On the Cyanogen Halides by P
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Spectrophotometric Determination of Thiocyanate In
Investigative Forensic Sciences © All rights are reserved by Buddha D. Paul and Thomas Research Article Open Access Spectrophotometric Determination of Thiocyanate in Human Saliva by a Unique Iodine-Azide-Chromogenic Substrate Reaction and its Application in Distinguishing Tobacco Smokers from Non-Smokers† Buddha D. Paul and Thomas Bosy Division of Forensic Toxicology, Office of the Armed Forces Medical Examiner, Dover AFB, Delaware, USA. Abstract A method to detect thiocyanate (SCN) in human saliva is presented. Thiocyanate concentrations appear to be diagnostic when classifying smokers or non-smokers, and in determining some clinical conditions. The method involves the reaction of SCN with excess iodine and azide, and spectroscopic detection of unreacted iodine by a chromogenic substrate, ABTS. The calibration was linear over the range of 12.5-150 µmol/L (slope = 0.0086 delta-Abs/SCN µmol/L, intercept = -0.0160 delta-Abs, R2 0.9998). The method was applied to analyze 29 saliva specimens. The results were similar to those obtained from a gas chromatography-mass 2 spectrometry method (slope = 0.9595, R 0.9790). Based on Grubbs equation applied to specimens from non-smoking subjects, a threshold concentration of 1100 µmol/L for SCN was determined to distinguish smokers from the non-smokers. The SCN concentrations in 18 out of 20 saliva specimens collected from 2 smokers were above this threshold. The specimens from smokers were also examined for nicotine and cotinine by a GCMS method. While nicotine concentrations were found to vary, the cotinine concentrations remained stable, 134+29 ng/mL. Generally, the presence of nicotine/cotinine in specimens only indicates exposure to tobacco products, but the presence of any of these compounds with elevated SCN, is an indication of smoking. -

Calarp) Program
California Accidental Release Prevention (CalARP) Program Administering Agency Guidance January 31, 2005 Preface This document provides general guidance to help Administering Agencies (AAs) implement and enforce the California Accidental Release Prevention (CalARP) Program. The intent is to identify the elements of the Program applicable to each regulated business, and assist AAs with oversight of the CalARP Program statutes and regulations. This document is not a substitute for the CalARP Program regulations; it does not impose legally binding requirements. About This Document This document follows the format of the California Code of Regulations, Title 19, Division 2, Chapter 4.5: California Accidental Release Prevention (CalARP) Program. The regulatory sections are presented in parentheses for ease of reference. Acknowledgements The California Emergency Management Agency (Cal EMA) would like to thank the following people for their valuable assistance in the preparation of this document: Howard Wines, Hazardous Materials Specialist, City of Bakersfield Fire Department Robert Distaso P.E., Fire Safety Engineer, Orange County Fire Authority Randall L. Sawyer, Supervisor, Accidental Release Prevention Programs, Contra Costa County Health Services Department Beronia Beniamine, Senior Hazardous Materials Specialist, Stanislaus County Environmental Resources Department Angie Proboszcz, Risk Management Program Coordinator, USEPA Region 9 Jon Christenson, Senior Environmental Health Specialist, Merced County Department of Public Health Teresa -

Solubility and Solution-Phase Chemistry of Isocyanic Acid, Methyl Isocyanate, 2 and Cyanogen Halides 3 4 5 6 James M
Atmos. Chem. Phys. Discuss., https://doi.org/10.5194/acp-2018-1160 Manuscript under review for journal Atmos. Chem. Phys. Discussion started: 9 November 2018 c Author(s) 2018. CC BY 4.0 License. 1 Solubility and Solution-phase Chemistry of Isocyanic Acid, Methyl Isocyanate, 2 and Cyanogen Halides 3 4 5 6 James M. Roberts1, and Yong Liu2 7 8 1. NOAA/ESRL Chemical Sciences Division, Boulder, Colorado, 80305 9 2. Department of Chemistry, University of Colorado, Denver, Denver, Colorado, 80217 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 Atmos. Chem. Phys. Discuss., https://doi.org/10.5194/acp-2018-1160 Manuscript under review for journal Atmos. Chem. Phys. Discussion started: 9 November 2018 c Author(s) 2018. CC BY 4.0 License. 52 Abstract 53 54 Condensed phase uptake and reaction are important atmospheric removal processes for reduced nitrogen 55 species, isocyanic acid (HNCO), methyl isocyanate (CH3NCO) and cyanogen halides (XCN, X =Cl, Br, I), yet many 56 of the fundamental quantities that govern this chemistry have not been measured or are understudied. Solubilities 57 and first-order reaction rates of these species were measured for a variety of solutions using a bubble flow reactor 58 method with total reactive nitrogen (Nr) detection. The aqueous solubility of HNCO was measured as a function of 59 pH, and exhibited the classic behavior of a weak acid, with an intrinsic Henry’s law solubility of 20 (±2) M/atm, and -4 60 a Ka of 2.0 (±0.28) ×10 M (which corresponds to pKa = 3.7 ±0.06) at 298K. -

