Additional Menopause Learning Issues And
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

United States July 2016 2 Table of Contents
Deuterium Labelled Compounds United States July 2016 2 Table of Contents International Distributors 3 Corporate Overview 4 General Information 5 Pricing and Payment 5 Quotations 5 Custom Synthesis 5 Shipping 5 Quality Control 6 Quotations 6 Custom Synthesis 6 Shipping 6 Quality Control 6 Chemical Abstract Service Numbers 6 Handling Hazardous Compounds 6 Our Products are Not Intended for Use in Humans 7 Limited Warranty 7 Packaging Information 7 Alphabetical Listings 8 Stock Clearance 236 Products by Category 242 n-Alkanes 243 α-Amino Acids, N-Acyl α-Amino Acids, N-t-BOC Protected α-Amino Acid 243 and N-FMOC Protected α-Amino Acids Buffers and Reagents for NMR Studies 245 Detergents 245 Environmental Standards 246 Fatty Acids and Fatty Acid Esters 249 Flavours and Fragrances 250 Gases 253 Medical Research Products 254 Nucleic Acid Bases and Nucleosides 255 Pesticides and Pesticide Metabolites 256 Pharmaceutical Standards 257 Polyaromatic Hydrocarbons (PAHs), Alkyl-PAHs, Amino-PAHs, 260 Hydroxy-PAHs and Nitro-PAHs Polychlorinated Biphenyls (PCBs) 260 Spin Labels 261 Steroids 261 3 International Distributors C Beijng Zhenxiang H EQ Laboratories GmbH Australia K Technology Company Graf-von-Seyssel-Str. 10 Rm. 15A01, Changyin Bld. 86199 Augsburg Austria H No. 88, YongDingLu Rd. Germany Beijing 100039 Tel.: (49) 821 71058246 Belgium J China Fax: (49) 821 71058247 Tel.: (86) 10-58896805 [email protected] China C Fax: (86) 10-58896158 www.eqlabs.de Czech Republic H [email protected] Germany, Austria, China Czech Republic, Greece, Denmark I Hungary, -

C:\Data\Ndaenjuvia\AP LTR 05-07-04
NDA 21-443 Package Insert ENJUVIA™ (synthetic conjugated estrogens, B) Tablets Rx only ESTROGENS INCREASE THE RISK OF ENDOMETRIAL CANCER Close clinical surveillance of all women taking estrogens is important. Adequate diagnostic measures, including endometrial sampling when indicated, should be undertaken to rule out malignancy in all cases of undiagnosed persistent or recurring abnormal vaginal bleeding. There is no evidence that the use of “natural” estrogens results in a different endometrial risk profile than synthetic estrogens at equivalent estrogen doses. (See WARNINGS, Malignant neoplasms, Endometrial cancer.) CARDIOVASCULAR AND OTHER RISKS Estrogens with or without progestins should not be used for the prevention of cardiovascular disease. (See WARNINGS, Cardiovascular disorders.) The Women’s Health Initiative (WHI) study reported increased risks of myocardial infarction, stroke, invasive breast cancer, pulmonary emboli, and deep vein thrombosis in postmenopausal women (50 to 79 years of age) during 5 years of treatment with oral conjugated estrogens (CE 0.625 mg) combined with medroxyprogesterone acetate (MPA 2.5 mg) relative to placebo. (See CLINICAL PHARMACOLOGY, Clinical Studies). The Women’s Health Initiative Memory Study (WHIMS), a substudy of WHI, reported increased risk of developing probable dementia in postmenopausal women 65 years of age or older during 4 years of treatment with oral conjugated estrogens plus medroxyprogesterone acetate relative to placebo. It is unknown whether this finding applies to younger postmenopausal women or to women taking estrogen alone therapy. (See CLINICAL PHARMACOLOGY, Clinical Studies.) Other doses of oral conjugated estrogens with medroxyprogesterone acetate, and other combinations and dosage forms of estrogens and progestins were not studied in the WHI clinical trials and, in the absence of comparable data, these risks should be assumed to be similar. -

