Product Data Sheet
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

PEMD-91-12BR Off-Label Drugs: Initial Results of a National Survey
11 1; -- __...._-----. ^.-- ______ -..._._ _.__ - _........ - t Ji Jo United States General Accounting Office Washington, D.C. 20648 Program Evaluation and Methodology Division B-242851 February 25,199l The Honorable Edward M. Kennedy Chairman, Committee on Labor and Human Resources United States Senate Dear Mr. Chairman: In September 1989, you asked us to conduct a study on reimbursement denials by health insurers for off-label drug use. As you know, the Food and Drug Administration designates the specific clinical indications for which a drug has been proven effective on a label insert for each approved drug, “Off-label” drug use occurs when physicians prescribe a drug for clinical indications other than those listed on the label. In response to your request, we surveyed a nationally representative sample of oncologists to determine: . the prevalence of off-label use of anticancer drugs by oncologists and how use varies by clinical, demographic, and geographic factors; l the extent to which third-party payers (for example, Medicare intermediaries, private health insurers) are denying payment for such use; and l whether the policies of third-party payers are influencing the treatment of cancer patients. We randomly selected 1,470 members of the American Society of Clinical Oncologists and sent them our survey in March 1990. The sam- pling was structured to ensure that our results would be generalizable both to the nation and to the 11 states with the largest number of oncologists. Our response rate was 56 percent, and a comparison of respondents to nonrespondents shows no noteworthy differences between the two groups. -

Title 16. Crimes and Offenses Chapter 13. Controlled Substances Article 1
TITLE 16. CRIMES AND OFFENSES CHAPTER 13. CONTROLLED SUBSTANCES ARTICLE 1. GENERAL PROVISIONS § 16-13-1. Drug related objects (a) As used in this Code section, the term: (1) "Controlled substance" shall have the same meaning as defined in Article 2 of this chapter, relating to controlled substances. For the purposes of this Code section, the term "controlled substance" shall include marijuana as defined by paragraph (16) of Code Section 16-13-21. (2) "Dangerous drug" shall have the same meaning as defined in Article 3 of this chapter, relating to dangerous drugs. (3) "Drug related object" means any machine, instrument, tool, equipment, contrivance, or device which an average person would reasonably conclude is intended to be used for one or more of the following purposes: (A) To introduce into the human body any dangerous drug or controlled substance under circumstances in violation of the laws of this state; (B) To enhance the effect on the human body of any dangerous drug or controlled substance under circumstances in violation of the laws of this state; (C) To conceal any quantity of any dangerous drug or controlled substance under circumstances in violation of the laws of this state; or (D) To test the strength, effectiveness, or purity of any dangerous drug or controlled substance under circumstances in violation of the laws of this state. (4) "Knowingly" means having general knowledge that a machine, instrument, tool, item of equipment, contrivance, or device is a drug related object or having reasonable grounds to believe that any such object is or may, to an average person, appear to be a drug related object. -

Hormone Replacement Therapy and Osteoporosis
This report may be used, in whole or in part, as the basis for development of clinical practice guidelines and other quality enhancement tools, or a basis for reimbursement and coverage policies. AHRQ or U.S. Department of Health and Human Services endorsement of such derivative products may not be stated or implied. AHRQ is the lead Federal agency charged with supporting research designed to improve the quality of health care, reduce its cost, address patient safety and medical errors, and broaden access to essential services. AHRQ sponsors and conducts research that provides evidence-based information on health care outcomes; quality; and cost, use, and access. The information helps health care decisionmakers— patients and clinicians, health system leaders, and policymakers—make more informed decisions and improve the quality of health care services. Systematic Evidence Review Number 12 Hormone Replacement Therapy and Osteoporosis Prepared for: Agency for Healthcare Research and Quality U.S. Department of Health and Human Services 2101 East Jefferson Street Rockville, MD 20852 http://www.ahrq.gov Contract No. 290-97-0018 Task Order No. 2 Technical Support of the U.S. Preventive Services Task Force Prepared by: Oregon Health Sciences University Evidence-based Practice Center, Portland, Oregon Heidi D. Nelson, MD, MPH August 2002 Preface The Agency for Healthcare Research and Quality (AHRQ) sponsors the development of Systematic Evidence Reviews (SERs) through its Evidence-based Practice Program. With guidance from the third U.S. Preventive Services Task Force∗ (USPSTF) and input from Federal partners and primary care specialty societies, two Evidence-based Practice Centers—one at the Oregon Health Sciences University and the other at Research Triangle Institute-University of North Carolina—systematically review the evidence of the effectiveness of a wide range of clinical preventive services, including screening, counseling, immunizations, and chemoprevention, in the primary care setting. -

