(HLXB9) in Infant Acute Myeloid Leukemia
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Prognostic Utility and Clinical Outcomes of MNX1-AS1 Expression in Cancers: a Systematic Review and Meta-Analysis
The prognostic utility and clinical outcomes of MNX1-AS1 expression in cancers: a systematic review and meta-analysis Juan Li The rst aliated hospital, college of medicine, zhejiang university https://orcid.org/0000-0002-0121-7098 Wen Jin The rst aliated hospital, college of medicine, zhejiang university Zhengyu Zhang The rst aliated hospital, college of medicine, zhejiang university Jingjing Chu The rst aliated hospital, college of medicine, zhejiang university Hui Yang The rst aliated hospital, college of meicine, zhejiang university Chang Li the rst aliated hospital, college of medicine, zhejiang university Ruiyin Dong The rst aliated hospital, college of medicine, zhejiang university Cailian Zhao ( [email protected] ) https://orcid.org/0000-0001-8337-0610 Primary research Keywords: Long non-coding RNA, MNX1-AS1, Cancer, Prognosis Posted Date: March 25th, 2020 DOI: https://doi.org/10.21203/rs.3.rs-19089/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 1/9 Abstract Background: Recently, emerging studies have identied that MNX1-AS1 highly expressed among variety of cancers and related with worse prognosis of cancer patients. The purpose of this study was to evaluate the relationship between MNX1-AS1 expression with clinical features and prognosis in different cancers. Methods: In this study, we searched the Web of Science, PubMed, CNKI, and Wanfang databases to nd relevant studies of MNX1-AS1. Pooled hazard ratios (HRs) and odds ratios (ORs) with 95% condence intervals (CIs) were applied to explore the prognostic and clinical signicance of MNX1-AS1. Results: A total of 9 literatures were included in this study, including 882 cancer patients. -

Watsonjn2018.Pdf (1.780Mb)
UNIVERSITY OF CENTRAL OKLAHOMA Edmond, Oklahoma Department of Biology Investigating Differential Gene Expression in vivo of Cardiac Birth Defects in an Avian Model of Maternal Phenylketonuria A THESIS SUBMITTED TO THE GRADUATE FACULTY In partial fulfillment of the requirements For the degree of MASTER OF SCIENCE IN BIOLOGY By Jamie N. Watson Edmond, OK June 5, 2018 J. Watson/Dr. Nikki Seagraves ii J. Watson/Dr. Nikki Seagraves Acknowledgements It is difficult to articulate the amount of gratitude I have for the support and encouragement I have received throughout my master’s thesis. Many people have added value and support to my life during this time. I am thankful for the education, experience, and friendships I have gained at the University of Central Oklahoma. First, I would like to thank Dr. Nikki Seagraves for her mentorship and friendship. I lucked out when I met her. I have enjoyed working on this project and I am very thankful for her support. I would like thank Thomas Crane for his support and patience throughout my master’s degree. I would like to thank Dr. Shannon Conley for her continued mentorship and support. I would like to thank Liz Bullen and Dr. Eric Howard for their training and help on this project. I would like to thank Kristy Meyer for her friendship and help throughout graduate school. I would like to thank my committee members Dr. Robert Brennan and Dr. Lilian Chooback for their advisement on this project. Also, I would like to thank the biology faculty and staff. I would like to thank the Seagraves lab members: Jailene Canales, Kayley Pate, Mckayla Muse, Grace Thetford, Kody Harvey, Jordan Guffey, and Kayle Patatanian for their hard work and support. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

High Definition Analyses of Single Cohort, Whole Genome Sequencing Data Provides a Direct Route
medRxiv preprint doi: https://doi.org/10.1101/2021.08.28.21262560; this version posted September 1, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. All rights reserved. No reuse allowed without permission. High definition analyses of single cohort, whole genome sequencing data provides a direct route to defining sub-phenotypes and personalising medicine Joyce KE1,2, Onabanjo E3, Brownlow S3, Nur F3, Olupona KO3, Fakayode K3, Sroya M4, Thomas G4, Ferguson T3, Redhead J3, Millar CM3,5, Cooper N3,5, Layton DM3,5, Boardman-Pretty F6, Caulfield MJ6,7, Genomics England Research Consortium6, Shovlin CL2,3,8* 1Imperial College School of Medicine, Imperial College, London UK; 2Genomics England Respiratory Clinical Interpretation Partnership (GeCIP); 3West London Genomic Medicine Centre, Imperial College Healthcare NHS Trust, London UK; 4Department of Surgery and Cancer, Imperial College London, UK; 5Centre for Haematology, Department of Immunology and Inflammation, Imperial College London UK; 6Genomics England, UK; 7 William Harvey Research Institute, Queen Mary University of London, London UK; 8National Heart and Lung Institute, Imperial College London UK. Word Count 4778 Abstract 150 Figures – 5 Data Supplement File- 1 *Corresponding Author: Claire L. Shovlin PhD FRCP, National Heart and Lung Institute, Imperial Centre for Translational and Experimental Medicine, Imperial College London, Hammersmith Campus, Du Cane Road, London W12 0NN, UK. Email [email protected] NOTE: This preprint reports new research that has not been certified by peer review and should not be used to guide clinical practice. -

