Hormone Replacement Therapy and Risk of Venous Thromboembolism
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Trauma-Associated Pulmonary Laceration in Dogs—A Cross Sectional Study of 364 Dogs
veterinary sciences Article Trauma-Associated Pulmonary Laceration in Dogs—A Cross Sectional Study of 364 Dogs Giovanna Bertolini 1,* , Chiara Briola 1, Luca Angeloni 1, Arianna Costa 1, Paola Rocchi 2 and Marco Caldin 3 1 Diagnostic and Interventional Radiology Division, San Marco Veterinary Clinic and Laboratory, via dell’Industria 3, 35030 Veggiano, Padova, Italy; [email protected] (C.B.); [email protected] (L.A.); [email protected] (A.C.) 2 Intensive Care Unit, San Marco Veterinary Clinic and Laboratory, via dell’Industria 3, 35030 Veggiano, Padova, Italy; [email protected] 3 Clinical Pathology Division, San Marco Veterinary Clinic and Laboratory, via dell’Industria 3, 35030 Veggiano, Padova, Italy; [email protected] * Correspondence: [email protected]; Tel.: +39-0498561098 Received: 5 March 2020; Accepted: 8 April 2020; Published: 12 April 2020 Abstract: In this study, we describe the computed tomography (CT) features of pulmonary laceration in a study population, which included 364 client-owned dogs that underwent CT examination for thoracic trauma, and compared the characteristics and outcomes of dogs with and without CT evidence of pulmonary laceration. Lung laceration occurred in 46/364 dogs with thoracic trauma (prevalence 12.6%). Dogs with lung laceration were significantly younger than dogs in the control group (median 42 months (interquartile range (IQR) 52.3) and 62 months (IQR 86.1), respectively; p = 0.02). Dogs with lung laceration were significantly heavier than dogs without laceration (median 20.8 kg (IQR 23.3) and median 8.7 kg (IQR 12.4 kg), respectively p < 0.0001). When comparing groups of dogs with thoracic trauma with and without lung laceration, the frequency of high-energy motor vehicle accident trauma was more elevated in dogs with lung laceration than in the control group. -

Download Product Insert (PDF)
PRODUCT INFORMATION Quinestrol Item No. 10006320 CAS Registry No.: 152-43-2 Formal Name: 3-(cyclopentyloxy)-19-norpregna- 1,3,5(10)-trien-20-yn-17α-ol Synonyms: Ethylnyl Estradiol-3-cyclopentyl ether, W 3566 MF: C25H32O2 FW: 3645 Purity: ≥98% UV/Vis.: λmax: 202, 281 nm Supplied as: A crystalline solid Storage: -20°C Stability: ≥2 years Information represents the product specifications. Batch specific analytical results are provided on each certificate of analysis. Laboratory Procedures Quinestrol is supplied as a crystalline solid. A stock solution may be made by dissolving the quinestrol in an organic solvent purged with an inert gas. Quinestrol is soluble in organic solvents such as ethanol, DMSO, and dimethyl formamide (DMF). The solubility of quinestrol in ethanol is approximately 20 mg/ml and approximately 30 mg/ml in DMSO and DMF. Quinestrol is sparingly soluble in aqueous buffers. For maximum solubility in aqueous buffers, should first be dissolved in DMSO and then diluted with the aqueous buffer of choice. Quinestrol has a solubility of approximately 0.5 mg/ml in a 1:1 solution of DMSO:PBS (pH 7.2) using this method. We do not recommend storing the aqueous solution for more than one day. Description Quinestrol is a synthetic estrogen that is effective in hormone replacement therapy.1,2 It is a 3-cyclopentyl ether of ethynyl estradiol. After gastrointestinal absorption, it is stored in adipose tissue, where it is slowly released and metabolized in the liver to its active form, ethynyl estradiol. Quinestrol has found limited use in suppressing lactation in postpartum women and, in combination with synthetic progestogens, as contraceptive therapy, although additional studies are needed for both applications. -
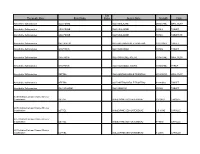
Therapeutic Class Brand Name P a Status Generic
P A Therapeutic Class Brand Name Status Generic Name Strength Form Absorbable Sulfonamides AZULFIDINE SULFASALAZINE 250MG/5ML ORAL SUSP Absorbable Sulfonamides AZULFIDINE SULFASALAZINE 500MG TABLET Absorbable Sulfonamides AZULFIDINE SULFASALAZINE 500MG TABLET DR Absorbable Sulfonamides BACTRIM DS SULFAMETHOXAZOLE/TRIMETHO 800-160MG TABLET Absorbable Sulfonamides GANTRISIN SULFISOXAZOLE 500MG TABLET Absorbable Sulfonamides GANTRISIN SULFISOXAZOLE ACETYL 500MG/5ML ORAL SUSP Absorbable Sulfonamides GANTRISIN SULFISOXAZOLE ACETYL 500MG/5ML SYRUP Absorbable Sulfonamides SEPTRA SULFAMETHOXAZOLE/TRIMETHO 200-40MG/5 ORAL SUSP Absorbable Sulfonamides SEPTRA SULFAMETHOXAZOLE/TRIMETHO 400-80MG TABLET Absorbable Sulfonamides SULFADIAZINE SULFADIAZINE 500MG TABLET ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 10-20MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 2.5-10MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 5-10MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 5-20MG CAPSULE P A Therapeutic Class Brand Name Status Generic Name Strength Form ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 5-40MG CAPSULE ACE Inhibitor/Calcium Channel Blocker Combination LOTREL AMLODIPINE BESYLATE/BENAZ 10-40MG CAPSULE Acne Agents, Systemic ACCUTANE ISOTRETINOIN 10MG CAPSULE Acne Agents, Systemic ACCUTANE ISOTRETINOIN 20MG CAPSULE Acne Agents, Systemic ACCUTANE -