Depo-Estradiol (Estradiol Cypionate) Injection
Market Applicability Market DC GA KY MD NJ NY WA Applicable X X X X X X NA Depo-Estradiol (estradiol cypionate) injection Override(s) Approval Duration Prior Authorization 1 year Medications Depo-Estradiol (estradiol cypionate) injection APPROVAL CRITERIA Requests for Depo-Estradiol (estradiol cypionate) injection may be approved when the following criteria are met: I. Individual is using to treat moderate to severe vasomotor symptoms associated with menopause; OR II. Individual has been diagnosed with hypoestrogenism due to hypogonadism; OR III. Individual has a diagnosis of gender dysphoria or gender identity disorder (WPATH 2012, Endocrine Society 2017) ; AND IV. Individual is 16 years of age or older; AND V. The goal of treatment is male-to-female gender reassignment. Requests for Depo-Estradiol (estradiol cypionate) injection may not be approved for any of the following: I. Individual has undiagnosed abnormal genital bleeding; OR II. Individual has known or suspected cancer of the breast; OR III. Individual has known or suspected estrogen-dependent neoplasia; OR IV. Individual has active deep vein thrombosis, pulmonary embolism or history of these conditions; OR V. Individual has active or recent (within the past year) arterial thromboembolic disease (such as stroke or myocardial infarction); OR VI. Individual has liver dysfunction or disease. PAGE 1 of 2 02/24/2020 New Program Date 02/24/2020 CRX-ALL-0519-20 This policy does not apply to health plans or member categories that do not have pharmacy benefits, nor does it apply to Medicare. Note that market specific restrictions or transition-of-care benefit limitations may apply. -
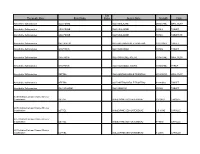
Therapeutic Class Brand Name P a Status Generic
P A Therapeutic Class Brand Name Status Generic Name Strength Form Absorbable Sulfonamides AZULFIDINE SULFASALAZINE 250MG/5ML ORAL SUSP Absorbable Sulfonamides AZULFIDINE SULFASALAZINE 500MG TABLET Absorbable Sulfonamides AZULFIDINE SULFASALAZINE 500MG TABLET DR Absorbable Sulfonamides BACTRIM DS SULFAMETHOXAZOLE/TRIMETHO 800-160MG TABLET Absorbable Sulfonamides GANTRISIN SULFISOXAZOLE 500MG TABLET Absorbable Sulfonamides GANTRISIN SULFISOXAZOLE ACETYL 500MG/5ML ORAL SUSP Absorbable Sulfonamides GANTRISIN SULFISOXAZOLE ACETYL 500MG/5ML SYRUP Absorbable Sulfonamides SEPTRA SULFAMETHOXAZOLE/TRIMETHO 200-40MG/5 ORAL SUSP Absorbable Sulfonamides SEPTRA SULFAMETHOXAZOLE/TRIMETHO 400-80MG TABLET Absorbable Sulfonamides SULFADIAZINE SULFADIAZINE 500MG TABLET ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 10-20MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 2.5-10MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 5-10MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 5-20MG CAPSULE P A Therapeutic Class Brand Name Status Generic Name Strength Form ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 5-40MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 10-40MG CAPSULE Acne Agents, Systemic ACCUTANE ISOTRETINOIN 10MG CAPSULE Acne Agents, Systemic ACCUTANE ISOTRETINOIN 20MG CAPSULE Acne Agents, Systemic ACCUTANE -

PEMD-91-12BR Off-Label Drugs: Initial Results of a National Survey
11 1; -- __...._-----. ^.-- ______ -..._._ _.__ - _........ - t Ji Jo United States General Accounting Office Washington, D.C. 20648 Program Evaluation and Methodology Division B-242851 February 25,199l The Honorable Edward M. Kennedy Chairman, Committee on Labor and Human Resources United States Senate Dear Mr. Chairman: In September 1989, you asked us to conduct a study on reimbursement denials by health insurers for off-label drug use. As you know, the Food and Drug Administration designates the specific clinical indications for which a drug has been proven effective on a label insert for each approved drug, “Off-label” drug use occurs when physicians prescribe a drug for clinical indications other than those listed on the label. In response to your request, we surveyed a nationally representative sample of oncologists to determine: . the prevalence of off-label use of anticancer drugs by oncologists and how use varies by clinical, demographic, and geographic factors; l the extent to which third-party payers (for example, Medicare intermediaries, private health insurers) are denying payment for such use; and l whether the policies of third-party payers are influencing the treatment of cancer patients. We randomly selected 1,470 members of the American Society of Clinical Oncologists and sent them our survey in March 1990. The sam- pling was structured to ensure that our results would be generalizable both to the nation and to the 11 states with the largest number of oncologists. Our response rate was 56 percent, and a comparison of respondents to nonrespondents shows no noteworthy differences between the two groups. -