Table S1. Identified Proteins with Exclusive Expression in Cerebellum of Rats of Control, 10Mg F/L and 50Mg F/L Groups
Table S1. Identified proteins with exclusive expression in cerebellum of rats of control, 10mg F/L and 50mg F/L groups. Accession PLGS Protein Name Group IDa Score Q3TXS7 26S proteasome non-ATPase regulatory subunit 1 435 Control Q9CQX8 28S ribosomal protein S36_ mitochondrial 197 Control P52760 2-iminobutanoate/2-iminopropanoate deaminase 315 Control Q60597 2-oxoglutarate dehydrogenase_ mitochondrial 67 Control P24815 3 beta-hydroxysteroid dehydrogenase/Delta 5-->4-isomerase type 1 84 Control Q99L13 3-hydroxyisobutyrate dehydrogenase_ mitochondrial 114 Control P61922 4-aminobutyrate aminotransferase_ mitochondrial 470 Control P10852 4F2 cell-surface antigen heavy chain 220 Control Q8K010 5-oxoprolinase 197 Control P47955 60S acidic ribosomal protein P1 190 Control P70266 6-phosphofructo-2-kinase/fructose-2_6-bisphosphatase 1 113 Control Q8QZT1 Acetyl-CoA acetyltransferase_ mitochondrial 402 Control Q9R0Y5 Adenylate kinase isoenzyme 1 623 Control Q80TS3 Adhesion G protein-coupled receptor L3 59 Control B7ZCC9 Adhesion G-protein coupled receptor G4 139 Control Q6P5E6 ADP-ribosylation factor-binding protein GGA2 45 Control E9Q394 A-kinase anchor protein 13 60 Control Q80Y20 Alkylated DNA repair protein alkB homolog 8 111 Control P07758 Alpha-1-antitrypsin 1-1 78 Control P22599 Alpha-1-antitrypsin 1-2 78 Control Q00896 Alpha-1-antitrypsin 1-3 78 Control Q00897 Alpha-1-antitrypsin 1-4 78 Control P57780 Alpha-actinin-4 58 Control Q9QYC0 Alpha-adducin 270 Control Q9DB05 Alpha-soluble NSF attachment protein 156 Control Q6PAM1 Alpha-taxilin 161 -

Genetic Colocalization Atlas Points to Common Regulatory Sites and Genes for Hematopoietic Traits and Hematopoietic Contributions to Disease Phenotypes Christopher S
Thom and Voight BMC Medical Genomics (2020) 13:89 https://doi.org/10.1186/s12920-020-00742-9 RESEARCH ARTICLE Open Access Genetic colocalization atlas points to common regulatory sites and genes for hematopoietic traits and hematopoietic contributions to disease phenotypes Christopher S. Thom1,2,3,4 and Benjamin F. Voight2,3,4* Abstract Background: Genetic associations link hematopoietic traits and disease end-points, but most causal variants and genes underlying these relationships are unknown. Here, we used genetic colocalization to nominate loci and genes related to shared genetic signal for hematopoietic, cardiovascular, autoimmune, neuropsychiatric, and cancer phenotypes. Methods: Our aim was to identify colocalization sites for human traits among established genome-wide significant loci. Using genome-wide association study (GWAS) summary statistics, we determined loci where multiple traits colocalized at a false discovery rate < 5%. We then identified quantitative trait loci among colocalization sites to highlight related genes. In addition, we used Mendelian randomization analysis to further investigate certain trait relationships genome-wide. Results: Our findings recapitulated developmental hematopoietic lineage relationships, identified loci that linked traits with causal genetic relationships, and revealed novel trait associations. Out of 2706 loci with genome-wide significant signal for at least 1 blood trait, we identified 1779 unique sites (66%) with shared genetic signal for 2+ hematologic traits. We could assign some sites to specific developmental cell types during hematopoiesis based on affected traits, including those likely to impact hematopoietic progenitor cells and/or megakaryocyte-erythroid progenitor cells. Through an expanded analysis of 70 human traits, we defined 2+ colocalizing traits at 2123 loci from an analysis of 9852 sites (22%) containing genome-wide significant signal for at least 1 GWAS trait. -