Excess Heat Production in the Redox Couple Reaction of Ferricyanide and Ferrocyanide
www.nature.com/scientificreports OPEN Excess heat production in the redox couple reaction of ferricyanide and ferrocyanide Atsushi Sugiyama1,2,3*, Makoto Miura4, Yoshinobu Oshikiri5, Yena Kim3, Ryoichi Morimoto6, Miki Miura7, Tetsuya Osaka2, Iwao Mogi8, Yusuke Yamauchi3,9* & Ryoichi Aogaki3,10* In order to establish the universality of the excess heat production in electrochemical reaction, under a high magnetic feld, as one of the most fundamental electrochemical reactions, the case of ferricyanide-ferrocyanide redox reaction was examined, where ionic vacancies with ± 1 unit charge were collided by means of magnetohydrodynamic (MHD) fow. As a result, from the pair annihilation of the vacancies with opposite signs, beyond 7 T, excess heat production up to 25 kJ·mol−1 in average at 15 T was observed, which was attributed to the liberation of the solvation energy stored in a pair of the vacancy cores with a 0.32 nm radius, i.e., 112 kJ·mol−1. Diference between the observed and expected energies comes from the small collision efciency of 0.22 due to small radius of the vacancy core. Ionic vacancy initially created as a by-product of electrode reaction is unstable in solution phase, stabilized by releasing solvation energy. Ionic vacancy utilizes the energy to enlarge the core and stores the energy in it. As a result, solvated ionic vacancy consists of a polarized free space of the enlarged core surrounded by oppositely charged ionic cloud. The accuracy and precision of the measured values were ascertained by in situ standard additive method. Recently, in copper redox reaction under a high magnetic feld, great excess heat production up to 410 kJ·mol−1 in average has been observed, which is 1.5 times larger than the molar combustion heat of hydrogen (285.8 kJ·mol−1)1. -

