(HPG) Axis Published: 18 April 2017 Matthew D
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Duplication 9P and Their Implication to Phenotype
Guilherme et al. BMC Medical Genetics (2014) 15:142 DOI 10.1186/s12881-014-0142-1 RESEARCH ARTICLE Open Access Duplication 9p and their implication to phenotype Roberta Santos Guilherme1, Vera Ayres Meloni1, Ana Beatriz Alvarez Perez1, Ana Luiza Pilla1, Marco Antonio Paula de Ramos1, Anelisa Gollo Dantas1, Sylvia Satomi Takeno1, Leslie Domenici Kulikowski2 and Maria Isabel Melaragno1* Abstract Background: Trisomy 9p is one of the most common partial trisomies found in newborns. We report the clinical features and cytogenomic findings in five patients with different chromosome rearrangements resulting in complete 9p duplication, three of them involving 9p centromere alterations. Methods: The rearrangements in the patients were characterized by G-banding, SNP-array and fluorescent in situ hybridization (FISH) with different probes. Results: Two patients presented de novo dicentric chromosomes: der(9;15)t(9;15)(p11.2;p13) and der(9;21)t(9;21) (p13.1;p13.1). One patient presented two concomitant rearranged chromosomes: a der(12)t(9;12)(q21.13;p13.33) and an psu i(9)(p10) which showed FISH centromeric signal smaller than in the normal chromosome 9. Besides the duplication 9p24.3p13.1, array revealed a 7.3 Mb deletion in 9q13q21.13 in this patient. The break in the psu i(9) (p10) probably occurred in the centromere resulting in a smaller centromere and with part of the 9q translocated to the distal 12p with the deletion 9q occurring during this rearrangement. Two patients, brother and sister, present 9p duplication concomitant to 18p deletion due to an inherited der(18)t(9;18)(p11.2;p11.31)mat. -

WO 2012/174282 A2 20 December 2012 (20.12.2012) P O P C T
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2012/174282 A2 20 December 2012 (20.12.2012) P O P C T (51) International Patent Classification: David [US/US]; 13539 N . 95th Way, Scottsdale, AZ C12Q 1/68 (2006.01) 85260 (US). (21) International Application Number: (74) Agent: AKHAVAN, Ramin; Caris Science, Inc., 6655 N . PCT/US20 12/0425 19 Macarthur Blvd., Irving, TX 75039 (US). (22) International Filing Date: (81) Designated States (unless otherwise indicated, for every 14 June 2012 (14.06.2012) kind of national protection available): AE, AG, AL, AM, AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, English (25) Filing Language: CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, Publication Language: English DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, KR, (30) Priority Data: KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, ME, 61/497,895 16 June 201 1 (16.06.201 1) US MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, 61/499,138 20 June 201 1 (20.06.201 1) US OM, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SC, SD, 61/501,680 27 June 201 1 (27.06.201 1) u s SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, 61/506,019 8 July 201 1(08.07.201 1) u s TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. -

DNMT Inhibitors Increase Methylation at Subset of Cpgs in Colon
bioRxiv preprint doi: https://doi.org/10.1101/395467; this version posted August 25, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. 1 Title: DNMT inhibitors increase methylation at subset of CpGs in colon, bladder, lymphoma, 2 breast, and ovarian, cancer genome 3 Running title: Decitabine/azacytidine increases DNA methylation 4 Anil K Giri1, Tero Aittokallio1,2 5 1Institute for Molecular Medicine Finland, FIMM, University of Helsinki, Helsinki, Finland. 6 2Department of Mathematics and Statistics, University of Turku, Turku, Finland. 7 Correspondence to 8 Dr. Anil K Giri 9 Institute for Molecular Medicine Finland FIMM, University of Helsinki, Helsinki, Finland. 10 Email: [email protected] 11 Financial disclosure: This work was funded by the Academy of Finland (grants 269862, 292611, 12 310507 and 313267), Cancer Society of Finland, and the Sigrid Juselius Foundation. 13 Ethical disclosure: This study is an independent analysis of existing data available in the public 14 domain and does not involve any animal or human samples that have been collected by the authors 15 themselves. 16 Author contribution: AKG conceptualized, analyzed the data and wrote the manuscript. TA 17 critically revised and edited the manuscript. The authors report no conflict of interest. 18 19 Word count: 20 Figure number: 5 21 Table number: 1 22 23 Abstract 24 Background: DNA methyltransferase inhibitors (DNMTi) decitabine and azacytidine are approved 25 therapies for acute myeloid leukemia and myelodysplastic syndrome. -