Spatial and Temporal Specification of Neural Fates by Transcription Factor Codes François Guillemot
REVIEW 3771 Development 134, 3771-3780 (2007) doi:10.1242/dev.006379 Spatial and temporal specification of neural fates by transcription factor codes François Guillemot The vertebrate central nervous system contains a great diversity Box 1. Neurons and glial cells of neurons and glial cells, which are generated in the embryonic neural tube at specific times and positions. Several classes of transcription factors have been shown to control various steps in the differentiation of progenitor cells in the neural tube and to determine the identity of the cells produced. Recent evidence indicates that combinations of transcription factors of the homeodomain and basic helix-loop-helix families establish molecular codes that determine both where and when the different kinds of neurons and glial cells are generated. Introduction Neuron Oligodendrocyte Astrocyte A multitude of neurons of different types, as well as oligodendrocytes and astrocytes (see Box 1), are generated as the vertebrate central The vertebrate central nervous system comprises three primary cell nervous system develops. These different neural cells are generated types, including neurons and two types of glial cells. Neurons are at defined times and positions by multipotent progenitors located in electrically excitable cells that process and transmit information via the walls of the embryonic neural tube. Progenitors located in the the release of neurotransmitters at synapses. Different subtypes of ventral neural tube at spinal cord level first produce motor neurons, neurons can be distinguished by the morphology of their cell body which innervate skeletal muscles and later produce oligodendrocytes and dendritic tree, the type of cells they connect with via their axon, the type of neurotransmitter used, etc. -

Additional File 3.Pdf
************ globins ************ >Pdu_Egb_A1a MNGITVFLILAMASASLADDCTQLDMIKVKHQWAEVYGVESNRQEFGLAVFKRFFVIHPD RSLFVNVHGDNVYSPEFQAHVARVLAGVDILISSMDQEAIFKAAQKHYADFHKSKFGEVPLVEFGTAMRDVLP KYVGLRNYDNDSWSRCYAYITSKVE >Pdu_Egb_A1b MKGLLVFLVLASVSASLASECSSLDKIKVKNQWA RIHGSPSNRKAFGTAVFKRFFEDHPDRSLFANVNGNDIYSADFQAHVQRVFGGLDILIVSLDQDDLFTAAKSH YSEFHKKLGDVPFAEFGVAFLDTLSDFLPLRDYNQDPWSRCYNYIIS >Pdu_Egb_A1c MNTVTVVLVLLG CIASAMTGDCNTLQRTKVKYQWSIVYGATDNRQAFGTLVWRDFFGLYPDRSLFSGVRGENIYSPEFRAHVVRV FAGFDILISLLDQEDILNSALAHYAAFHKQFPSIPFKEFGVVLLEALAKTIPEQFDQDAWSQCYAVIVAGVTA >Pdu_Egb_A1d_alpha MYQILSVAVLVLSCLALGTLGEEVCGPLERIKVQHQWVSVYGADHDRLKVSTL VWKDFFEHHPEERARFERVNSDNIFSGDFRAHMVRVFAGFDLLIGVLNEEEIFKSAMIHYTKMHNDLGVTTEI IKEFGKSIARVLPEFMDGKPDITAWRPCFNLIAAGVSE >Pdu_Egb_A1d_beta MYFSYFTAAASYLSVAVLVLSCLVQGILGEEVCGPLEKIKVQHQWASAYRGDHD RLKMSTLVWKDFFAHNPEERARFERVHSDDIYSGDFRAHMVRVFAGFDLLIGALNQEDIFRSAMIHYTKMHKK LGVTYEIGIEFGKSIGRVLPEFIDGKLDITAWRPCYKLIATGVDE >Pdu_Egb_A1d_gamma MYLSVAVLVLSCLALGTQGEEVCGPLEKIKVQHQWASAYRGDHDRLKMSTLVW KDFFAHHPEERARFERVHSDDIYSGDFRAHMVRVFAGFDLLIGVLNQDEIFKSAMIHYTKMHNDLGVKTEIVL EFGKSIARVLPDFIDGKPDITAWRPCFKLIAAGVSE >Pdu_Egb_A2 MNNLVILVGLLCLGLTSATKCGPL QRLKVKQQWAKAYGVGHERLELGIALWKSIFAQDPESRSIFTRVHGDDVRHPAFEAHIARVFNGFDRIISSLT DEDVLQAQLAHLKAQHIKLGISAHHFKLMRTGLSYVLPAQLGRCFDKEAWGSCWDEVIYPGIKSL >Pdu_Egb_B1 MLVLAVFVAALGLAAADQCCSIEDRNEVQALWQSIWSAENTGKRTIIGHQIFEELFDINP GTKDLFKRVNVEDTSSPEFEAHVLRVMNGLDTLIGVLDDPATGYSLITHLAEQHKAREGFKPSYFKDIGVALK RVLPQVASCFNPEAWDHCFNGFVEAITNKMNAL >Pdu_Egb_B2 MLVLVLSLAFLGSALAEDCCSAADRKTVLRDWQSVWSAEFTGRRVAIGTAIFEELFAIDA GAKDVFKNVAVDKPESAEWAAHVIRVINGLDLAINLLEDPRALKEELLHLAKQHRERDGVKAVYFDEIGRALL -