Pharmacology/Therapeutics II Block III Lectures 2013-14
Pharmacology/Therapeutics II Block III Lectures 2013‐14 66. Hypothalamic/pituitary Hormones ‐ Rana 67. Estrogens and Progesterone I ‐ Rana 68. Estrogens and Progesterone II ‐ Rana 69. Androgens ‐ Rana 70. Thyroid/Anti‐Thyroid Drugs – Patel 71. Calcium Metabolism – Patel 72. Adrenocorticosterioids and Antagonists – Clipstone 73. Diabetes Drugs I – Clipstone 74. Diabetes Drugs II ‐ Clipstone Pharmacology & Therapeutics Neuroendocrine Pharmacology: Hypothalamic and Pituitary Hormones, March 20, 2014 Lecture Ajay Rana, Ph.D. Neuroendocrine Pharmacology: Hypothalamic and Pituitary Hormones Date: Thursday, March 20, 2014-8:30 AM Reading Assignment: Katzung, Chapter 37 Key Concepts and Learning Objectives To review the physiology of neuroendocrine regulation To discuss the use neuroendocrine agents for the treatment of representative neuroendocrine disorders: growth hormone deficiency/excess, infertility, hyperprolactinemia Drugs discussed Growth Hormone Deficiency: . Recombinant hGH . Synthetic GHRH, Recombinant IGF-1 Growth Hormone Excess: . Somatostatin analogue . GH receptor antagonist . Dopamine receptor agonist Infertility and other endocrine related disorders: . Human menopausal and recombinant gonadotropins . GnRH agonists as activators . GnRH agonists as inhibitors . GnRH receptor antagonists Hyperprolactinemia: . Dopamine receptor agonists 1 Pharmacology & Therapeutics Neuroendocrine Pharmacology: Hypothalamic and Pituitary Hormones, March 20, 2014 Lecture Ajay Rana, Ph.D. 1. Overview of Neuroendocrine Systems The neuroendocrine -

A Pharmaceutical Product for Hormone Replacement Therapy Comprising Tibolone Or a Derivative Thereof and Estradiol Or a Derivative Thereof
Europäisches Patentamt *EP001522306A1* (19) European Patent Office Office européen des brevets (11) EP 1 522 306 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: (51) Int Cl.7: A61K 31/567, A61K 31/565, 13.04.2005 Bulletin 2005/15 A61P 15/12 (21) Application number: 03103726.0 (22) Date of filing: 08.10.2003 (84) Designated Contracting States: • Perez, Francisco AT BE BG CH CY CZ DE DK EE ES FI FR GB GR 08970 Sant Joan Despi (Barcelona) (ES) HU IE IT LI LU MC NL PT RO SE SI SK TR • Banado M., Carlos Designated Extension States: 28033 Madrid (ES) AL LT LV MK (74) Representative: Markvardsen, Peter et al (71) Applicant: Liconsa, Liberacion Controlada de Markvardsen Patents, Sustancias Activas, S.A. Patent Department, 08028 Barcelona (ES) P.O. Box 114, Favrholmvaenget 40 (72) Inventors: 3400 Hilleroed (DK) • Palacios, Santiago 28001 Madrid (ES) (54) A pharmaceutical product for hormone replacement therapy comprising tibolone or a derivative thereof and estradiol or a derivative thereof (57) A pharmaceutical product comprising an effec- arate or sequential use in a method for hormone re- tive amount of tibolone or derivative thereof, an effective placement therapy or prevention of hypoestrogenism amount of estradiol or derivative thereof and a pharma- associated clinical symptoms in a human person, in par- ceutically acceptable carrier, wherein the product is pro- ticular wherein the human is a postmenopausal woman. vided as a combined preparation for simultaneous, sep- EP 1 522 306 A1 Printed by Jouve, 75001 PARIS (FR) 1 EP 1 522 306 A1 2 Description [0008] The review article of Journal of Steroid Bio- chemistry and Molecular Biology (2001), 76(1-5), FIELD OF THE INVENTION: 231-238 provides a review of some of these compara- tive studies. -