(12) United States Patent (10) Patent No.: US 9,498,481 B2 Rao Et Al
USOO9498481 B2 (12) United States Patent (10) Patent No.: US 9,498,481 B2 Rao et al. (45) Date of Patent: *Nov. 22, 2016 (54) CYCLOPROPYL MODULATORS OF P2Y12 WO WO95/26325 10, 1995 RECEPTOR WO WO99/O5142 2, 1999 WO WOOO/34283 6, 2000 WO WO O1/92262 12/2001 (71) Applicant: Apharaceuticals. Inc., La WO WO O1/922.63 12/2001 olla, CA (US) WO WO 2011/O17108 2, 2011 (72) Inventors: Tadimeti Rao, San Diego, CA (US); Chengzhi Zhang, San Diego, CA (US) OTHER PUBLICATIONS Drugs of the Future 32(10), 845-853 (2007).* (73) Assignee: Auspex Pharmaceuticals, Inc., LaJolla, Tantry et al. in Expert Opin. Invest. Drugs (2007) 16(2):225-229.* CA (US) Wallentin et al. in the New England Journal of Medicine, 361 (11), 1045-1057 (2009).* (*) Notice: Subject to any disclaimer, the term of this Husted et al. in The European Heart Journal 27, 1038-1047 (2006).* patent is extended or adjusted under 35 Auspex in www.businesswire.com/news/home/20081023005201/ U.S.C. 154(b) by Od en/Auspex-Pharmaceuticals-Announces-Positive-Results-Clinical M YW- (b) by ayS. Study (published: Oct. 23, 2008).* This patent is Subject to a terminal dis- Concert In www.concertpharma. com/news/ claimer ConcertPresentsPreclinicalResultsNAMS.htm (published: Sep. 25. 2008).* Concert2 in Expert Rev. Anti Infect. Ther. 6(6), 782 (2008).* (21) Appl. No.: 14/977,056 Springthorpe et al. in Bioorganic & Medicinal Chemistry Letters 17. 6013-6018 (2007).* (22) Filed: Dec. 21, 2015 Leis et al. in Current Organic Chemistry 2, 131-144 (1998).* Angiolillo et al., Pharmacology of emerging novel platelet inhibi (65) Prior Publication Data tors, American Heart Journal, 2008, 156(2) Supp. -

