Prostacyclin Synthesis by COX-2 Endothelial Cells
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Diclofenac Enhances Docosahexaenoic Acid-Induced Apoptosis in Vitro in Lung Cancer Cells
cancers Article Diclofenac Enhances Docosahexaenoic Acid-Induced Apoptosis in Vitro in Lung Cancer Cells Rosemary A. Poku 1, Kylee J. Jones 2, Megan Van Baren 2 , Jamie K. Alan 3 and Felix Amissah 2,* 1 Department of Foundational Sciences, College of Medicine, Central Michigan University, Warriner Hall, 319, Mt Pleasant, MI 48859, USA; [email protected] 2 Department of Pharmaceutical Sciences, Ferris State University, College of Pharmacy, 220 Ferris Dr, Big Rapids, MI 49307, USA; [email protected] (K.J.J.); [email protected] (M.V.B.) 3 Department of Pharmacology and Toxicology, Michigan State University, East Lansing, MI 48824, USA; [email protected] * Correspondence: [email protected]; Tel.: +1-231-591-3790 Received: 9 September 2020; Accepted: 17 September 2020; Published: 20 September 2020 Simple Summary: Polyunsaturated fatty acids (PUFAs) and non-steroidal anti-inflammatory drugs (NSAIDs) have limited anticancer capacities when used alone. We examined whether combining NSAIDs with docosahexaenoic (DHA) would increase their anticancer activity on lung cancer cell lines. Our results indicate that combining DHA and NSAIDs increased their anticancer activities by altering the expression of critical proteins in the RAS/MEK/ERK and PI3K/Akt pathways. The data suggest that DHA combined with low dose diclofenac provides more significant anticancer potential, which can be further developed for chemoprevention and adjunct therapy in lung cancer. Abstract: Polyunsaturated fatty acids (PUFAs) and non-steroidal anti-inflammatory drugs (NSAIDs) show anticancer activities through diverse molecular mechanisms. However, the anticancer capacities of either PUFAs or NSAIDs alone is limited. We examined whether combining NSAIDs with docosahexaenoic (DHA), commonly derived from fish oils, would possibly synergize their anticancer activity. -

Prostaglandin E2 Deficiency Uncovers a Dominant Role for Thromboxane A2
Prostaglandin E2 deficiency uncovers a dominant role for thromboxane A2 in house dust mite-induced allergic pulmonary inflammation Tao Liua,b, Tanya M. Laidlawa,b,c, Chunli Fenga,b, Wei Xinga,b, Shiliang Shena,b, Ginger L. Milned, and Joshua A. Boycea,b,c,1 Departments of aMedicine and cPediatrics, Harvard Medical School, Boston, MA 02115; bDivision of Rheumatology, Immunology, and Allergy, Jeff and Penny Vinik Center for Allergic Disease Research, Brigham and Women’s Hospital, Boston, MA 02115; and dDepartment of Pharmacology, Vanderbilt University, Nashville, TN 37232 Edited* by K. Frank Austen, Brigham and Women’s Hospital, Boston, MA, and approved June 20, 2012 (received for review May 10, 2012) fl – Prostaglandin E2 (PGE2) is an abundant lipid in ammatory media- PGE2 production (16 19) and reduced expression of the EP2 re- tor with potent but incompletely understood anti-inflammatory ceptor (20, 21), as well as marked tissue eosinophilia and bron- actions in the lung. Deficient PGE2 generation in the lung predis- choconstrictive responses to the administration of nonselective poses to airway hyperresponsiveness and aspirin intolerance in COX inhibitors. These findings suggest potential therapeutic fi −/− asthmatic individuals. PGE2-de cient ptges mice develop exag- applications of PGE2 in asthma and AERD if the mechanisms gerated pulmonary eosinophilia and pulmonary arteriolar smooth- responsible for the homeostatic functions of PGE2 in asthma can fi muscle hyperplasia compared with PGE2-suf cient controls when be fully defined. challenged intranasally with a house dust mite extract. We now An extract (Df) of the house dust mite Dermatophagoides demonstrate that both pulmonary eosinophilia and vascular farinae contains clinically relevant protease allergens, as well as fi remodeling in the setting of PGE2 de ciency depend on thrombox- adjuvants (glycans, endotoxin) that elicit sensitization through ane A2 and signaling through the T prostanoid (TP) receptor. -