Steroidal Estrogens
FINAL Report on Carcinogens Background Document for Steroidal Estrogens December 13 - 14, 2000 Meeting of the NTP Board of Scientific Counselors Report on Carcinogens Subcommittee Prepared for the: U.S. Department of Health and Human Services Public Health Service National Toxicology Program Research Triangle Park, NC 27709 Prepared by: Technology Planning and Management Corporation Canterbury Hall, Suite 310 4815 Emperor Blvd Durham, NC 27703 Contract Number N01-ES-85421 Dec. 2000 RoC Background Document for Steroidal Estrogens Do not quote or cite Criteria for Listing Agents, Substances or Mixtures in the Report on Carcinogens U.S. Department of Health and Human Services National Toxicology Program Known to be Human Carcinogens: There is sufficient evidence of carcinogenicity from studies in humans, which indicates a causal relationship between exposure to the agent, substance or mixture and human cancer. Reasonably Anticipated to be Human Carcinogens: There is limited evidence of carcinogenicity from studies in humans which indicates that causal interpretation is credible but that alternative explanations such as chance, bias or confounding factors could not adequately be excluded; or There is sufficient evidence of carcinogenicity from studies in experimental animals which indicates there is an increased incidence of malignant and/or a combination of malignant and benign tumors: (1) in multiple species, or at multiple tissue sites, or (2) by multiple routes of exposure, or (3) to an unusual degree with regard to incidence, site or type of tumor or age at onset; or There is less than sufficient evidence of carcinogenicity in humans or laboratory animals, however; the agent, substance or mixture belongs to a well defined, structurally-related class of substances whose members are listed in a previous Report on Carcinogens as either a known to be human carcinogen, or reasonably anticipated to be human carcinogen or there is convincing relevant information that the agent acts through mechanisms indicating it would likely cause cancer in humans. -

Appendix E: Hormonal Replacement Therapy Regimens E
Appendix E: Hormonal Replacement Therapy Regimens E he proportion of post-menopausal women 14 days and a three-day drug-free period) or in who use hormone replacement therapy continuous combination with estrogen. These (HRT) has increased in the United States progestin/estrogen therapies (PERTs) alter the during the last two decades (13). While benefit-risk profile of HRT. Tuse of HRT has increased, the average dose and In the United States, conjugated equine estro- duration of use of postmenopausal estrogens has gen (CEE) (Premarin, Wyeth-Ayerst) is the most decreased until recently (64). This is due in part to commonly used form of estrogen for HRT.1,2 the discovery that the cancer-causing effects of There are a number of other estrogens used for postmenopausal estrogen are related to its dose HRT.3 Table E-1 lists the estrogens either ap- and duration. proved for osteoporosis by the Food and Drug Ad- This appendix describes the range of choice re- ministration or accepted for this use by a garding the dose, routes of administration, and committee of the United States Pharmacopoeia. In combinations of hormones currently in use or un- addition to the estrogens listed in the table, the es- der study as treatment for postmenopausal HRT. trogens quinestrol (Estrovis tablets, Parke-Davis) This appendix also describes the appropriate fol- and chlorotrianisene (TACE capsules, Marion low-up of women on HRT. Finally, this appendix Merrel Dow) are approved by the FDA for treat- describes how dosing regimens may affect com- ment of menopausal symptoms (61). pliance with HRT and describes other factors that affect HRT compliance. -
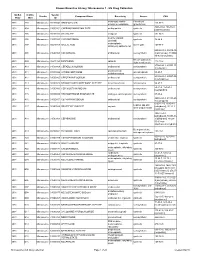
Known Bioactive Library: Microsource 1 - US Drug Collection
Known Bioactive Library: Microsource 1 - US Drug Collection ICCB-L ICCB-L Vendor Vendor Compound Name Bioactivity Source CAS Plate Well ID antifungal, inhibits Penicillium 2091 A03 Microsource 00200046 GRISEOFULVIN 126-07-8 mitosis in metaphase griseofulvum 3505-38-2, 486-16-8 2091 A04 Microsource 01500161 CARBINOXAMINE MALEATE antihistaminic synthetic [carbinoxamine] 2091 A05 Microsource 00200331 SALSALATE analgesic synthetic 552-94-3 muscle relaxant 2091 A06 Microsource 01500162 CARISOPRODOL synthetic 78-44-4 (skeletal) antineoplastic, 2091 A07 Microsource 00210369 GALLIC ACID insect galls 149-91-7 astringent, antibacterial 66592-87-8, 50370-12- 2091 A08 Microsource 01500163 CEFADROXIL antibacterial semisynthetic 2 [anhydrous], 119922- 89-9 [hemihydrate] Rheum palmatum, 2091 A09 Microsource 00211468 DANTHRON cathartic 117-10-2 Xyris semifuscata 27164-46-1, 25953-19- 2091 A10 Microsource 01500164 CEFAZOLIN SODIUM antibacterial semisynthetic 9 [cefazolin] glucocorticoid, 2091 A11 Microsource 00300024 HYDROCORTISONE adrenal glands 50-23-7 antiinflammatory 64485-93-4, 63527-52- 2091 A12 Microsource 01500165 CEFOTAXIME SODIUM antibacterial semisynthetic 6 [cefotaxime] 2091 A13 Microsource 00300029 DESOXYCORTICOSTERONE ACETATE mineralocorticoid adrenocortex 56-47-3 58-71-9, 153-61-7 2091 A14 Microsource 01500166 CEPHALOTHIN SODIUM antibacterial semisynthetic [cephalothin] 2091 A15 Microsource 00300034 TESTOSTERONE PROPIONATE androgen, antineoplastic semisynthetic 57-85-2 24356-60-3, 21593-23- 2091 A16 Microsource 01500167 CEPHAPIRIN SODIUM -