PEMD-91-12BR Off-Label Drugs: Initial Results of a National Survey
11 1; -- __...._-----. ^.-- ______ -..._._ _.__ - _........ - t Ji Jo United States General Accounting Office Washington, D.C. 20648 Program Evaluation and Methodology Division B-242851 February 25,199l The Honorable Edward M. Kennedy Chairman, Committee on Labor and Human Resources United States Senate Dear Mr. Chairman: In September 1989, you asked us to conduct a study on reimbursement denials by health insurers for off-label drug use. As you know, the Food and Drug Administration designates the specific clinical indications for which a drug has been proven effective on a label insert for each approved drug, “Off-label” drug use occurs when physicians prescribe a drug for clinical indications other than those listed on the label. In response to your request, we surveyed a nationally representative sample of oncologists to determine: . the prevalence of off-label use of anticancer drugs by oncologists and how use varies by clinical, demographic, and geographic factors; l the extent to which third-party payers (for example, Medicare intermediaries, private health insurers) are denying payment for such use; and l whether the policies of third-party payers are influencing the treatment of cancer patients. We randomly selected 1,470 members of the American Society of Clinical Oncologists and sent them our survey in March 1990. The sam- pling was structured to ensure that our results would be generalizable both to the nation and to the 11 states with the largest number of oncologists. Our response rate was 56 percent, and a comparison of respondents to nonrespondents shows no noteworthy differences between the two groups. -

Postmenopausal Pharmacotherapy Newsletter
POSTMENOPAUSAL PHARMACOTHERAPY September, 1999 As Canada's baby boomers age, more and more women will face the option of Hormone Replacement Therapy (HRT). The HIGHLIGHTS decision can be a difficult one given the conflicting pros and cons. M This RxFiles examines the role and use of HRT, as well as newer Long term HRT carries several major benefits but also risks SERMS and bisphosphonates in post-menopausal (PM) patients. which should be evaluated on an individual and ongoing basis MContinuous ERT is appropriate for women without a uterus HRT MWomen with a uterus should receive progestagen (at least 12 HRT is indicated for the treatment of PM symptoms such as days per month or continuous low-dose) as part of their HRT vasomotor disturbances and urogenital atrophy, and is considered MLow-dose ERT (CEE 0.3mg) + Ca++ appears to prevent PMO primary therapy for prevention and treatment of postmenopausal MBisphosphinates (e.g. alendronate, etidronate) and raloxifene are osteoporosis (PMO).1 Contraindications are reviewed in Table 2. alternatives to HRT in treating and preventing PMO Although HRT is contraindicated in women with active breast or M"Natural" HRT regimens can be compounded but data is lacking uterine cancer, note that a prior or positive family history of these does not necessarily preclude women from receiving HRT.1 Comparative Safety: Because of differences between products, some side effects may be alleviated by switching from one product Estrogen Replacement Therapy (ERT) 2 to another, particularly from equine to plant sources or from oral to Naturally secreted estrogens include: topical (see Table 3 - Side Effects & Their Management). -

Case Report a Case of a Patient Who Is Diagnosed with Mild Acquired Hemophilia a After Tooth Extraction Died of Acute Subdural Hematoma Due to Head Injury
Hindawi Case Reports in Dentistry Volume 2018, Article ID 7185263, 3 pages https://doi.org/10.1155/2018/7185263 Case Report A Case of a Patient Who Is Diagnosed with Mild Acquired Hemophilia A after Tooth Extraction Died of Acute Subdural Hematoma due to Head Injury Tomohisa Kitamura,1 Tsuyoshi Sato ,1 Eiji Ikami,1 Yosuke Fukushima,1 and Tetsuya Yoda2 1Department of Oral and Maxillofacial Surgery, Saitama Medical University, 38 Moro-hongou, Moroyama-machi, Iruma-gun, Saitama 350-0495, Japan 2Department of Maxillofacial Surgery, Tokyo Medical and Dental University, Tokyo, Japan Correspondence should be addressed to Tsuyoshi Sato; [email protected] Received 13 September 2018; Revised 12 November 2018; Accepted 25 November 2018; Published 9 December 2018 Academic Editor: Yuk-Kwan Chen Copyright © 2018 Tomohisa Kitamura et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Background. Acquired hemophilia A (AHA) is a rare disorder which results from the presence of autoantibodies against blood coagulation factor VIII. The initial diagnosis is based on the detection of an isolated prolongation of the activated partial thromboplastin time (aPTT) with negative personal and family history of bleeding disorder. Definitive diagnosis is the identification of reduced FVIII levels with evidence of FVIII neutralizing activity. Case report. We report a case of a 93-year-old female who was diagnosed as AHA after tooth extraction at her home clinic. Prolongation of aPTT and a reduction in factor VIII activity levels were observed with the presence of factor VIII inhibitor. -