Differential Gene Expression Profiling of Dystrophic Dog
Differential Gene Expression Profiling of Dystrophic Dog Muscle after MuStem Cell Transplantation Florence Robriquet, Aurélie Lardenois, Candice Babarit, Thibaut Larcher, Laurence Dubreil, Isabelle Leroux, Céline Zuber, Mireille Ledevin, Jack-Yves Deschamps, Yves Fromes, et al. To cite this version: Florence Robriquet, Aurélie Lardenois, Candice Babarit, Thibaut Larcher, Laurence Dubreil, et al.. Differential Gene Expression Profiling of Dystrophic Dog Muscle after MuStem Cell Transplantation. PLoS ONE, Public Library of Science, 2015, 10 (5), 10.1371/journal.pone.0123336. hal-01222898 HAL Id: hal-01222898 https://hal.archives-ouvertes.fr/hal-01222898 Submitted on 30 Oct 2015 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Distributed under a Creative Commons Attribution| 4.0 International License RESEARCH ARTICLE Differential Gene Expression Profiling of Dystrophic Dog Muscle after MuStem Cell Transplantation Florence Robriquet1,2,3☯, Aurélie Lardenois1,2☯, Candice Babarit1,2, Thibaut Larcher1,2, Laurence Dubreil1,2, Isabelle Leroux1,2, Céline Zuber1,2, Mireille Ledevin1,2, Jack- Yves Deschamps1,2, Yves Fromes2,4, Yan Cherel1,2, Laetitia Guevel1,2,3‡*, Karl Rouger1,2‡ 1 INRA, UMR703 PAnTher, Nantes, France, 2 LUNAM Université, Oniris, École nationale vétérinaire, agro- alimentaire et de l’alimentation Nantes-Atlantique, Nantes, France, 3 Université de Nantes, Nantes, France, 4 Laboratoire RMN AIM-CEA, Institut de Myologie, Hôpital Pitié-Salpêtrière, Paris, France ☯ These authors contributed equally to this work. -

Molecular Targeting and Enhancing Anticancer Efficacy of Oncolytic HSV-1 to Midkine Expressing Tumors
University of Cincinnati Date: 12/20/2010 I, Arturo R Maldonado , hereby submit this original work as part of the requirements for the degree of Doctor of Philosophy in Developmental Biology. It is entitled: Molecular Targeting and Enhancing Anticancer Efficacy of Oncolytic HSV-1 to Midkine Expressing Tumors Student's name: Arturo R Maldonado This work and its defense approved by: Committee chair: Jeffrey Whitsett Committee member: Timothy Crombleholme, MD Committee member: Dan Wiginton, PhD Committee member: Rhonda Cardin, PhD Committee member: Tim Cripe 1297 Last Printed:1/11/2011 Document Of Defense Form Molecular Targeting and Enhancing Anticancer Efficacy of Oncolytic HSV-1 to Midkine Expressing Tumors A dissertation submitted to the Graduate School of the University of Cincinnati College of Medicine in partial fulfillment of the requirements for the degree of DOCTORATE OF PHILOSOPHY (PH.D.) in the Division of Molecular & Developmental Biology 2010 By Arturo Rafael Maldonado B.A., University of Miami, Coral Gables, Florida June 1993 M.D., New Jersey Medical School, Newark, New Jersey June 1999 Committee Chair: Jeffrey A. Whitsett, M.D. Advisor: Timothy M. Crombleholme, M.D. Timothy P. Cripe, M.D. Ph.D. Dan Wiginton, Ph.D. Rhonda D. Cardin, Ph.D. ABSTRACT Since 1999, cancer has surpassed heart disease as the number one cause of death in the US for people under the age of 85. Malignant Peripheral Nerve Sheath Tumor (MPNST), a common malignancy in patients with Neurofibromatosis, and colorectal cancer are midkine- producing tumors with high mortality rates. In vitro and preclinical xenograft models of MPNST were utilized in this dissertation to study the role of midkine (MDK), a tumor-specific gene over- expressed in these tumors and to test the efficacy of a MDK-transcriptionally targeted oncolytic HSV-1 (oHSV). -

