Evolution of Oxygenic Photosynthesis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Prokaryotes (Domains Bacteria & Archaea)
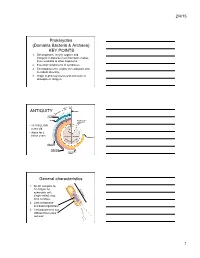
2/4/15 Prokaryotes (Domains Bacteria & Archaea) KEY POINTS 1. Decomposers: recycle organic and inorganic molecules in environment; makes them available to other organisms. 2. Essential components of symbioses. 3. Encompasses the origins of metabolism and metabolic diversity. 4. Origin of photosynthesis and formation of atmospheric Oxygen Ceno- Meso- zoic zoic ANTIQUITY Humans Paleozoic Colonization of land Animals Origin of solar system and Earth • >3.5 BILLION years old. • Alone for 2 1 4 billion years Proterozoic Archaean Prokaryotes Billions of 2 years ago3 Multicellular eukaryotes Single-celled eukaryotes Atmospheric oxygen General characteristics 1. Small: compare to 10-100µm for 0.5-5µm eukaryotic cell; single-celled; may form colonies. 2. Lack membrane- enclosed organelles. 3. Cell wall present, but different from plant cell wall. 1 2/4/15 General characteristics 4. Occur everywhere, most numerous organisms. – More individuals in a handful of soil then there are people that have ever lived. – By far more individuals in our gut than eukaryotic cells that are actually us. General characteristics 5. Metabolic diversity established nutritional modes of eukaryotes. General characteristics 6. Important decomposers and recyclers 2 2/4/15 General characteristics 6. Important decomposers and recyclers • Form the basis of global nutrient cycles. General characteristics 7. Symbionts!!!!!!! • Parasites • Pathogenic organisms. • About 1/2 of all human diseases are caused by Bacteria General characteristics 7. Symbionts!!!!!!! • Parasites • Pathogenic organisms. • Extremely important in agriculture as well. Pierce’s disease is caused by Xylella fastidiosa, a Gamma Proteobacteria. It causes over $56 million in damage annually in California. That’s with $34 million spent to control it! = $90 million in California alone. -

Characterization of the Aerobic Anoxygenic Phototrophic Bacterium Sphingomonas Sp
microorganisms Article Characterization of the Aerobic Anoxygenic Phototrophic Bacterium Sphingomonas sp. AAP5 Karel Kopejtka 1 , Yonghui Zeng 1,2, David Kaftan 1,3 , Vadim Selyanin 1, Zdenko Gardian 3,4 , Jürgen Tomasch 5,† , Ruben Sommaruga 6 and Michal Koblížek 1,* 1 Centre Algatech, Institute of Microbiology, Czech Academy of Sciences, 379 81 Tˇreboˇn,Czech Republic; [email protected] (K.K.); [email protected] (Y.Z.); [email protected] (D.K.); [email protected] (V.S.) 2 Department of Plant and Environmental Sciences, University of Copenhagen, Thorvaldsensvej 40, 1871 Frederiksberg C, Denmark 3 Faculty of Science, University of South Bohemia, 370 05 Ceskˇ é Budˇejovice,Czech Republic; [email protected] 4 Institute of Parasitology, Biology Centre, Czech Academy of Sciences, 370 05 Ceskˇ é Budˇejovice,Czech Republic 5 Research Group Microbial Communication, Technical University of Braunschweig, 38106 Braunschweig, Germany; [email protected] 6 Laboratory of Aquatic Photobiology and Plankton Ecology, Department of Ecology, University of Innsbruck, 6020 Innsbruck, Austria; [email protected] * Correspondence: [email protected] † Present Address: Department of Molecular Bacteriology, Helmholtz-Centre for Infection Research, 38106 Braunschweig, Germany. Abstract: An aerobic, yellow-pigmented, bacteriochlorophyll a-producing strain, designated AAP5 Citation: Kopejtka, K.; Zeng, Y.; (=DSM 111157=CCUG 74776), was isolated from the alpine lake Gossenköllesee located in the Ty- Kaftan, D.; Selyanin, V.; Gardian, Z.; rolean Alps, Austria. Here, we report its description and polyphasic characterization. Phylogenetic Tomasch, J.; Sommaruga, R.; Koblížek, analysis of the 16S rRNA gene showed that strain AAP5 belongs to the bacterial genus Sphingomonas M. Characterization of the Aerobic and has the highest pairwise 16S rRNA gene sequence similarity with Sphingomonas glacialis (98.3%), Anoxygenic Phototrophic Bacterium Sphingomonas psychrolutea (96.8%), and Sphingomonas melonis (96.5%). -