Blood Counts
Medicare National Coverage Determinations (NCD) Coding Policy Manual and Change Report April 2009 Clinical Diagnostic Laboratory Services Health & Human Services Department Centers for Medicare & Medicaid Services 7500 Security Boulevard Baltimore, MD 21244 CMS Email Point of Contact: [email protected] TDD 410.786.0727 Fu Associates, Ltd. Medicare National Coverage Determinations (NCD) Coding Policy Manual and Change Report This is CMS Logo. NCD Manual Changes Date Reason Release Change Edit The following section represents NCD Manual updates for April 2009. 04/01/09 Per CR 6383 add 2009200 *525.71 Osseointegration *190.15 Blood Counts ICD-9-CM codes failure of dental implant 525.71, 525.72 and 525.73 to the list of ICD-9-CM codes that *525.72 Post- do not support osseointegration Medical necessity for biological failure of the Blood Counts dental implant NCD. *525.73 Post- Transmittal # 1684 osseointegration mechanic failure of dental implant 04/01/09 Per CR 6383 add 2009200 *535.70 Eosinophilic *190.16 Partial ICD-9-CM codes gastritis, without Thromboplastin Time 535.70 and 535.71 to mention of obstruction (PTT) the list of ICD-9-CM codes covered by Medicare for the *535.71 Eosinophilic Partial gastritis, with Thromboplastin Time obstruction NCD. Transmittal # 1684 04/01/09 Per CR 6383 add 2009200 *414.3 Coronary *190.17 Prothrombin ICD-9-CM codes atherosclerosis due to Time 414.3, 535.70 and lipid rich plaque 535.71 to the list of ICD-9-CM codes *535.70 Eosinophilic covered by Medicare gastritis, without mention for the Prothrombin of obstruction Time NCD. -

The Reactivity of Human and Equine Estrogen Quinones Towards Purine Nucleosides
S S symmetry Article The Reactivity of Human and Equine Estrogen Quinones towards Purine Nucleosides Zsolt Benedek †, Peter Girnt † and Julianna Olah * Department of Inorganic and Analytical Chemistry, Budapest University of Technology and Economics, Szent Gellért tér 4, H-1111 Budapest, Hungary; [email protected] (Z.B.); [email protected] (P.G.) * Correspondence: [email protected] † These authors contributed equally to this work. Abstract: Conjugated estrogen medicines, which are produced from the urine of pregnant mares for the purpose of menopausal hormone replacement therapy (HRT), contain the sulfate conjugates of estrone, equilin, and equilenin in varying proportions. The latter three steroid sex hormones are highly similar in molecular structure as they only differ in the degree of unsaturation of the sterane ring “B”: the cyclohexene ring in estrone (which is naturally present in both humans and horses) is replaced by more symmetrical cyclohexadiene and benzene rings in the horse-specific (“equine”) hormones equilin and equilenin, respectively. Though the structure of ring “B” has only moderate influence on the estrogenic activity desired in HRT, it might still significantly affect the reactivity in potential carcinogenic pathways. In the present theoretical study, we focus on the interaction of estrogen orthoquinones, formed upon metabolic oxidation of estrogens in breast cells with purine nucleosides. This multistep process results in a purine base loss in the DNA chain (depurination) and the formation of a “depurinating adduct” from the quinone and the base. The point mutations induced in this manner are suggested to manifest in breast cancer development in the long run. -