Clomifene Citrate(BANM, Rinnm) ⊗
2086 Sex Hormones and their Modulators Profasi; UK: Choragon; Ovitrelle; Pregnyl; USA: Chorex†; Choron; Gonic; who received the drug for a shorter period.6 No association be- 8. Werler MM, et al. Ovulation induction and risk of neural tube Novarel; Ovidrel; Pregnyl; Profasi; Venez.: Ovidrel; Pregnyl; Profasi†. tween gonadotrophin therapy and ovarian cancer was noted in defects. Lancet 1994; 344: 445–6. Multi-ingredient: Ger.: NeyNormin N (Revitorgan-Dilutionen N Nr this study. The conclusions of this study were only tentative, 9. Greenland S, Ackerman DL. Clomiphene citrate and neural tube 65)†; Mex.: Gonakor. defects: a pooled analysis of controlled epidemiologic studies since the numbers who developed ovarian cancer were small; it and recommendations for future studies. Fertil Steril 1995; 64: has been pointed out that a successfully achieved pregnancy may 936–41. reduce the risk of some other cancers, and that the risks and ben- 10. Whiteman D, et al. Reproductive factors, subfertility, and risk efits of the procedure are not easy to balance.7 A review8 of epi- of neural tube defects: a case-control study based on the Oxford Clomifene Citrate (BANM, rINNM) ⊗ Record Linkage Study Register. Am J Epidemiol 2000; 152: demiological and cohort studies concluded that clomifene was 823–8. Chloramiphene Citrate; Citrato de clomifeno; Clomifène, citrate not associated with any increase in the risk of ovarian cancer 11. Sørensen HT, et al. Use of clomifene during early pregnancy de; Clomifeni citras; Clomiphene Citrate (USAN); Klomifeenisi- when used for less than 12 cycles, but noted conflicting results, and risk of hypospadias: population based case-control study. -

Pms-ESTRADIOL VALERATE INJECTION
PRODUCT MONOGRAPH Prpms-ESTRADIOL VALERATE INJECTION Estradiol Valerate 10 mg/mL Estrogen PHARMASCIENCE INC. Date of Revision: 6111 Royalmount Avenue, Suite 100 June 30, 2009 Montreal, Quebec H4P 2T4 Control Number: 120632 Table of Contents PART I: HEALTH PROFESSIONAL INFORMATION……………………………...3 SUMMARY PRODUCT INFORMATION……………………………………….3 INDICATIONS AND CLINICAL USE…………………………………………...3 CONTRAINDICATIONS…………………………………………………………4 WARNINGS AND PRECAUTIONS…………………………………….……….5 ADVERSE REACTIONS………………………………………………………...13 DRUG INTERACTIONS………………………………………………………...15 DOSAGE AND ADMINISTRATION…………………………………………...17 OVERDOSAGE………………………………………………………………….19 ACTION AND CLINICAL PHARMACOLOGY……………………………….19 STORAGE AND STABILITY…………………………………………………...22 DOSAGE FORMS, COMPOSITION AND PACKAGING……………………..22 PART II: SCIENTIFIC INFORMATION…………………………………………….23 PHARMACEUTICAL INFORMATION………………………………………...23 CLINICAL TRIALS……………………………………………………………...24 DETAILED PHARMACOLOGY………………………………………………..24 REFERENCES…………………………………………………………………...25 PART III: CONSUMER INFORMATION……………………………………………………27 2 PRODUCT MONOGRAPH Prpms- ESTRADIOL VALERATE INJECTION (Estradiol Valerate) 10 mg/mL PART I: HEALTH PROFESSIONAL INFORMATION SUMMARY PRODUCT INFORMATION Route of Dosage Form / Strength Clinically Relevant Nonmedicinal Administration Ingredients Intramuscular Injection / 10 mg/mL Sesame Oil For a complete listing see Dosage Forms, Composition and Packaging section. INDICATIONS AND CLINICAL USE pms-ESTRADIOL VALERATE INJECTION is indicated in the treatment of: I. amenorrhea (primary -