Cumulative Cross Index to Iarc Monographs
PERSONAL HABITS AND INDOOR COMBUSTIONS volume 100 e A review of humAn cArcinogens This publication represents the views and expert opinions of an IARC Working Group on the Evaluation of Carcinogenic Risks to Humans, which met in Lyon, 29 September-6 October 2009 LYON, FRANCE - 2012 iArc monogrAphs on the evAluAtion of cArcinogenic risks to humAns CUMULATIVE CROSS INDEX TO IARC MONOGRAPHS The volume, page and year of publication are given. References to corrigenda are given in parentheses. A A-α-C .............................................................40, 245 (1986); Suppl. 7, 56 (1987) Acenaphthene ........................................................................92, 35 (2010) Acepyrene ............................................................................92, 35 (2010) Acetaldehyde ........................36, 101 (1985) (corr. 42, 263); Suppl. 7, 77 (1987); 71, 319 (1999) Acetaldehyde associated with the consumption of alcoholic beverages ..............100E, 377 (2012) Acetaldehyde formylmethylhydrazone (see Gyromitrin) Acetamide .................................... 7, 197 (1974); Suppl. 7, 56, 389 (1987); 71, 1211 (1999) Acetaminophen (see Paracetamol) Aciclovir ..............................................................................76, 47 (2000) Acid mists (see Sulfuric acid and other strong inorganic acids, occupational exposures to mists and vapours from) Acridine orange ...................................................16, 145 (1978); Suppl. 7, 56 (1987) Acriflavinium chloride ..............................................13, -

Federal Register/Vol. 68, No. 71/Monday, April 14, 2003/Notices
Federal Register / Vol. 68, No. 71 / Monday, April 14, 2003 / Notices 17953 CFR Number of Responses per Avg burden per Total annual reference Data collection respondents respondent response (in hrs.) burden (in hrs.) 73.6 (c–e) Request for exemption .............................. 17 1 70/60 20 73.14 ........ Transfer of select agent ............................ 1,000 5 1.75 8,750 73.6 (a)(2) Clinical and diagnostic laboratory exemp- 1,000 4 1 4,000 tion report. 73.7(i) ....... Notification of inactivation .......................... 6 1 30/60 3 73.8(g) ..... Request expedited review ......................... 6 1 30/60 3 73.10(b) ... Documentation of self-inspection .............. 1,000 1 1 1,000 73.13(f) .... Documentation of training ......................... 1,000 1 2 8,700 73.18 ........ Administrative review ................................. 14 1 4 56 73.15(d) ... Ensure secure recordkeeping system ....... 1,000 1 30/60 4,000 Total .. .................................................................... 1,000 ................................ ................................ 34,804 Dated: April 7, 2003. and other comments should be patients not improved by estrogen Thomas Bartenfeld, identified with Docket No. 76N–0377 alone.’’ Acting Associate Director for Policy, Planning and submitted to the Dockets In the Federal Register of December and Evaluation, Centers for Disease Control Management Branch (HFA–305), Food 17, 1998 (63 FR 69631), FDA withdrew and Prevention. and Drug Administration, 5630 Fishers approval of estrogen-containing drugs [FR Doc. 03–9019 Filed 4–11–03; 8:45 am] Lane, rm. 1061, Rockville, MD 20852. A insofar as they are indicated for BILLING CODE 4163–18–P request for an opinion on the postpartum breast engorgement because applicability of this notice to a specific estrogens have not been shown to be drug product should be directed to the safe for this use. -