Reversibility of Ferri-/Ferrocyanide Redox During Operando Soft X-Ray Spectroscopy
Reversibility of Ferri-/Ferrocyanide Redox during Operando Soft X-ray Spectroscopy The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation Risch, Marcel, Kelsey A. Stoerzinger, Tom Z. Regier, Derek Peak, Sayed Youssef Sayed, and Yang Shao-Horn. “Reversibility of Ferri-/ Ferrocyanide Redox During Operando Soft X-Ray Spectroscopy.” The Journal of Physical Chemistry C 119, no. 33 (August 20, 2015): 18903–18910. As Published http://dx.doi.org/10.1021/acs.jpcc.5b04609 Publisher American Chemical Society (ACS) Version Author's final manuscript Citable link http://hdl.handle.net/1721.1/109590 Terms of Use Article is made available in accordance with the publisher's policy and may be subject to US copyright law. Please refer to the publisher's site for terms of use. Reversibility of Ferri-Ferrocyanide Redox During Operando Soft X-Ray Spectroscopy ┴ Marcel Risch,†* Kelsey A. Stoerzinger,§ Tom Z. Regier,‡ Derek Peak,º Sayed Youssef Sayed,† Yang Shao-Horn†§||* †Research Laboratory of Electronics, Massachusetts Institute of Technology, Cambridge, MA, USA 02139 §Department of Materials Science and Engineering, Massachusetts Institute of Technology, Cambridge, MA, USA 02139 ||Department of Mechanical Engineering, Massachusetts Institute of Technology, Cambridge, MA, USA 02139 ‡Canadian Light Source, Inc., Saskatoon, SK, Canada S7N 2V3 ºDepartment of Soil Sciences, University of Saskatchewan, Saskatoon, SK, Canada S7N 5A8 KEYWORDS. Electrochemistry; In-situ; redox shuttle; iron cyanide; X-ray absorption; radiolysis; radiation damage. ABSTRACT The ferri-ferrocyanide redox couple is ubiquitous in many fields of physical chemistry. We studied its photochemical response to intense synchrotron radiation by in-situ X-Ray absorption spectroscopy. -
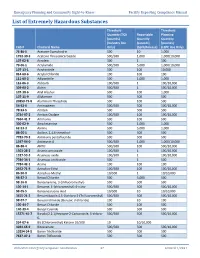
List of Extremely Hazardous Substances
Emergency Planning and Community Right-to-Know Facility Reporting Compliance Manual List of Extremely Hazardous Substances Threshold Threshold Quantity (TQ) Reportable Planning (pounds) Quantity Quantity (Industry Use (pounds) (pounds) CAS # Chemical Name Only) (Spill/Release) (LEPC Use Only) 75-86-5 Acetone Cyanohydrin 500 10 1,000 1752-30-3 Acetone Thiosemicarbazide 500/500 1,000 1,000/10,000 107-02-8 Acrolein 500 1 500 79-06-1 Acrylamide 500/500 5,000 1,000/10,000 107-13-1 Acrylonitrile 500 100 10,000 814-68-6 Acrylyl Chloride 100 100 100 111-69-3 Adiponitrile 500 1,000 1,000 116-06-3 Aldicarb 100/500 1 100/10,000 309-00-2 Aldrin 500/500 1 500/10,000 107-18-6 Allyl Alcohol 500 100 1,000 107-11-9 Allylamine 500 500 500 20859-73-8 Aluminum Phosphide 500 100 500 54-62-6 Aminopterin 500/500 500 500/10,000 78-53-5 Amiton 500 500 500 3734-97-2 Amiton Oxalate 100/500 100 100/10,000 7664-41-7 Ammonia 500 100 500 300-62-9 Amphetamine 500 1,000 1,000 62-53-3 Aniline 500 5,000 1,000 88-05-1 Aniline, 2,4,6-trimethyl- 500 500 500 7783-70-2 Antimony pentafluoride 500 500 500 1397-94-0 Antimycin A 500/500 1,000 1,000/10,000 86-88-4 ANTU 500/500 100 500/10,000 1303-28-2 Arsenic pentoxide 100/500 1 100/10,000 1327-53-3 Arsenous oxide 100/500 1 100/10,000 7784-34-1 Arsenous trichloride 500 1 500 7784-42-1 Arsine 100 100 100 2642-71-9 Azinphos-Ethyl 100/500 100 100/10,000 86-50-0 Azinphos-Methyl 10/500 1 10/10,000 98-87-3 Benzal Chloride 500 5,000 500 98-16-8 Benzenamine, 3-(trifluoromethyl)- 500 500 500 100-14-1 Benzene, 1-(chloromethyl)-4-nitro- 500/500 -

No, Potassium Ferrocyanide in Some Salt Brands Is Not at Toxic Levels
Fact check: No, potassium ferrocyanide in some salt brands is not at toxic levels Shubashree Desikan JULY 02, 2019 Citing a report from U.S.-based West American Analytical Laboratories, Shiv Shankar Gupta, chairman of Godhum Grains and Food Products, accused some salt manufacturers in India of selling food-grade salt that contained high levels of potassium ferrocyanide. This caused a panic, which was aggravated by widespread sharing of the news item on social media. Indeed, potassium ferrocyanide is being used in salt to give it anti-caking properties. But the answer to whether it is toxic is no! Firstly, while potassium cyanide is a toxic substance and releases the cyanide anion when consumed by a person, potassium ferrocyanide is not. In potassium ferrocyanide, the cyanide anion is strongly bonded to ferrous ion and hence does not get hydrolysed. An image widely shared in social media. Secondly, there is the factor known as the LD50 value. This is expanded as “Lethal Dose 50%”. LD50 is the amount of any chemical that can cause death in 50% of the group of animals that it enters by consumption or absorption through the skin. According to inorganic chemist Dr. Sayam Sengupta of IISER Kolkata, the LD50 value for common salt — sodium chloride — for rats is greater than 3 gram per kilogram body weight of the animal. He points out that for potassium Ferrocyanide, LD 50 value is 3.6 gram per kilogram weight of the animal, when taken orally. As this indicates, the LD50 value of potassium ferrocyanide is almost the same as common salt. -