Estrone Sulfate
Available online at www.sciencedirect.com Journal of Steroid Biochemistry & Molecular Biology 109 (2008) 158–167 Estrone sulfate (E1S), a prognosis marker for tumor aggressiveness in prostate cancer (PCa)ଝ Frank Giton a,∗, Alexandre de la Taille b, Yves Allory b, Herve´ Galons c, Francis Vacherot b, Pascale Soyeux b, Claude Clement´ Abbou b, Sylvain Loric b, Olivier Cussenot b, Jean-Pierre Raynaud d, Jean Fiet b a AP-HP CIB INSERM IMRB U841eq07, Henri Mondor, Facult´edeM´edecine, 94010 Cr´eteil, France b INSERM IMRB U841 eq07, CHU Henri Mondor, Facult´edeM´edecine, 94010 Cr´eteil, France c Service de Chimie organique, Facult´e de Pharmacie Paris V, 75006 Paris, France d Universit´e Pierre et Marie Curie, 75252 Paris, France Received 26 December 2006; accepted 26 October 2007 Abstract Seeking insight into the possible role of estrogens in prostate cancer (PCa) evolution, we assayed serum E2, estrone (E1), and estrone sulfate (E1S) in 349 PCa and 100 benign prostatic hyperplasia (BPH) patients, and in 208 control subjects in the same age range (50–74 years). E1 (pmol/L ± S.D.) and E1S (nmol/L ± S.D.) in the PCa and BPH patients (respectively 126.1 ± 66.1 and 2.82 ± 1.78, and 127.8 ± 56.4 and 2.78 ± 2.12) were significantly higher than in the controls (113.8 ± 47.6 and 2.11 ± 0.96). E2 was not significantly different among the PCa, BPH, and control groups. These assays were also carried out in PCa patients after partition by prognosis (PSA, Gleason score (GS), histological stage, and surgical margins (SM)). -

MENEST ® Brand of Esterified Estrogens Tablets, USP
PRESCRIBING INFORMATION MENEST ® brand of esterified estrogens tablets, USP WARNINGS 1. ESTROGENS HAVE BEEN REPORTED TO INCREASE THE RISK OF ENDOMETRIAL CARCINOMA. Three independent case control studies have shown an increased risk of endometrial cancer in postmenopausal women exposed to exogenous estrogens for prolonged periods.1–3 This risk was independent of the other known risk factors for endometrial cancer. These studies are further supported by the finding that incidence rates of endometrial cancer have increased sharply since 1969 in eight different areas of the United States with population-based cancer reporting systems, an increase which may be related to the rapidly expanding use of estrogens during the last decade.4 The three case control studies reported that the risk of endometrial cancer in estrogen users was about 4.5 to 13.9 times greater than in nonusers. The risk appears to depend on both duration of treatment1 and on estrogen dose.3 In view of these findings, when estrogens are used for the treatment of menopausal symptoms, the lowest dose that will control symptoms should be utilized and medication should be discontinued as soon as possible. When prolonged treatment is medically indicated, the patient should be reassessed on at least a semiannual basis to determine the need for continued therapy. Although the evidence must be considered preliminary, one study sug- gests that cyclic administration of low doses of estrogen may carry less risk than continuous administration3; it therefore appears prudent to utilize such a regimen. Close clinical surveillance of all women taking estrogens is important. In all cases of undiagnosed persistent or recurring abnormal vaginal bleeding, adequate diagnostic measures should be undertaken to rule out malignancy. -

Mass Spectrometry Analysis of Hormones in Water by Direct Injection
Application Note Environmental Mass Spectrometry Analysis of Hormones in Water by Direct Injection Using the Agilent 6470 Triple Quadrupole Mass Spectrometer Authors Abstract Imma Ferrer, E. Michael Thurman, and Some hormones are included in the Contaminant Candidate List CCL4 of the Jerry A. Zweigenbaum Environmental Protection Agency (EPA) to be assessed for regulation in drinking University of Colorado and water. These compounds are also of regulatory interest in the EU, China, and other Agilent Technologies, Inc. countries. Therefore, the environmental community often desires the analysis of these compounds in water samples. This Application Note describes the methodology used for the determination of eight hormones (17-α-ethinylestradiol, 17-β-estradiol, estriol, 4-androstene-3,17-dione, equilin, estrone, progesterone, and testosterone) in tap water using an Agilent 6470 triple quadrupole mass spectrometer. A direct injection method using 100 µL of water sample was carried out. This method saves time, reduces handling errors and analytical variability, and is sensitive enough to detect hormones in surface and drinking water at ng/L levels. Introduction Both positive and negative ion modes and stored at −18 °C. A mix of all were used, depending on the specific the hormones was prepared at a The presence of hormones in compound. We also used a novel concentration of 1 μg/mL. Serial dilutions environmental waters has always been mobile phase approach that included were prepared to obtain a calibration controversial because either none have ammonium fluoride to enhance the curve ranging from 1 to 500 ng/L. been detected, or very low traces have negative ion signal4. -