A Study on the Phototrophic Microbial Mat Communities of Sulphur Mountain Thermal Springs and Their Association with the Endangered, Endemic Snail Physella Johnsoni
A Study on the Phototrophic Microbial Mat Communities of Sulphur Mountain Thermal Springs and their Association with the Endangered, Endemic Snail Physella johnsoni By Michael Bilyj A thesis submitted to the Faculty of Graduate Studies in partial fulfillment of the requirements for the degree of Master of Science Department of Microbiology Faculty of Science University of Manitoba Winnipeg, Manitoba October 2011 © Copyright 2011, Michael A. Bilyj 1 Abstract The seasonal population fluctuation of anoxygenic phototrophs and the diversity of cyanobacteria at the Sulphur Mountain thermal springs of Banff, Canada were investigated and compared to the drastic population changes of the endangered snail Physella johnsoni. A new species and two strains of Rhodomicrobium were taxonomically characterized in addition to new species of Rhodobacter and Erythromicrobium. Major mat-forming organisms included Thiothrix-like species, oxygenic phototrophs of genera Spirulina, Oscillatoria, and Phormidium and purple nonsulfur bacteria Rhodobacter, Rhodopseudomonas and Rhodomicrobium. Aerobic anoxygenic phototrophs comprised upwards of 9.6 x 104 CFU/cm2 of mat or 18.9% of total aerobic heterotrophic bacterial isolates at certain sites, while maximal purple nonsulfur and purple sulfur bacteria were quantified at 3.2 x 105 and 2.0 x 106 CFU/cm2 of mat, respectively. Photosynthetic activity measurements revealed incredibly productive carbon fixation rates averaging 40.5 mg C/cm2/24 h. A temporal mismatch was observed for mat area and prokaryote-based organics to P. johnsoni population flux in a ―tracking inertia‖ manner. 2 Acknowledgements It is difficult to express sufficient gratitude to my supervisor Dr. Vladimir Yurkov for his unfaltering patience, generosity and motivation throughout this entire degree. -

Anoxygenic Photosynthesis in Photolithotrophic Sulfur Bacteria and Their Role in Detoxication of Hydrogen Sulfide
antioxidants Review Anoxygenic Photosynthesis in Photolithotrophic Sulfur Bacteria and Their Role in Detoxication of Hydrogen Sulfide Ivan Kushkevych 1,* , Veronika Bosáková 1,2 , Monika Vítˇezová 1 and Simon K.-M. R. Rittmann 3,* 1 Department of Experimental Biology, Faculty of Science, Masaryk University, 62500 Brno, Czech Republic; [email protected] (V.B.); [email protected] (M.V.) 2 Department of Biology, Faculty of Medicine, Masaryk University, 62500 Brno, Czech Republic 3 Archaea Physiology & Biotechnology Group, Department of Functional and Evolutionary Ecology, Universität Wien, 1090 Vienna, Austria * Correspondence: [email protected] (I.K.); [email protected] (S.K.-M.R.R.); Tel.: +420-549-495-315 (I.K.); +431-427-776-513 (S.K.-M.R.R.) Abstract: Hydrogen sulfide is a toxic compound that can affect various groups of water microorgan- isms. Photolithotrophic sulfur bacteria including Chromatiaceae and Chlorobiaceae are able to convert inorganic substrate (hydrogen sulfide and carbon dioxide) into organic matter deriving energy from photosynthesis. This process takes place in the absence of molecular oxygen and is referred to as anoxygenic photosynthesis, in which exogenous electron donors are needed. These donors may be reduced sulfur compounds such as hydrogen sulfide. This paper deals with the description of this metabolic process, representatives of the above-mentioned families, and discusses the possibility using anoxygenic phototrophic microorganisms for the detoxification of toxic hydrogen sulfide. Moreover, their general characteristics, morphology, metabolism, and taxonomy are described as Citation: Kushkevych, I.; Bosáková, well as the conditions for isolation and cultivation of these microorganisms will be presented. V.; Vítˇezová,M.; Rittmann, S.K.-M.R. -