Aphrodisiac & Tonic Activity of Tribulus Leaf
No. 57 January 2011 Aphrodisiac & Tonic Activity of Tribulus Leaf Key Points at a Glance Tribulus Leaf Constituents & Quality • consumed as a vegetable and used traditionally as a tonic • contains furostanol glycosides (which are steroidal • concentrated extract of Tribulus leaf (of Eastern European saponins), in particular, protodioscin origin and/or containing furostanol saponins, predominantly • protodioscin level may be critical for efficacy protodioscin) clinically demonstrated to: • fruit as well as plant grown in locations other than Eastern − improve sperm and semen parameters in men with Europe may contain furostanol saponins, but not necessarily infertility with sufficient protodioscin − improve libido in men • method of analysis determines the accuracy of furostanol − improve ovulation in some women with infertility saponin content − relieve menopausal symptoms • much clinical research published on Tribulus but often − possibly enhance the body’s natural production of sex undefined for plant part, saponin profile or content of hormones, especially when at low levels, but is unlikely protodioscin to increase beyond normal range • steroidal saponins may act by binding to vacant receptors in the hypothalamus Traditional Uses The high content of steroidal saponins is a characteristic feature of Tribulus terrestris . The composition of the There is little information available on the traditional use saponins correlates with the place of origin of the plant. of the leaf of Tribulus ( Tribulus terrestris ). In Ayurveda the The main constituent of Tribulus aerial parts (leaf and plant and fruit have been used to treat spermatorrhoea, stem) from Bulgaria is the furostanol glycoside gonorrhoea, impotence, uterine disorders after parturition, protodioscin. 10 cystitis, painful urination, kidney stones and gout. -

Steroid Sex Hormones Non Steroid Hormones Fig. A1. Chemical
Electronic Supplementary Material (ESI) for Analytical Methods. This journal is © The Royal Society of Chemistry 2015 Steroid sex hormones O OH H H H H H H O Testosterone (T) HO Estrone (E1) OH O H H H H H H O HO 17β-Estradiol (17β-E2) 4-Androstene-3,17-dione (AND) OH OH H OH H H H H H H O HO Nandrolone (NAN) Estriol (E3) OH OH H H H H H H O 17α-Methyltestosterone (17α-MT) HO Ethinylestradiol (EE2) OH O O H HO OH H H H H H H O Prednisolone (PRED) O Progesterone (P) Non steroid hormones HO CH3 HO CH3 H C OH H C OH 3 Diethylstilbestrol (DES) 3 Hexestrol (HEX) Fig. A1. Chemical structure of selected endocrine disruptors. Fig. A2. Scheme of SPE procedure: a) PTFE disks, b) nylon filter membrane. Table A1. Characterization data for mesoporous silicas a Material BET surface Pore volume Pore L0C18 Particle morphology Average particle size 2 -1 3 -1 -1 (m g ) (cm g ) diameter (Å) (mmol C18 g ) (length x wide) SBA-15-C18 796 0.88 76 0.69 Cylindrical 1.4 µm x 750 nm a Amount of octadecyl groups per gram of silica Q3 Q4 Q2 DH 50 0 -50 -100 -150 -200 (ppm) 29 Fig. A3. Si NMR spectrum of SBA-15-C18. 140 0 % Weight Loss T 120 -5 100 s s o L Exothermic Procces 80 t -10 ) h C º g i 60 ( e T W Endothermic Procces -15 % 40 20 -20 0 -25 -20 100 200 300 400 500 600 700 800 T (ºC) Fig. -

Enhanced Solubility and Dissolution Rate of Raloxifene Using Cycloencapsulation Technique
Journal of Analytical & Pharmaceutical Research Enhanced Solubility and Dissolution Rate of Raloxifene using Cycloencapsulation Technique Research Article Abstract Volume 2 Issue 5 - 2016 The aim of this study was to improve the water solubility of raloxifene by aqueous solution and solid state was evaluated by the phase solubility diagram, powdercomplexing X-ray it diffractometer,with sulphobutylether-β-cyclodextrin. Fourier-transform infrared Inclusion spectroscopy, complexation nuclear in magnetic resonance, scanning electron microscopy, hot stage microscopy and transmission electron microscopy. The inclusion complex behavior of raloxifene 1Department of Pharmaceutical Technology (Formulation), National Institute of Pharmaceutical Education and Research, classifiedand sulphobutylether-β-cyclodextrin as AL-type curve, indicating the were formation examined of 1:1 by stochiomatric molecular modelinginclusion India method. The phase solubility diagram with sulphobutylether-β-cyclodextrin was complex. The apparent solubility constants calculated from phase solubility 2 -1 Technology Development Center, National Institute of diagram was 753 M . Aqueous solubility and dissolution studies indicated that Pharmaceutical Education and Research, India the dissolution rates were remarkably increased in inclusion complex, compared 3Department of Pharmacoinformatics, National Institute of with the physical mixture and drug alone. In conclusion, inclusion complexation Pharmaceutical Education and Research, India 4 solubility and dissolution rate