Coexpression of Microsomal Prostaglandin E Synthase With
Coexpression of Microsomal Prostaglandin E Synthase with Cyclooxygenase-2 in Human Rheumatoid Synovial Cells FUMIAKI KOJIMA, HIROAKI NARABA, YASUHARU SASAKI, RENZO OKAMOTO, TOMIHISA KOSHINO, and SHINICHI KAWAI ABSTRACT. Objective. Recently, microsomal prostaglandin (PG) E synthase (mPGES) was cloned as a terminal enzyme catalyzing PGH2 to PGE2. We investigated mPGES as well as cyclooxygenase (COX)-2, catalyzing arachidonic acid to PGH2, in synovial cells from patients with rheumatoid arthritis (RA). The effect of dexamethasone on mPGES expression was also studied. Methods. Synovial cells were treated with interleukin 1ß (IL-1ß) and dexamethasone under various conditions, and expression of mPGES mRNA and protein was analyzed by Northern blot and Western blot, respectively. Conversions of arachidonic acid or PGH2 to PGE2 were measured by ELISA. Subcellular localization of mPGES and COX-2 was determined by immunofluorescent microscopic analysis. Results. mPGES mRNA and protein expression were significantly upregulated by IL-1ß in synovial cells. COX-2 mRNA and protein were also upregulated by IL-1ß, but with a different time course from that of mPGES. Conversion of PGH2 to PGE2 was increased by IL-1ß and was correlated with mPGES expression. Increased conversion of arachidonic acid to PGE2 was maintained when mPGES and COX-2 were coexpressed. Subcellular localization of mPGES and COX-2 overlapped in the perinuclear region in IL-1ß stimulated synovial cells. Dexamethasone inhibited mRNA and protein expression for mPGES and increased conversion of arachidonic acid to PGE2, but inhibition of mPGES was weaker compared with that of COX-2 in IL-1ß stimulated cells. Conclusion. The results suggest that abundant PGE2 production at inflammation sites such as rheumatoid synovia is caused by the coordinated upregulation of mPGES and COX-2. -

Expression and Regulation of Microsomal Prostaglandin E Synthase
Expression and Regulation of Microsomal Prostaglandin E Synthase-1 in Human Osteoarthritic Cartilage and Chondrocytes XINFANG LI, HASSAN AFIF, SARANETTE CHENG, JOHANNE MARTEL-PELLETIER, JEAN-PIERRE PELLETIER, PIERRE RANGER, and HASSAN FAHMI ABSTRACT. Objective. Elevated production of prostaglandin E2 (PGE2) plays an important role in the pathogen- esis of arthritis. Recently, an inducible microsomal prostaglandin E synthase-1 (mPGES-1) was identified. This enzyme is functionally coupled with cyclooxygenase-2 (COX-2) and converts the COX product PGH2 to PGE2. We analyzed expression of mPGES-1 in human normal and osteoarthritic (OA) cartilage and determined the effect of different inflammatory agonists on the expression of mPGES-1 in OA chondrocytes. Methods. Expression of mPGES-1 mRNA and protein in cartilage was determined by quantitative real-time reverse transcriptase-polymerase chain reaction and immunohistochemistry, respectively. OA chondrocytes were treated with different inflammatory agents, and mPGES-1 protein expression was evaluated by Western blot. Activation of the mPGES-1 promoter was assessed in transient trans- fection experiments. Results. Levels of mPGES-1 mRNA and protein were markedly elevated in OA versus normal car- tilage. Treatment of chondrocytes with interleukin 1ß (IL-1ß) induced expression of mPGES-1 pro- tein in a dose- and time-dependent manner. This appears to occur at the transcriptional level, as IL- 1ß induced expression of mPGES-1 mRNA and the activity of this gene promoter. Tumor necrosis factor-α (TNF-α) and IL-17 also upregulated expression of mPGES-1 protein and displayed a syn- ergistic effect with IL-1ß. Peroxisome proliferator-activated receptor-γ ligands, 15-deoxy-∆12,14- prostaglandin J2 and troglitazone, inhibited IL-1ß-induced mPGES-1 protein expression, an effect that was reversed by exogenous PGE2. -

