experiment to see that water can be split into its constituent ions through the process of electrolysis.