Oxygen Evolution
1 BISC 367 Plant Physiology Laboratory Simon Fraser University Chloroplast photosynthesis Effects of varying light intensities and wavelengths on the activity of the oxygen evolving complex Photosynthesis can be divided into two major pathways, the “light reactions and the carbon reduction reactions”. The light reactions consisting of the light regulated splitting of water and subsequent transport of electrons takes place in the grana of the chloroplast. The rate of this reaction can be monitored by several methods. For example, the rate of NADPH production (the end product of the electron transport chain) can be monitored with a dye such as Neotetrasolium chloride. Another frequently used method to detect chloroplast activity is to monitor the rate of oxygen evolution. Although the final electron acceptor in vivo is NADP, in isolated chloroplasts, the activity of this compound is quite low. Therefore artificial electron acceptors such as potassium ferricyanide or methyl violagen are used to sustain the transport of electrons, received from the splitting of water at photosystem II, across the electron transport chain. In this exercise we will be using isolated spinach chloroplasts to study the effects of quantity (different light intensities) and quality (different wave lengths) of light on the rate of oxygen evolution. Also, the effectiveness of two photosystem inhibitors on O2 production will be compared. The mode of action of these inhibitors is different. DCMU (3-3,4 dichlorophenyl-1.1 dimethyl urea) is an herbicide that accepts electrons from one of the intermediates in the electron transport chain (ETC). FMN (flavin mononucleotide) on the other hand inhibits oxygen production by accepting electrons and transferring them to O2 (Mehler reaction). -

The Position of Cyanobacteria in the World of Phototrophs
Carlsberg Res. Commun. Vol. 42, p. 77-98, 1977 THE POSITION OF CYANOBACTERIA IN THE WORLD OF PHOTOTROPHS. by ROGER Y STANIER D~partementde Biochimieet G6n6tiqueMicrobienne 28, rue du Dr Roux, 75015 Paris, France EMIL CHR. nANSENlecture delivered at the Carlsberg Laboratory Centennial and the Inauguration of the Carlsberg Research Center on September 28th, 1976 Key words: prokaryotic photosynthetic apparatus; obligate photoautotrophy; oxidative and reductive pentose phosphate cycles; nitrogen fixation; oxygenic photosynthesis; facultative anoxygenic photosynthesis; cyanobacterial ecology; cyanobacterial developmental patterns Recent advances in our knowledge of the biological properties of the cyanobacteria (former ,blue-green algae,) are reviewed. The subjects discussed include: cyanobacterial cell structure and development; comparative as- pects of photosyntetic structure and function in prokaryotes; carbon metabolism and its relation to obligate photoautotrophy; and special properties of cyanobacteria that are of ecological significance (temperature rela- tionships, nitrogen fixation and facultative anoxygenicphotosynthesis). 1. INTRODUCTION seated functional differences between the two The largest evolutionary discontinuity among kinds of cells could be appreciated only after contemporary organisms lies at the cellular the molecular biological revolution. level; it permits us to distinguish two biological In addition to the organisms long recognized superkingdoms, eukaryotes and prokaryotes as bacteria, the superkingdom of prokaryotes -