Endogenous Biosynthesis of Prostacyclin and Thromboxane and Platelet Function During Chronic Administration of Aspirin in Man
Endogenous biosynthesis of prostacyclin and thromboxane and platelet function during chronic administration of aspirin in man. G A FitzGerald, … , J A Lawson, A R Brash J Clin Invest. 1983;71(3):676-688. https://doi.org/10.1172/JCI110814. Research Article To assess the pharmacologic effects of aspirin on endogenous prostacyclin and thromboxane biosynthesis, 2,3-dinor-6- keto PGF1 alpha (PGI-M) and 2,3-dinor-thromboxane B2 (Tx-M) were measured in urine by mass spectrometry during continuing administration of aspirin. To define the relationship of aspirin intake to endogenous prostacyclin biosynthesis, sequential urines were initially collected in individuals prior to, during, and subsequent to administration of aspirin. Despite inter- and intra-individual variations, PGI-M excretion was significantly reduced by aspirin. However, full mass spectral identification confirmed continuing prostacyclin biosynthesis during aspirin therapy. Recovery of prostacyclin biosynthesis was incomplete 5 d after drug administration was discontinued. To relate aspirin intake to indices of thromboxane biosynthesis and platelet function, volunteers received 20 mg aspirin daily followed by 2,600 mg aspirin daily, each dose for 7 d in sequential weeks. Increasing aspirin dosage inhibited Tx-M excretion from 70 to 98% of pretreatment control values; platelet TxB2 formation from 4.9 to 0.5% and further inhibited platelet function. An extended study was performed to relate aspirin intake to both thromboxane and prostacyclin generation over a wide range of doses. Aspirin, in the range of 20 to 325 mg/d, resulted in a dose-dependent decline in both Tx-M and PGI-M excretion. At doses of 325-2,600 mg/d Tx-M excretion ranged from 5 to 3% of control values while PGI-M remained at 37-23% of control. -

Challenging the FDA Black Box Warning for High Aspirin Dose with Ticagrelor in Patients with Diabetes James J
PERSPECTIVES IN DIABETES Challenging the FDA Black Box Warning for High Aspirin Dose With Ticagrelor in Patients With Diabetes James J. DiNicolantonio and Victor L. Serebruany – Ticagrelor, a novel reversible antiplatelet agent, has a Food and ASA 300 325 mg (53.6%) in the U.S. compared with the Drug Administration (FDA) black box warning to avoid mainte- rest of the world (1.7%) was the only factor to explain nance doses of aspirin (ASA) .100 mg/daily. This restriction is (out of 37 variables explored) the regional interaction based on the hypothesis that ASA doses .100 mg somehow de- that ticagrelor was less effective and potentially more creased ticagrelor’s benefit in the Platelet Inhibition and Patient harmful than clopidogrel (2). However, this interaction Outcomes (PLATO) U.S. cohort. However, these data are highly was not significant, is highly postrandomized, comes postrandomized, come from a very small subgroup in PLATO from a very small subgroup of PLATO, and makes no (57% of patients in the U.S. site), and make no biological sense. biological sense (3). Moreover, the ticagrelor-ASA interaction was not significant by any multivariate Cox regression analyses. The Complete Re- sponse Review for ticagrelor indicates that for U.S. PLATO patients, an ASA dose .300 mg was not a significant interaction TICAGRELOR-ASA HYPOTHESIS for vascular outcomes. In the ticagrelor-ASA .300 mg cohort, all- The claimed ticagrelor-ASA interaction states that ticagrelor cause and vascular mortality were not significantly increased fi – P plus low-dose ASA is bene cial, whereas increasing doses (hazard ratio [HR] 1.27 [95% CI 0.84 1.93], = 0.262 and 1.39 of ASA in combination with ticagrelor produces adverse [0.87–2.2], P = 0.170), respectively. -