Aberrant Development of Pancreatic Beta Cells Derived from Human Ipscs with FOXA2 Deficiency Ahmed K
Elsayed et al. Cell Death and Disease (2021) 12:103 https://doi.org/10.1038/s41419-021-03390-8 Cell Death & Disease ARTICLE Open Access Aberrant development of pancreatic beta cells derived from human iPSCs with FOXA2 deficiency Ahmed K. Elsayed1, Ihab Younis2, Gowher Ali1, Khalid Hussain3 and Essam M. Abdelalim 1,4 Abstract FOXA2 has been identified as an essential factor for pancreas development and emerging evidence supports an association between FOXA2 and diabetes. Although the role of FOXA2 during pancreatic development is well-studied in animal models, its role during human islet cell development remains unclear. Here, we generated induced pluripotent stem cells (iPSCs) from a patient with FOXA2 haploinsufficiency (FOXA2+/− iPSCs) followed by beta-cell differentiation to understand the role of FOXA2 during pancreatic beta-cell development. Our results showed that FOXA2 haploinsufficiency resulted in aberrant expression of genes essential for the differentiation and proper functioning of beta cells. At pancreatic progenitor (PP2) and endocrine progenitor (EPs) stages, transcriptome analysis showed downregulation in genes associated with pancreatic development and diabetes and upregulation in genes associated with nervous system development and WNT signaling pathway. Knockout of FOXA2 in control iPSCs (FOXA2−/− iPSCs) led to severe phenotypes in EPs and beta-cell stages. The expression of NGN3 and its downstream targets at EPs as well as INSUILIN and GLUCAGON at the beta-cell stage, were almost absent in the cells derived from FOXA2−/− iPSCs. These findings indicate that FOXA2 is crucial for human pancreatic endocrine development and its defect may lead to diabetes based on FOXA2 dosage. 1234567890():,; 1234567890():,; 1234567890():,; 1234567890():,; Introduction TFs lead to neonatal diabetes, which can be associated During human development, early endodermal tissue with pancreatic hypoplasia/agenesis in some mutations4. -
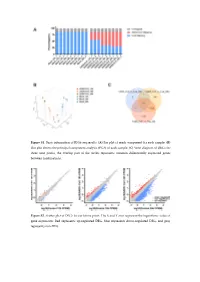
Figure S1. Basic Information of RNA-Seq Results. (A) Bar Plot of Reads Component for Each Sample
Figure S1. Basic information of RNA-seq results. (A) Bar plot of reads component for each sample. (B) Dot plot shows the principal component analysis (PCA) of each sample. (C) Venn diagram of DEGs for three time points, the overlap part of the circles represents common differentially expressed genes between combinations. Figure S2. Scatter plot of DEGs for each time point. The X and Y axes represent the logarithmic value of gene expression. Red represents up-regulated DEG, blue represents down-regulated DEG, and gray represents non-DEG. Table S1. Primers used for quantitative real-time PCR analysis of DEGs. Gene Primer Sequence Forward 5’-CTACGAGTGGATGGTCAAGAGC-3’ FOXO1 Reverse 5’-CCAGTTCCTTCATTCTGCACACG-3’ Forward 5’-GACGTCCGGCATCAGAGAAA-3’ IRS2 Reverse 5’-TCCACGGCTAATCGTCACAG-3’ Forward 5’-CACAACCAGGACCTCACACC-3’ IRS1 Reverse 5’-CTTGGCACGATAGAGAGCGT-3’ Forward 5’-AGGATACCACTCCCAACAGACCT-3’ IL6 Reverse 5’-CAAGTGCATCATCGTTGTTCATAC-3’ Forward 5’-TCACGTTGTACGCAGCTACC-3’ CCL5 Reverse 5’-CAGTCCTCTTACAGCCTTTGG-3’ Forward 5’-CTGTGCAGCCGCAGTGCCTACC-3’ BMP7 Reverse 5’-ATCCCTCCCCACCCCACCATCT-3’ Forward 5’-CTCTCCCCCTCGACTTCTGA-3’ BCL2 Reverse 5’-AGTCACGCGGAACACTTGAT-3’ Forward 5’-CTGTCGAACACAGTGGTACCTG-3’ FGF7 Reverse 5’-CCAACTGCCACTGTCCTGATTTC-3’ Forward 5’-GGGAGCCAAAAGGGTCATCA-3’ GAPDH Reverse 5’-CGTGGACTGTGGTCATGAGT-3’ Supplementary material: Differentially expressed genes log2(SADS-CoV_12h/ Qvalue (SADS-CoV _12h/ Gene Symbol Control_12h) Control_12h) PTGER4 -1.03693 6.79E-04 TMEM72 -3.08132 3.66E-04 IFIT2 -1.02918 2.11E-07 FRAT2 -1.09282 4.66E-05 -