Title Hydrogen Atom Formation from the Photodissociation of Water Ice at 193 Nm Author(S)
Hydrogen atom formation from the photodissociation of water Title ice at 193 nm Yabushita, A; Hashikawa, Y; Ikeda, A; Kawasaki, M; Author(s) Tachikawa, H JOURNAL OF CHEMICAL PHYSICS (2004), 120(11): 5463- Citation 5468 Issue Date 2004-03-15 URL http://hdl.handle.net/2433/39757 Copyright 2004 American Institute of Physics. This article may be downloaded for personal use only. Any other use requires Right prior permission of the author and the American Institute of Physics. Type Journal Article Textversion none; publisher Kyoto University JOURNAL OF CHEMICAL PHYSICS VOLUME 120, NUMBER 11 15 MARCH 2004 Hydrogen atom formation from the photodissociation of water ice at 193 nm Akihiro Yabushita, Yuichi Hashikawa, Atsushi Ikeda, and Masahiro Kawasaki Department of Molecular Engineering, and Graduate School of Global Environmental Studies, Kyoto University, Kyoto 615-8510, Japan Hiroto Tachikawa Department of Molecular Chemistry, Graduate School of Engineering, Hokkaido University, Sapporo 060-8628, Japan ͑Received 21 April 2003; accepted 27 August 2003͒ The TOF spectra of photofragment hydrogen atoms from the 193 nm photodissociation of amorphous ice at 90–140 K have been measured. The spectra consist of both a fast and a slow ϭ Ϯ components that are characterized by average translational energies of 2kBTtrans 0.39 0.04 eV (2300Ϯ200 K) and 0.02 eV (120Ϯ20 K), respectively. The incident laser power dependency of the hydrogen atom production suggests one-photon process. The electronic excitation energy of a branched cluster, (H2O)6ϩ1 , has been theoretically calculated, where (H2O)6ϩ1 is a (H2O)6 cyclic cluster attached by a water molecule with the hydrogen bond. -

Enhancing the Effectiveness of Oxygen Evolution Reaction by Electrodeposition of Transition Metal Nanoparticles on Nickel Foam Material
catalysts Article Enhancing the Effectiveness of Oxygen Evolution Reaction by Electrodeposition of Transition Metal Nanoparticles on Nickel Foam Material Mateusz Łuba 1 , Tomasz Mikołajczyk 1,* , Mateusz Kuczy ´nski 1, Bogusław Pierozy˙ ´nski 1,* and Ireneusz M. Kowalski 2 1 Department of Chemistry, Faculty of Agriculture and Forestry, University of Warmia and Mazury in Olsztyn, Plac Lodzki 4, 10-727 Olsztyn, Poland; [email protected] (M.Ł.); [email protected] (M.K.) 2 Department of Rehabilitation, Faculty of Medical Sciences, University of Warmia and Mazury in Olsztyn, Zolnierska 14C Street, 10-561 Olsztyn, Poland; [email protected] * Correspondence: [email protected] (T.M.); [email protected] (B.P.); Tel.: +48-89-523-4177 (B.P.) Abstract: Electrochemical oxygen evolution reaction (OER) activity was studied on nickel foam-based electrodes. The OER was investigated in 0.1 M NaOH solution at room temperature on as-received and Co- or Mo-modified Ni foam anodes. Corresponding values of charge-transfer resistance, exchange current-density for the OER and other electrochemical parameters for the examined Ni foam composites were recorded. The electrodeposition of Co or Mo on Ni foam base-materials resulted in a significant enhancement of the OER electrocatalytic activity. The quality and extent Citation: Łuba, M.; Mikołajczyk, T.; of Co, and Mo electrodeposition on Ni foam were characterized by means of scanning electron Kuczy´nski,M.; Pierozy´nski,B.;˙ microscopy (SEM) and energy-dispersive X-ray spectroscopy (EDX) analysis. Kowalski, I.M. Enhancing the Effectiveness of Oxygen Evolution Keywords: oxygen evolution reaction; Ni foam; Co-modified Ni foam; Mo-modified Ni foam; Reaction by Electrodeposition of electrodeposition; electrochemical impedance spectroscopy Transition Metal Nanoparticles on Nickel Foam Material. -