GTH 2021 State of the Art—Cardiac Surgery: the Perioperative Management of Heparin-Induced Thrombocytopenia in Cardiac Surgery
Review Article 59 GTH 2021 State of the Art—Cardiac Surgery: The Perioperative Management of Heparin-Induced Thrombocytopenia in Cardiac Surgery Laura Ranta1 Emmanuelle Scala1 1 Department of Anesthesiology, Cardiothoracic and Vascular Address for correspondence Emmanuelle Scala, MD, Centre Anesthesia, Lausanne University Hospital (CHUV), Lausanne, Hospitalier Universitaire Vaudois, Rue du Bugnon 46, BH 05/300, 1011 Switzerland Lausanne, Suisse, Switzerland (e-mail: [email protected]). Hämostaseologie 2021;41:59–62. Abstract Heparin-induced thrombocytopenia (HIT) is a severe, immune-mediated, adverse drug Keywords reaction that paradoxically induces a prothrombotic state. Particularly in the setting of ► Heparin-induced cardiac surgery, where full anticoagulation is required during cardiopulmonary bypass, thrombocytopenia the management of HIT can be highly challenging, and requires a multidisciplinary ► cardiac surgery approach. In this short review, the different perioperative strategies to run cardiopul- ► state of the art monary bypass will be summarized. Introduction genicity of the antibodies and is diagnostic for HIT. The administration of heparin to a patient with circulating Heparin-induced thrombocytopenia (HIT) is a severe, im- pathogenic HITabs puts the patient at immediate risk of mune-mediated, adverse drug reaction that paradoxically severe thrombotic complications. induces a prothrombotic state.1,2 Particularly in the setting The time course of HIT can be divided into four distinct of cardiac surgery, where full anticoagulation is required phases.6 Acute HIT is characterized by thrombocytopenia during cardiopulmonary bypass (CPB), the management of and/or thrombosis, the presence of HITabs, and confirma- HIT can be highly challenging, and requires a multidisciplin- tion of their platelet activating capacity by a functional ary approach. -

Inverse Agonism of SQ 29,548 and Ramatroban on Thromboxane A2 Receptor
Inverse Agonism of SQ 29,548 and Ramatroban on Thromboxane A2 Receptor Raja Chakraborty1,3, Rajinder P. Bhullar1, Shyamala Dakshinamurti2,3, John Hwa4, Prashen Chelikani1,2,3* 1 Department of Oral Biology, University of Manitoba, Winnipeg, Manitoba, Canada, 2 Departments of Pediatrics, Physiology, University of Manitoba, Winnipeg, Manitoba, Canada, 3 Biology of Breathing Group- Manitoba Institute of Child Health, Winnipeg, Manitoba, Canada, 4 Department of Internal Medicine (Cardiology), Cardiovascular Research Center, Yale University School of Medicine, New Haven, Connecticut, United States of America Abstract G protein-coupled receptors (GPCRs) show some level of basal activity even in the absence of an agonist, a phenomenon referred to as constitutive activity. Such constitutive activity in GPCRs is known to have important pathophysiological roles in human disease. The thromboxane A2 receptor (TP) is a GPCR that promotes thrombosis in response to binding of the prostanoid, thromboxane A2. TP dysfunction is widely implicated in pathophysiological conditions such as bleeding disorders, hypertension and cardiovascular disease. Recently, we reported the characterization of a few constitutively active mutants (CAMs) in TP, including a genetic variant A160T. Using these CAMs as reporters, we now test the inverse agonist properties of known antagonists of TP, SQ 29,548, Ramatroban, L-670596 and Diclofenac, in HEK293T cells. Interestingly, SQ 29,548 reduced the basal activity of both, WT-TP and the CAMs while Ramatroban was able to reduce the basal activity of only the CAMs. Diclofenac and L-670596 showed no statistically significant reduction in basal activity of WT-TP or CAMs. To investigate the role of these compounds on human platelet function, we tested their effects on human megakaryocyte based system for platelet activation. -