List of Lists
United States Office of Solid Waste EPA 550-B-10-001 Environmental Protection and Emergency Response May 2010 Agency www.epa.gov/emergencies LIST OF LISTS Consolidated List of Chemicals Subject to the Emergency Planning and Community Right- To-Know Act (EPCRA), Comprehensive Environmental Response, Compensation and Liability Act (CERCLA) and Section 112(r) of the Clean Air Act • EPCRA Section 302 Extremely Hazardous Substances • CERCLA Hazardous Substances • EPCRA Section 313 Toxic Chemicals • CAA 112(r) Regulated Chemicals For Accidental Release Prevention Office of Emergency Management This page intentionally left blank. TABLE OF CONTENTS Page Introduction................................................................................................................................................ i List of Lists – Conslidated List of Chemicals (by CAS #) Subject to the Emergency Planning and Community Right-to-Know Act (EPCRA), Comprehensive Environmental Response, Compensation and Liability Act (CERCLA) and Section 112(r) of the Clean Air Act ................................................. 1 Appendix A: Alphabetical Listing of Consolidated List ..................................................................... A-1 Appendix B: Radionuclides Listed Under CERCLA .......................................................................... B-1 Appendix C: RCRA Waste Streams and Unlisted Hazardous Wastes................................................ C-1 This page intentionally left blank. LIST OF LISTS Consolidated List of Chemicals -

Chemical Hygiene Plan Ii Revised 03/2021 Table of Contents
MAR 2021 Office of Environmental Health and Safety Principal Author/Editor: David Webber, PhD/Chemical Hygiene Officer Contributing Authors/Editors: Nikolai Evdokimov, PhD, James Gibson, PhD, Tania Guardado, PhD, Amanda Jevons, Deona Willes, MPH Graphics/Design: Alfred M. Bouziane, MS, Brent Pantell USC Chemical Hygiene Plan ii Revised 03/2021 Table of Contents i.0 2021 Revision Summary Section 3.0 vi Section 4.0 vi Section 5.0 vii Section 7.0 vii Section 8.0 viii Section 10.0 x Appendix D x Appendix G x 1.0 Introduction Purpose and Scope 1.1 Sources of Safety Information 1.2 2.0 Regulatory Requirements 3.0 Roles and Responsibilities Research Safety Oversight Committee (RSOC) 3.1 Campus-Wide Chemical Safety Committee (CCSC) 3.1 Other Safety Committees 3.2 Office of Environmental Health & Safety 3.2 Principal Investigator (PI) 3.3 Training Requirements 3.5 4.0 Basics of Laboratory Safety Hazard, Risk, and Safety Management 4.1 Hierarchy of Safety Controls 4.1 Group Safety Management and Safety Culture 4.4 USC Chemical Hygiene Plan iii Revised 03/2021 Basics of Lab Facilities, Equipment, and Emergency Supplies 4.7 Emergency Equipment and Supplies 4.15 Open Flames 4.20 5.0 Hazard Communication Labeling and Signage Systems 5.2 Labelling and Signage in the Lab: What You Need to Do 5.5 Safety Data Sheets (SDSs): What Are They? 5.7 SDSs in The Lab: What You Need to Do 5.8 6.0 Hazardous Chemicals and Hazard Classification Introduction 6.1 Health-Hazardous Chemicals: Routes of Exposure 6.2 Particularly Hazardous Substances (PHS) 6.16 7.0 Chemical -

The Analysis of Cyanide Silver-Plating Solutions
RP384 THE ANALYSIS OF CYANIDE SILVER-PLATING SOLUTIONS By R. M. Wick ABSTRACT Methods for the analysis of cyanide silver-plating solutions were studied, including the determination of free cyanide, total cyanide, carbonate, chloride, ammonia, silver, iron, copper, and mercury. Electrometric titrations showed that the Liebig method for alkali cyanide is correct to better than 0.2 per cent. Addition of iodide makes the method still more accurate and overcomes the effects of impurities on the titration. The nonagreement of the Liebig and Hannay methods was studied by electro- metric titrations, which showed that the visual Hannay method gives high results although the electrometric Hannay titration is accurate. The determination of total cyanide was accomplished by distillation with sulphuric acid, and of total effective cyanide by titration with iodine. Silver may be separated as silver sulphide, by precipitation with zinc, or by decomposition with acid, after which a determination by any standard method is applicable. Carbonate may be determined by precipitating and filtering out barium car- bonate and titrating it with acid or by titrating a sample with acid after adding silver nitrate to combine with the free cyanide. The usual method for the determination of chloride involves prolonged treat- ment with nitric acid, which converts the precipitated silver cyanide to silver chloride. A new method for separating chloride was investigated in which the silver is precipitated as sulphide and the iron is removed by precipitation as manganous ferrocyanide. The chloride in the filtrate can be determined by the usual methods. Iron and copper can be determined by the regular analytical methods after decomposition of the plating solution with sulphuric acid. -