Transdifferentiation to Pancreatic Progenitors
UNIVERSITY OF MANCHESTER Transdifferentiation to pancreatic progenitors A thesis submitted to the University of Manchester for the degree of Doctor of Philosophy in the Faculty of Biology, Medicine and Health 2019 Stephen John Wearne School of Medical Sciences Division of Diabetes Endocrinology and Gastroenterology Contents Contents..................................................................................................................................................................................... 1 List of tables ............................................................................................................................................................................... 3 List of figures.............................................................................................................................................................................. 4 List of abbreviations ................................................................................................................................................................... 5 List of publications ..................................................................................................................................................................... 8 Abstract ...................................................................................................................................................................................... 9 Declaration .............................................................................................................................................................................. -

Related to Figure 1
Figure S1. Temporal and spatial documentation of Olig2Cre-mediated recombination (related to Figure 1). (A-I) Serial transverse spinal cord sections from E8.5-E9.5 Control (Olig2Cre; R26RGFP) embryos. Tissue was immunostained with Nkx2.2, Olig2, Cre, and GFP antibodies to assess where and when recombination occurred. Scale bars = 50 µm. (J-Q) Analysis of E10.5 Olig2Cre; R26RGFP Control mice shows that Cre recombination takes place in both the pMN (Nkx6.1+/Olig2+) and p3 (Nkx6.1+/Nxk2.2+) domains, but not in the floor plate (FoxA2+ or Shh+). At this time point, Cre recombination had occurred in nearly all MNs (Isl1/2+), including both LMC (FoxP1+) and MMC (Lhx3+) subgroups. Inset in (P) reveals a small population of non-recombined MNs. Note that Lhx3 is also prominently expressed by newborn V2 interneurons that form above the motor columns. (R-W) Notch-Off embryos display an absence of Rbpj protein from pMN, p3 progenitors, and differentiated (Islet+/Hb9+) MNs starting around E9.5. Note that Sox2 is maintained in pMN/p3 cells despite the loss of Rbpj. Scale bars = 50 µm. Figure S2. Notch signaling is activated in Notch-On embryos and reduced in Notch-Off embryos without any major disruptions to the neuroepithelial organization (related to Figures 1 and 2). (A-U) Analysis of Notch signaling activity in Control (A-G), Notch-On (H-N), and Notch-Off (O-U) E11.5 embryos. (A, H, O) Green brackets demarcate the dorsoventral extent of the GFP+ region of recombination and white brackets indicate the thickness of the Sox2+ ventricular zone (VZ) within this same region.