UC San Diego Electronic Theses and Dissertations
UC San Diego UC San Diego Electronic Theses and Dissertations Title Cardiac Stretch-Induced Transcriptomic Changes are Axis-Dependent Permalink https://escholarship.org/uc/item/7m04f0b0 Author Buchholz, Kyle Stephen Publication Date 2016 Peer reviewed|Thesis/dissertation eScholarship.org Powered by the California Digital Library University of California UNIVERSITY OF CALIFORNIA, SAN DIEGO Cardiac Stretch-Induced Transcriptomic Changes are Axis-Dependent A dissertation submitted in partial satisfaction of the requirements for the degree Doctor of Philosophy in Bioengineering by Kyle Stephen Buchholz Committee in Charge: Professor Jeffrey Omens, Chair Professor Andrew McCulloch, Co-Chair Professor Ju Chen Professor Karen Christman Professor Robert Ross Professor Alexander Zambon 2016 Copyright Kyle Stephen Buchholz, 2016 All rights reserved Signature Page The Dissertation of Kyle Stephen Buchholz is approved and it is acceptable in quality and form for publication on microfilm and electronically: Co-Chair Chair University of California, San Diego 2016 iii Dedication To my beautiful wife, Rhia. iv Table of Contents Signature Page ................................................................................................................... iii Dedication .......................................................................................................................... iv Table of Contents ................................................................................................................ v List of Figures ................................................................................................................... -

Integrative Analysis of Next Generation Sequencing for Small Non-Coding
Beck et al. BMC Medical Genomics 2011, 4:19 http://www.biomedcentral.com/1755-8794/4/19 RESEARCHARTICLE Open Access Integrative analysis of next generation sequencing for small non-coding RNAs and transcriptional regulation in Myelodysplastic Syndromes Dominik Beck1,2, Steve Ayers4, Jianguo Wen3, Miriam B Brandl1,2, Tuan D Pham1, Paul Webb4, Chung-Che Chang3*, Xiaobo Zhou1* Abstract Background: Myelodysplastic Syndromes (MDSS) are pre-leukemic disorders with increasing incident rates worldwide, but very limited treatment options. Little is known about small regulatory RNAs and how they contribute to pathogenesis, progression and transcriptome changes in MDS. Methods: Patients’ primary marrow cells were screened for short RNAs (RNA-seq) using next generation sequencing. Exon arrays from the same cells were used to profile gene expression and additional measures on 98 patients obtained. Integrative bioinformatics algorithms were proposed, and pathway and ontology analysis performed. Results: In low-grade MDS, observations implied extensive post-transcriptional regulation via microRNAs (miRNA) and the recently discovered Piwi interacting RNAs (piRNA). Large expression differences were found for MDS- associated and novel miRNAs, including 48 sequences matching to miRNA star (miRNA*) motifs. The detected species were predicted to regulate disease stage specific molecular functions and pathways, including apoptosis and response to DNA damage. In high-grade MDS, results suggested extensive post-translation editing via transfer RNAs (tRNAs), providing a potential link for reduced apoptosis, a hallmark for this disease stage. Bioinformatics analysis confirmed important regulatory roles for MDS linked miRNAs and TFs, and strengthened the biological significance of miRNA*. The “RNA polymerase II promoters” were identified as the tightest controlled biological function. -
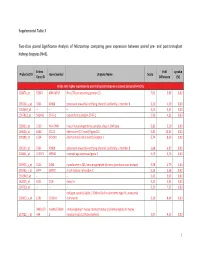
Supplemental Table 3 Two-Class Paired Significance Analysis of Microarrays Comparing Gene Expression Between Paired
Supplemental Table 3 Two‐class paired Significance Analysis of Microarrays comparing gene expression between paired pre‐ and post‐transplant kidneys biopsies (N=8). Entrez Fold q‐value Probe Set ID Gene Symbol Unigene Name Score Gene ID Difference (%) Probe sets higher expressed in post‐transplant biopsies in paired analysis (N=1871) 218870_at 55843 ARHGAP15 Rho GTPase activating protein 15 7,01 3,99 0,00 205304_s_at 3764 KCNJ8 potassium inwardly‐rectifying channel, subfamily J, member 8 6,30 4,50 0,00 1563649_at ‐‐ ‐‐ ‐‐ 6,24 3,51 0,00 1567913_at 541466 CT45‐1 cancer/testis antigen CT45‐1 5,90 4,21 0,00 203932_at 3109 HLA‐DMB major histocompatibility complex, class II, DM beta 5,83 3,20 0,00 204606_at 6366 CCL21 chemokine (C‐C motif) ligand 21 5,82 10,42 0,00 205898_at 1524 CX3CR1 chemokine (C‐X3‐C motif) receptor 1 5,74 8,50 0,00 205303_at 3764 KCNJ8 potassium inwardly‐rectifying channel, subfamily J, member 8 5,68 6,87 0,00 226841_at 219972 MPEG1 macrophage expressed gene 1 5,59 3,76 0,00 203923_s_at 1536 CYBB cytochrome b‐245, beta polypeptide (chronic granulomatous disease) 5,58 4,70 0,00 210135_s_at 6474 SHOX2 short stature homeobox 2 5,53 5,58 0,00 1562642_at ‐‐ ‐‐ ‐‐ 5,42 5,03 0,00 242605_at 1634 DCN decorin 5,23 3,92 0,00 228750_at ‐‐ ‐‐ ‐‐ 5,21 7,22 0,00 collagen, type III, alpha 1 (Ehlers‐Danlos syndrome type IV, autosomal 201852_x_at 1281 COL3A1 dominant) 5,10 8,46 0,00 3493///3 IGHA1///IGHA immunoglobulin heavy constant alpha 1///immunoglobulin heavy 217022_s_at 494 2 constant alpha 2 (A2m marker) 5,07 9,53 0,00 1 202311_s_at -