Phenethyl Isothiocyanate (Peitc) Decreases Specificity
PHENETHYL ISOTHIOCYANATE (PEITC) DECREASES SPECIFICITY PROTEIN (SP) TRANSCRIPTION FACTORS THROUGH AN ROS- DEPENDENT MECHANISM A Thesis by AARON SHANE GUTHRIE Submitted to the Office of Graduate Studies of Texas A&M University in partial fulfillment of the requirements for the degree of MASTER OF SCIENCE Approved by: Chair of Committee, Stephen Safe Committee Members, Robert Burghardt Xiuren Zhang Head of Department, Gregory D. Reinhart December 2012 Major Subject: Biochemistry Copyright 2012 Aaron Shane Guthrie ABSTRACT Isothiocyanates (ITCs) are phytochemicals highly expressed in cruciferous vegetables and these compounds are associated with the decreased incidence of cancers in populations consuming high levels of cruciferous vegetables. Several individual ITCs including phenethyl isothiocyanate (PEITC) inhibit tumor growth and angiogenesis and their anticancer activity has been linked to inhibition of cancer cell growth, survival and inflammation (NFB). It has also been demonstrated that PEITC induces reactive oxygen species (ROS) and that ROS is largely responsible for PEITC-induced cell death. To confirm PEITC-induced cancer cell death we have investigated the mechanism of action of PEITC in pancreatic cancer cell lines and PEITC induces ROS and inhibits growth and induces apoptosis (PARP cleavage). In addition, PEITC downregulates expression of several gene products including vascular endothelial growth factor (VEGF), cyclin D1 (CD1), Bcl2 and survivin and these have previously been reported in other studies. However, since these gene products are all regulated by specificity protein (Sp) transcription factors Sp1, Sp3 and Sp4, which are overexpressed in cancer cells and tumors, we investigated the effects of PEITC on Sp proteins and observed that PEITC decreased expression of Sp1, Sp3 and Sp4 in pancreatic cancer cells. -

Massachusetts Oil and Hazardous Material List
SUBPART P: MASSACHUSETTS OIL AND HAZARDOUS MATERIAL LIST TABLE OF CONTENTS TABLE 1 - MASSACHUSETTS OIL AND HAZARDOUS MATERIAL LIST (ALPHABETICAL LISTING) TABLE 2 - MASSACHUSETTS OIL AND HAZARDOUS MATERIAL LIST (BY CAS NUMBER ORDER) NOTES: The Massachusetts Oil and Hazardous Materials List (MOHML) contains oils and hazardous materials subject to 310 CMR 40.0000 and their reportable quantities (RQs) and reportable concentrations (RCs). These values are referred to in the notification requirements (310 CMR 40.0300). This list is provided both alphabetically in Table 1 and by Chemical Abstracts Service Number (CAS Number) in Table 2. The CAS number is a unique number assigned to a substance. Both tables identify other lists on which a substance appears by using name source codes. These codes are as follows: Name Source 1 - The Department of Transportation (DOT) Hazardous Materials List (49 CFR Part 172.101 Hazardous Materials Table) Name Source 2 - The Resource Conservation and Recovery Act Appendix VIII List (40 CFR Part 261 - Appendix VIII Hazardous Constituents) Name Source 3 - The Comprehensive Environmental Response, Compensation, and Liability Act (CERCLA) Hazardous Substance and Waste Stream Lists (40 CFR Part 302 - Table 302.4) Name Source 4 - The Extremely Hazardous Substance List as mandated by Superfund Amendments and Reauthorization Act, Title III, Section 302 (40 CFR Part 355 Appendices A and B) Name Source 5 - DEP Allowable Ambient Limits (AALs) and Drinking Water Guidelines Name Source 6 - The Massachusetts Substance List (MSL)(105 CMR 670.000: “Right to Know” Appendix A) Name Source 7 - The Chemical Abstracts name, 9th collective period, 1972-1976 Name Source 8 - The EPA Right to Know list, Section 313 of the Emergency Planning and Community Right to Know Act of 1986 (40 CFR Part 372.65).