Chemical Compounds As Carcinogenic Agents Second Supplementary Report: Literature of 1938 and 1939 Biological Considerations
CHEMICAL COMPOUNDS AS CARCINOGENIC AGENTS SECONDSUPPLEMENTARY REPORT: LITERATURE OF 1938 AND 1939 J. W. COOK AND E. L. KENNAWAY (From the Royal Cancer Hospital (Free), London, and the University of Glasgow) BIOLOGICALCONSIDERATIONS (Continued from page 428, Jitly 1940) II. Action of Carcinogenic Compounds in Difierent Species and Tissues A number of reports of the action of carcinogenic compounds on human tis- sues have appeared. Klar (601) developed a nodule on the forearm after completion of a series of experiments with 3:4-benzpyrene. He applied a solution of the hydrocarbon (0.25 per cent in benzol) to the skin of mice with a paint brush and for at least part of the period wore rubber gloves. He also conducted experiments, of which no description is given, with the powdered hydrocarbon contained in a glass vessel. Three months after the completion of the experiments a small nodule appeared on the dorsum of the left forearm, This was excised in May 1938 and described by Professor Huckel as a “ so- called benign calcifying epithelioma.” The growth extended into the subcu- taneous fatty tissue; the connection with the superficial epithelium is not described nor is it evident in the two photomicrographs which illustrate the report. The author does not state his age. Gordonoff and Walthard (562) record the occurrence of a tumor in a labo- ratory assistant, aged forty-two, engaged in applying methylcholanthrene (0.3 per cent in benzol) to the skin of mice. The site was in the nasolabial fold, at a spot often touched by the patient when smoking. The microscopic ap- pearance was that of a “ still well delimited stage of an incipient squamous- cell sarcoma.” Cottini and Mazzone (479) deliberately applied 3 :4-benzpyrene ( 1 per cent in benzene) to the skin, generally of the arm or thigh, of 26 patients with various cutaneous diseases, usually daily for periods up to 120 days. -

Full Text Pdf
Eastern Biologist No. 4 2015 Protective Effects of Conjugated Equine Estrogens and 17-β Estradiol on Oxidatively Stressed Astrocytes Whitley E. Grimes and Kathleen S. Hughes The Eastern Biologist . ♦ A peer-reviewed journal that publishes original articles focused on the many diverse disciplines of biological research, except for the natural history science disciplines (ISSN 2165-6657 [online]). ♦ Subject areas - The journal welcomes manuscripts based on original research and observations, as well as research summaries and general interest articles. Subject areas include, but are not limited to, biochemistry, biotechnology, cell biology, developmental biology, genetics and genomics, immunology, microbiology, molecular evolution, neurobiology, parasitology, physiology, toxicology, as well as scientific pedagogy. ♦ Offers article-by-article online publication for prompt distribution to a global audience ♦ Offers authors the option of publishing large files such as data tables, audio and video clips, and even PowerPoint presentations as online supplemental files. ♦ Special issues - The Eastern Biologist welcomes proposals for special issues that are based on conference proceedings or on a series of invitational articles. Special issue editors can rely on the publisher’s years of experiences in efficiently handling most details relating to the publication of special issues. ♦ Indexing - As is the case with Eagle Hill's other journals, the Eastern Biologist is expected to be fully indexed in Elsevier, Thomson Reuters, Proquest, EBSCO, Google Scholar, and other databases. ♦ The journal staff is pleased to discuss ideas for manuscripts and to assist during all stages of manuscript preparation. The journal has a mandatory page charge to help defray a portion of the costs of publishing the manuscript.