2012; Nᵒ 17, 91-95 ISSN 0976 – 044X
Int. J. Pharm. Sci. Rev. Res., 16(1), 2012; nᵒ 17, 91-95 ISSN 0976 – 044X Research Article COMPARATIVE DOCKING STUDIES OF ESTROGEN RECEPTOR INHIBITORS AND THEIR BINDING INTERACTION ANALYSIS Nidhi Desai1*, Manoj Kumar Mahto2,5, B.Alekhya3, C.R.Naveen4, Prof. M. Bhaskar5 1. Dept. of Biotechnology, PESIT, Bangalore University, Bangalore, Karnataka, India. 2. Dept. of Biotechnology, Acharya Nagarjuna University, Guntur, AP, India. 3. Dept. of Biotechnology, MGR Arts and Science College, Periyar University, Hosur, TN, India. 4. Dept. of Medical Biochemistry, SRM Hospital College and Research Centre, TN, India. 5. Division of Animal Biotechnology, Dept. of Zoology, Sri Venkateswara University, Tirupati, AP, India. *Corresponding author’s E-mail: [email protected] Accepted on: 08-07-2012; Finalized on: 31-08-2012. ABSTRACT Comparative docking studies have been performed on 10 drug molecules, which play a vital role in the treatment of breast cancer. These 10 drug molecules have the same target called Estrogen Receptor which acts as a DNA-binding transcription factor that regulates gene expression. The glide scores of these 10 drugs were compared to each other using the module Glide which is a part of the Schrödinger software, which provided a better understanding of the binding interactions of the ten drug molecules. Among the 10 drugs taken, toremifene had the lowest glide score, i.e. -9.82, which shows that it had better binding interaction with the protein. Keywords: Breast Cancer, Estrogen Receptor, Molecular Docking, Glide, Binding Interactions, Schrödinger 2009. INTRODUCTION found in kidney, brain, bone, prostate, heart, lungs, and endothelial cells6. The binding and the functional Breast cancer is a cancer which originates in the tissues of selectivity of the ER helix with 12 domains play a vital role the breast, mostly from the inner lining of the lobules or in the determination of the binding interactions with the milk ducts which supply milk. -

PRAC Recommendations on Signals Adopted at the 13-16 January 2020 PRAC En
10 February 20201 EMA/PRAC/8637/2020 Pharmacovigilance Risk Assessment Committee (PRAC) PRAC recommendations on signals Adopted at the 13-16 January 2020 PRAC meeting This document provides an overview of the recommendations adopted by the Pharmacovigilance Risk Assessment Committee (PRAC) on the signals discussed during the meeting of 13-16 January 2020 (including the signal European Pharmacovigilance Issues Tracking Tool [EPITT]2 reference numbers). PRAC recommendations to provide supplementary information are directly actionable by the concerned marketing authorisation holders (MAHs). PRAC recommendations for regulatory action (e.g. amendment of the product information) are submitted to the Committee for Medicinal Products for Human Use (CHMP) for endorsement when the signal concerns Centrally Authorised Products (CAPs), and to the Co-ordination Group for Mutual Recognition and Decentralised Procedures – Human (CMDh) for information in the case of Nationally Authorised Products (NAPs). Thereafter, MAHs are expected to take action according to the PRAC recommendations. When appropriate, the PRAC may also recommend the conduct of additional analyses by the Agency or Member States. MAHs are reminded that in line with Article 16(3) of Regulation No (EU) 726/2004 and Article 23(3) of Directive 2001/83/EC, they shall ensure that their product information is kept up to date with the current scientific knowledge including the conclusions of the assessment and recommendations published on the European Medicines Agency (EMA) website (currently acting as the EU medicines webportal). For CAPs, at the time of publication, PRAC recommendations for update of product information have been agreed by the CHMP at their plenary meeting (27-30 January 2020) and corresponding variations will be assessed by the CHMP.