Thermophilic Lithotrophy and Phototrophy in an Intertidal, Iron-Rich, Geothermal Spring 2 3 Lewis M
bioRxiv preprint doi: https://doi.org/10.1101/428698; this version posted September 27, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. 1 Thermophilic Lithotrophy and Phototrophy in an Intertidal, Iron-rich, Geothermal Spring 2 3 Lewis M. Ward1,2,3*, Airi Idei4, Mayuko Nakagawa2,5, Yuichiro Ueno2,5,6, Woodward W. 4 Fischer3, Shawn E. McGlynn2* 5 6 1. Department of Earth and Planetary Sciences, Harvard University, Cambridge, MA 02138 USA 7 2. Earth-Life Science Institute, Tokyo Institute of Technology, Meguro, Tokyo, 152-8550, Japan 8 3. Division of Geological and Planetary Sciences, California Institute of Technology, Pasadena, CA 9 91125 USA 10 4. Department of Biological Sciences, Tokyo Metropolitan University, Hachioji, Tokyo 192-0397, 11 Japan 12 5. Department of Earth and Planetary Sciences, Tokyo Institute of Technology, Meguro, Tokyo, 13 152-8551, Japan 14 6. Department of Subsurface Geobiological Analysis and Research, Japan Agency for Marine-Earth 15 Science and Technology, Natsushima-cho, Yokosuka 237-0061, Japan 16 Correspondence: [email protected] or [email protected] 17 18 Abstract 19 Hydrothermal systems, including terrestrial hot springs, contain diverse and systematic 20 arrays of geochemical conditions that vary over short spatial scales due to progressive interaction 21 between the reducing hydrothermal fluids, the oxygenated atmosphere, and in some cases 22 seawater. At Jinata Onsen, on Shikinejima Island, Japan, an intertidal, anoxic, iron- and 23 hydrogen-rich hot spring mixes with the oxygenated atmosphere and sulfate-rich seawater over 24 short spatial scales, creating an enormous range of redox environments over a distance ~10 m. -

Molecular Biogeochemistry, Lecture 8
12.158 Lecture Pigment-derived Biomarkers (1) Colour, structure, distribution and function (2) Biosynthesis (3) Nomenclature (4) Aromatic carotenoids ● Biomarkers for phototrophic sulfur bacteria ● Alternative biological sources (5) Porphyrins and maleimides Many of the figures in this lecture were kindly provided by Jochen Brocks, RSES ANU 1 Carotenoid pigments ● Carotenoids are usually yellow, orange or red coloured pigments lutein β-carotene 17 18 19 2' 2 4 6 8 3 7 9 16 1 5 lycopenelycopene 2 Structural diversity ● More than 600 different natural structures are known, ● They are derived from the C40 carotenoid lycopene by varied hydrogenation, dehydrogenation, cyclization and oxidation reaction 17 18 19 2' 2 4 6 8 3 7 9 16 1 5 lycopene neurosporene α-carotene γ -carotene spirilloxanthin siphonaxanthin canthaxanthin spheroidenone 3 Structural diversity Purple non-sulfur bacteria peridinin 7,8-didehydroastaxanthin okenone fucoxanthin Biological distribution ● Carotenoids are biosynthesized de novo by all phototrophic bacteria, eukaryotes and halophilic archaea ● They are additionally synthesized by a large variety of non-phototrophs ● Vertebrates and invertebrates have to incorporate carotenoids through the diet, but have often the capacity to structurally modifiy them 4 Carotenoid function (1) Accessory pigments in Light Harvesting Complex (LHC) (annual production by marine phytoplancton alone: 4 million tons) e.g. LH-II Red and blue: protein complex Green: chlorophyll Yellow: lycopene (2) Photoprotection (3) photoreceptors for phototropism -