Computational Studies of the Genome Dynamics of Mammalian Transposable Elements and Their Relationships to Genes
COMPUTATIONAL STUDIES OF THE GENOME DYNAMICS OF MAMMALIAN TRANSPOSABLE ELEMENTS AND THEIR RELATIONSHIPS TO GENES by Ying Zhang M.Sc., Katholieke Universiteit Leuven (BELGIUM), 2004 B.E., Harbin Institute of Technology (CHINA), 1993 A THESIS SUBMITTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY in THE FACULTY OF GRADUATE STUDIES (Genetics) THE UNIVERSITY OF BRITISH COLUMBIA (Vancouver) May, 2012 © Ying Zhang, 2012 Abstract Sequences derived from transposable elements (TEs) comprise nearly 40 - 50% of the genomic DNA of most mammalian species, including mouse and human. However, what impact they may exert on their hosts is an intriguing question. Originally considered as merely genomic parasites or “selfish DNA”, these mobile elements show their detrimental effects through a variety of mechanisms, from physical DNA disruption to epigenetic regulation. On the other hand, evidence has been mounting to suggest that TEs sometimes may also play important roles by participating in essential biological processes in the host cell. The dual-roles of TE-host interactions make it critical for us to understand the relationship between TEs and the host, which may ultimately help us to better understand both normal cellular functions and disease. This thesis encompasses my three genome-wide computational studies of TE-gene dynamics in mammals. In the first, I identified high levels of TE insertional polymorphisms among inbred mouse strains, and systematically analyzed their distributional features and biological effects, through mining tens of millions of mouse genomic DNA sequences. In the second, I examined the properties of TEs located in introns, and identified key factors, such as the distance to the intron-exon boundary, insertional orientation, and proximity to splice sites, that influence the probability that TEs will be retained in genes. -
Identification and Interaction Analysis of Key Genes and Micrornas In
Mou et al. World Journal of Surgical Oncology (2017) 15:63 DOI 10.1186/s12957-017-1127-2 RESEARCH Open Access Identification and interaction analysis of key genes and microRNAs in hepatocellular carcinoma by bioinformatics analysis Tong Mou†, Di Zhu†, Xufu Wei, Tingting Li, Daofeng Zheng, Junliang Pu, Zhen Guo and Zhongjun Wu* Abstract Background: Hepatocellular carcinoma (HCC) is the most common liver malignancy worldwide. However, present studies of its multiple gene interaction and cellular pathways still could not explain the initiation and development of HCC perfectly. To find the key genes and miRNAs as well as their potential molecular mechanisms in HCC, microarray data GSE22058, GSE25097, and GSE57958 were analyzed. Methods: The microarray datasets GSE22058, GSE25097, and GSE57958, including mRNA and miRNA profiles, were downloaded from the GEO database and were analyzed using GEO2R. Functional and pathway enrichment analyses were performed using the DAVID database, and the protein–protein interaction (PPI) network was constructed using the Cytoscape software. Finally, miRDB was applied to predict the targets of the differentially expressed miRNAs (DEMs). Results: A total of 115 differentially expressed genes (DEGs) were found in HCC, including 52 up-regulated genes and 63 down-regulated genes. The gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses from DAVID showed that up-regulated genes were significantly enriched in chromosome segregation and cell division, while the down-regulated genes were mainly involved in complement activation, protein activation cascades, carboxylic acid metabolic processes, oxoacid metabolic processes, and the immune response. From the PPI network, the 18 nodes with the highest degree were screened as hub genes. -