Downloaded from Bioscientifica.Com at 09/25/2021 12:14:58PM Via Free Access
ID: 19-0149 8 6 Q Pang et al. PHO patients with soft tissue 8:6 736–744 giant tumor caused by HPGD mutation RESEARCH The first case of primary hypertrophic osteoarthropathy with soft tissue giant tumors caused by HPGD loss-of-function mutation Qianqian Pang1,2, Yuping Xu1,3, Xuan Qi1, Yan Jiang1, Ou Wang1, Mei Li1, Xiaoping Xing1, Ling Qin2 and Weibo Xia1 1Department of Endocrinology, Key Laboratory of Endocrinology, Ministry of Health, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences, Beijing, China 2Musculoskeletal Research Laboratory and Bone Quality and Health Assessment Centre, Department of Orthopedics & Traumatology, The Chinese University of Hong Kong, Hong Kong SAR, Hong Kong 3Department of Endocrinology, The First Affiliated Hospital of Shanxi Medicalniversity, U Taiyuan, Shanxi, China Correspondence should be addressed to L Qin or W Xia: [email protected] or [email protected] Abstract Background: Primary hypertrophic osteoarthropathy (PHO) is a rare genetic multi-organic Key Words disease characterized by digital clubbing, periostosis and pachydermia. Two genes, f primary hypertrophic HPGD and SLCO2A1, which encodes 15-hydroxyprostaglandin dehydrogenase (15-PGDH) osteoarthropathy and prostaglandin transporter (PGT), respectively, have been reported to be related to f PHO PHO. Deficiency of aforementioned two genes leads to failure of prostaglandin E2 (PGE2) f HPGD mutation degradation and thereby elevated levels of PGE2. PGE2 plays an important role in f soft tumor tumorigenesis. Studies revealed a tumor suppressor activity of 15-PGDH in tumors, such f COX2 selective inhibitor treatment as lung, bladder and breast cancers. However, to date, no HPGD-mutated PHO patients presenting concomitant tumor has been documented. -

Cyclooxygenase-Independent Inhibition by Aspirin
Proc. Natl. Acad. Sci. USA Vol. 93, pp. 11091-11096, October 1996 Medical Sciences Leukocyte lipid body formation and eicosanoid generation: Cyclooxygenase-independent inhibition by aspirin (nonsteroidal antiinflammatory agents/cyclooxygenase knockout/leukotrienes/prostaglandin endoperoxide H synthase/lipoxygenase) PATRICIA T. BOZZA*, JENNIFER L. PAYNE*, ScoTr G. MORHAMt, ROBERT LANGENBACHt, OLIVER SMITHIESt, AND PETER F. WELLER*§ *Harvard Thorndike Laboratory and Charles A. Dana Research Institute, Department of Medicine, Beth Israel Hospital, Harvard Medical School, Boston, MA 02215-5491; tDepartment of Pathology, University of North Carolina, Chapel Hill, NC 27599-7525; and *Laboratory of Experimental Carcinogenesis and Mutagenesis, National Institute of Environmental Health Sciences, Research Triangle Park, NC 27709 Communicated by Seymour J. Klenbanoff, University of Washington School of Medicine, Seattle, WA, July 31, 1996 (received for review April 8, 1996) ABSTRACT Lipid bodies, cytoplasmic inclusions that de- inducible structures. Lipid bodies are prominent in leukocytes velop in cells associated with inflammation, are inducible at sites of natural and experimental inflammation, including structures that might participate in generating inflammatory leukocytes from joints of patients with inflammatory arthritis eicosanoids. Cis-unsaturated fatty acids (arachidonic and (5-7), the airways of patients with acute respiratory distress oleic acids) rapidly induced lipid body formation in leuko- syndrome (8), and casein- or lipopolysaccharide-elicited cytes, and this lipid body induction was inhibited by aspirin guinea pig peritoneal exudates (9). The formation of lipid and nonsteroidal antiinflammatory drugs (NSAIDs). Several bodies is a regulated process that involves activation of intra- findings indicated that the inhibitory effect of aspirin and cellular signaling pathways, including protein kinase C and NSAIDs on lipid body formation was independent of cycloox- phospholipase C (10, 11). -