Detection of Purple Sulfur Bacteria in Purple and Non-Purple Dairy Wastewaters
Published September 16, 2015 Journal of Environmental Quality TECHNICAL REPORTS environmental microbiology Detection of Purple Sulfur Bacteria in Purple and Non-purple Dairy Wastewaters Robert S. Dungan* and April B. Leytem hototrophic microorganisms, which reside Abstract in aquatic, benthic, and terrestrial environments, contain The presence of purple bacteria in manure storage lagoons is pigments that allow them to use light as an energy source. often associated with reduced odors. In this study, our objec- PAnoxygenic photosynthesis among prokaryotes (in contrast to tives were to determine the occurrence of purple sulfur bacteria oxygenic photosynthesis) occurs in purple and green bacteria (PSB) in seven dairy wastewater lagoons and to identify possible linkages between wastewater properties and purple blooms. but does not result in the production of oxygen (Madigan and Community DNA was extracted from composited wastewater Martinko, 2006). Anoxyphototrophs, such as purple sulfur samples, and a conservative 16S rRNA gene sequence within bacteria (PSB) and some purple nonsulfur bacteria (PNSB), Chromatiaceae and pufM genes found in both purple sulfur and use reduced sulfur compounds (e.g., hydrogen sulfide [H2S], nonsulfur bacteria was amplified. Analysis of the genes indicated elemental S), thiosulfate, and molecular hydrogen as electron that all of the lagoons contained sequences that were 92 to 97% similar with Thiocapsa roseopersicina. Sequences from a few la- donors in photosynthesis (Dilling et al., 1995; Asao et al., 2007). goons were also found to be similar with other PSB, such as Mari- Purple sulfur bacteria can also photoassimilate a number of chromatium sp. (97%), Thiolamprovum pedioforme (93–100%), simple organic compounds in the presence of sulfide, including and Thiobaca trueperi (95–98%). -

Aerobic Respiration
Life is based on redox • All energy generation in biological systems is due to redox (reduction-oxidation) reactions Aerobic Respiration: + - C6H12O6 + 6 H2O ==> 6 CO2 + 24 H +24 e oxidation electron donor (aka energy source) + - (O2+ 4H + 4e ==> 2H2O) x6 reduction electron acceptor --------------------------------------- C6H12O6 + 6 O2 ==> 6 CO2 + 6 H2O overall reaction (24 electrons) Types of bacterial metabolisms • While eukaryotes only reduce O2 and oxidize organic compounds, prokaryotes can use a variety of electron donors and acceptors, organic and inorganic. - • Aerobic respiration: e acceptor is O2 - • Anaerobic respiration: e acceptor is not O2 • Fermentation: e- donor and acceptor are organic molecules • Chemolithotrophy: e- donor and acceptor are inorganic molecules • Phototrophy: e- donor is light and e- acceptor is either organic or inorganic all microorganisms energy source? chemical light chemotroph phototroph carbon source? carbon source? organic organic CO CO compound 2 compound 2 chemoheterotroph chemoautotroph photoheterotroph photoautotroph e- acceptor? Nitrifying and sulfur- use H O to reduce CO ? oxidizing bacteria 2 2 green non-sulfur and O Other than O 2 2 purple non-sulfur bacteria anoxygenic oxygenic photosynthesis: photosynthesis: green sulfur and most bacteria Organic Inorganic cyanobacteria compound compound purple sulfur bacteria fermentative organism anaerobic respiration: nitrate, sulfate, Fe(III) Aerobic or anaerobic respiration Chemolithotrophy Important molecules Redox Electron Carrier: for example the