Somatic Polyploidy Is Associated with the Upregulation of C-MYC Interacting Genes and EMT-Like Signature
www.impactjournals.com/oncotarget/ Oncotarget, Vol. 7, No. 46 Research Paper Somatic polyploidy is associated with the upregulation of c-MYC interacting genes and EMT-like signature Alejandro Vazquez-Martin1,*, Olga V. Anatskaya2,*, Alessandro Giuliani3, Jekaterina Erenpreisa1,**, Sui Huang4, Kristine Salmina1, Inna Inashkina1, Anda Huna1, Nikolai N. Nikolsky2, Alexander E. Vinogradov2,** 1Latvian Biomedical Research and Study Centre, Riga, Latvia 2Institute of Cytology, St-Petersburg, Russian Federation, Russia 3Istituto Superiore di Sanità, Rome, Italy 4Systems Biology Institute, Seattle, USA *Equal contribution **Senior authors Correspondence to: Olga V. Anatskaya, email: [email protected] Keywords: c-MYC interacting genes, polyploidy, Warburg, stress, EMT Received: June 03, 2016 Accepted: September 05, 2016 Published: September 19, 2016 ABSTRACT The dependence of cancer on overexpressed c-MYC and its predisposition for polyploidy represents a double puzzle. We address this conundrum by cross- species transcription analysis of c-MYC interacting genes in polyploid vs. diploid tissues and cells, including human vs. mouse heart, mouse vs. human liver and purified 4n vs. 2n mouse decidua cells. Gene-by-gene transcriptome comparison and principal component analysis indicated that c-MYC interactants are significantly overrepresented among ploidy-associated genes. Protein interaction networks and gene module analysis revealed that the most upregulated genes relate to growth, stress response, proliferation, stemness and unicellularity, as well as to the pathways of cancer supported by MAPK and RAS coordinated pathways. A surprising feature was the up-regulation of epithelial-mesenchymal transition (EMT) modules embodied by the N-cadherin pathway and EMT regulators from SNAIL and TWIST families. Metabolic pathway analysis also revealed the EMT-linked features, such as global proteome remodeling, oxidative stress, DNA repair and Warburg-like energy metabolism. -

(M) of SARS-Cov-2
Endomembrane systems are reorganized by ORF3a and Membrane (M) of SARS-CoV-2 Yun-Bin Lee1, Minkyo Jung2, Jeesoo Kim3, Myeong-Gyun Kang1, Chulhwan Kwak1,5, Jong-Seo Kim3,4,*, Ji- Young Mun2,*, Hyun-Woo Rhee1,4,* 1Department of Chemistry, Seoul National University, Seoul 08826, Republic of Korea 2Neural Circuit Research Group, Korea Brain Research Institute, 41062 Daegu, Republic of Korea 3Center for RNA research, Institute for Basic Science, Seoul 08826, Republic of Korea 4School of Biological Sciences, Seoul National University, Seoul 08826, Republic of Korea 5Department of Chemistry, Ulsan National Institute of Science and Technology, 44919 Ulsan, Korea *Correspondence: [email protected], [email protected], [email protected]. Table of Contents Supplementary Figure 1-15-----------------------------------------------------------------------------------------S2-24 Supplementary Table 1-3------------------------------------------------------------------------------------------S25-29 -S1- A GFP V5 BF Merged + Pearson R V5 - = 0.64 GFP GFP - SEC61B ORF3a Pearson R = 0.61 Pearson R + V5 - = 0.57 GFP GFP - M SEC61B Pearson R = 0.64 B GFP Flag BF Merged + + Pearson R = 0.87 Flag - GFP GFP - ORF3a ORF3a Pearson R = 0.80 + Pearson R = 0.67 Flag - GFP GFP - ORF6 ORF6 Pearson R = 0.64 + Pearson R Flag = 0.91 - GFP GFP - ORF7b ORF7b Pearson R = 0.90 + Pearson R = 0.91 Flag - GFP GFP - M M Pearson R = 0.89 Supplementary Figure 1. Additional confocal microscopy images of GFP-tagged vPOIs of SARS-CoV-2 (a) Confocal microscopy images of ORF3a-GFP (left) and M-GFP (right) with SEC61B-V5-TurboID. SEC61B-V5- TurboID was visualized by Anti-V5 antibody (AF647-conjugated secondary antibody, Cy5 fluorescence channel).