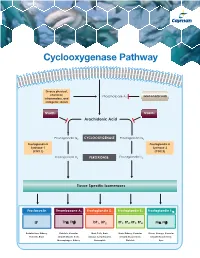
Cyclooxygenase Pathway
Cyclooxygenase Pathway Diverse physical, chemical, Phospholipase A Glucocorticoids inflammatory, and 2 mitogenic stimuli NSAIDs NSAIDs Arachidonic Acid Prostaglandin G2 CYCLOOXYGENASE Prostaglandin G2 Prostaglandin H Prostaglandin H Synthase-1 Synthase-2 (COX 1) (COX 2) Prostaglandin H2 PEROXIDASE Prostaglandin H2 Tissue Specific Isomerases Prostacyclin Thromboxane A2 Prostaglandin D2 Prostaglandin E2 Prostaglandin F2α IP TPα, TPβ DP1, DP2 EP1, EP2, EP3, EP4 FPα, FPβ Endothelium, Kidney, Platelets, Vascular Mast Cells, Brain, Brain, Kidney, Vascular Uterus, Airways, Vascular Platelets, Brain Smooth Muscle Cells, Airways, Lymphocytes, Smooth Muscle Cells, Smooth Muscle Cells, Macrophages, Kidney Eosinophils Platelets Eyes Prostacyclin Item No. Product Features Prostacyclin (Prostaglandin I2; PGI2) is formed from arachidonic acid primarily in the vascular endothelium and renal cortex by sequential 515211 6-keto • Sample Types: Culture Medium | Plasma Prostaglandin • Measure 6-keto PGF levels down to 6 pg/ml activities of COX and prostacyclin synthase. PGI2 is non-enzymatically 1α F ELISA Kit • Incubation : 18 hours | Development: 90-120 minutes | hydrated to 6-keto PGF1α (t½ = 2-3 minutes), and then quickly converted 1α Read: Colorimetric at 405-420 nm to the major metabolite, 2,3-dinor-6-keto PGF1α (t½= 30 minutes). Prostacyclin was once thought to be a circulating hormone that regulated • Assay 24 samples in triplicate or 36 samples in duplicate platelet-vasculature interactions, but the rate of secretion into circulation • NOTE: A portion of urinary 6-keto PGF1α is of renal origin coupled with the short half-life indicate that prostacyclin functions • NOTE : It has been found that normal plasma levels of 6-keto PGF may be low locally. -

Vasoactive Responses of U46619, PGF2 , Latanoprost, and Travoprost
Vasoactive Responses of U46619, PGF2␣, Latanoprost, and Travoprost in Isolated Porcine Ciliary Arteries Ineta Vysniauskiene, Reto Allemann, Josef Flammer, and Ivan O. Haefliger PURPOSE. To compare the vasoactive properties of the prosta- these responses can be modulated by SQ 29548 (TP-receptor noids U46619 (thromboxane A2 analogue), prostaglandin F2␣ antagonist) or AL-8810 (FP-receptor antagonist). (PGF2␣), latanoprost free acid, and travoprost free acid in isolated porcine ciliary arteries. METHODS. In a myograph system (isometric force measure- MATERIAL AND METHODS ment), quiescent vessels were exposed (cumulatively) to U46619, PGF , latanoprost, or travoprost (0.1 nM–0.1 mM). 2␣ Vessels Preparation Experiments were also conducted in the presence of SQ 29548 (TP-receptor antagonist; 3–10 M) or AL-8810 (FP-receptor Porcine eyes were obtained from an abattoir immediately after death. antagonist; 3–30 M). Contractions were expressed as the In cold modified Krebs-Ringer solution (NaCl 118 mM, KCl 4.7 mM, percentage of 100 mM potassium chloride-induced contrac- CaCl2 2.5 mM, K2PO4 1.2 mM, MgSO4 1.2 mM, NaHCO3 25 mM, tions. glucose 11.1 mM, and EDTA 0.026 mM), ciliary arteries were dissected 5 RESULTS. In quiescent vessels, contractions (concentration–re- and cut into 2-mm segments. In an organ chamber, two 45- m Ϯ tungsten wires were passed through the vessel’s lumen and attached to sponse curves) induced by (0.1 mM) PGF2␣ (87.9% 3.5%), U46619 (66.7% Ϯ 4.1%), and latanoprost (62.9% Ϯ 3.6%) were a force transducer for isometric force measurements (Myo-Interface; JP more pronounced (P Յ 0.001) than those induced by tra- Trading, Aarhus, Denmark).