Preferred Drug List
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Ayahuasca: Spiritual Pharmacology & Drug Interactions
Ayahuasca: Spiritual Pharmacology & Drug Interactions BENJAMIN MALCOLM, PHARMD, MPH [email protected] MARCH 28 TH 2017 AWARE PROJECT Can Science be Spiritual? “Science is not only compatible with spirituality; it is a profound source of spirituality. When we recognize our place in an immensity of light years and in the passage of ages, when we grasp the intricacy, beauty and subtlety of life, then that soaring feeling, that sense of elation and humility combined, is surely spiritual. The notion that science and spirituality are somehow mutually exclusive does a disservice to both.” – Carl Sagan Disclosures & Disclaimers No conflicts of interest to disclose – I don’t get paid by pharma and have no potential to profit directly from ayahuasca This presentation is for information purposes only, none of the information presented should be used in replacement of medical advice or be considered medical advice This presentation is not an endorsement of illicit activity Presentation Outline & Objectives Describe what is known regarding ayahuasca’s pharmacology Outline adverse food and drug combinations with ayahuasca as well as strategies for risk management Provide an overview of spiritual pharmacology and current clinical data supporting potential of ayahuasca for treatment of mental illness Pharmacology Terms Drug ◦ Term used synonymously with substance or medicine in this presentation and in pharmacology ◦ No offense intended if I call your medicine or madre a drug! Bioavailability ◦ The amount of a drug that enters the body and is able to have an active effect ◦ Route specific: bioavailability is different between oral, intranasal, inhalation (smoked), and injected routes of administration (IV, IM, SC) Half-life (T ½) ◦ The amount of time it takes the body to metabolize/eliminate 50% of a drug ◦ E.g. -

The Migraine Attack As a Homeostatic, Neuroprotective Response to Brain Oxidative Stress: Preliminary Evidence for a Theory
ISSN 0017-8748 Headache doi: 10.1111/head.13214 VC 2017 American Headache Society Published by Wiley Periodicals, Inc. Views and Perspectives The Migraine Attack as a Homeostatic, Neuroprotective Response to Brain Oxidative Stress: Preliminary Evidence for a Theory Jonathan M. Borkum, PhD Background.—Previous research has suggested that migraineurs show higher levels of oxidative stress (lipid peroxides) between migraine attacks and that migraine triggers may further increase brain oxidative stress. Oxidative stress is trans- duced into a neural signal by the TRPA1 ion channel on meningeal pain receptors, eliciting neurogenic inflammation, a key event in migraine. Thus, migraines may be a response to brain oxidative stress. Results.—In this article, a number of migraine components are considered: cortical spreading depression, platelet acti- vation, plasma protein extravasation, endothelial nitric oxide synthesis, and the release of serotonin, substance P, calcitonin gene-related peptide, and brain-derived neurotrophic factor. Evidence is presented from in vitro research and animal and human studies of ischemia suggesting that each component has neuroprotective functions, decreasing oxidant production, upregulating antioxidant enzymes, stimulating neurogenesis, preventing apoptosis, facilitating mitochondrial biogenesis, and/or releasing growth factors in the brain. Feedback loops between these components are described. Limitations and challenges to the model are discussed. Conclusions.—The theory is presented that migraines are an integrated -

PMA-Sio2: Heteropolyacid Catalysis for Michael Addition-Convenient Route to Substituted-3-Indoles
Available online at www.derpharmachemica.com ISSN 0975-413X Der Pharma Chemica, 2017, 9(13):112-117 CODEN (USA): PCHHAX (http://www.derpharmachemica.com/archive.html) PMA-SiO2: Heteropolyacid Catalysis for Michael Addition-Convenient Route to Substituted-3-Indoles Vijay Kumar Pasala* Deapartment of Chemistry, Osmania University, Hyderabad-500007, Telangana, India ABSTRACT Synthesis of 3-substituted indoles in a hassel-free, ecofriendly manner by treating indoles with α, β-unsaturated carbonyl or nitro compounds under acidic conditions to give good to excellent yields with shorter reaction durations are described. The catalyst is recyclable for three to four times without great loss in the activity. Keywords: Phosphomolybdic Acid (PMA), Substituted indoles, α, β-unsaturated carbonyl compounds, α, β-unsaturated nitro compounds, Michael addition INTRODUCTION Heterocyclic chemistry is one of the quintessential branches of organic synthesis beaconing towards new scaffolds with medicinal values, new methodologies to the existing active principles etc. One among such skeletons is indole. It is perhaps the most common heterocycles in chemistry and its derivatives are obtained from coal pitch, variety of plants or by the bacterial decay of tryptophan in the intestine [1]. Indole derivatives serve as signaling chemicals in plants and animals, as nucleus building blocks (serotonin [2] (A) a crucial neurotransmitter in the central nervous system [3]), as antibacterial [4], antiviral [5], protein kinase inhibitors [6], anticancer agents [7], entheogens (psilocybin (B) causes perceptional changes), hormones (melatonin (C) regulates sleep and wakefulness), antidepressants (roxindole (D), bufotenin (E)), sumatriptan (F) for the treatment of migraine and ondansetron (G) for the suppression of nausea and vastly found in natural products such as alkaloids (Corynanthe, Iboga, and Aspidosperma alkaloids) [8-10], indigoids etc., which originate, either fully or partly, from bio-oxidation of indoles [11]. -

Pharmacokinetics, Pharmacodynamics and Drug
pharmaceutics Review Pharmacokinetics, Pharmacodynamics and Drug–Drug Interactions of New Anti-Migraine Drugs—Lasmiditan, Gepants, and Calcitonin-Gene-Related Peptide (CGRP) Receptor Monoclonal Antibodies Danuta Szkutnik-Fiedler Department of Clinical Pharmacy and Biopharmacy, Pozna´nUniversity of Medical Sciences, Sw.´ Marii Magdaleny 14 St., 61-861 Pozna´n,Poland; [email protected] Received: 28 October 2020; Accepted: 30 November 2020; Published: 3 December 2020 Abstract: In the last few years, there have been significant advances in migraine management and prevention. Lasmiditan, ubrogepant, rimegepant and monoclonal antibodies (erenumab, fremanezumab, galcanezumab, and eptinezumab) are new drugs that were launched on the US pharmaceutical market; some of them also in Europe. This publication reviews the available worldwide references on the safety of these anti-migraine drugs with a focus on the possible drug–drug (DDI) or drug–food interactions. As is known, bioavailability of a drug and, hence, its pharmacological efficacy depend on its pharmacokinetics and pharmacodynamics, which may be altered by drug interactions. This paper discusses the interactions of gepants and lasmiditan with, i.a., serotonergic drugs, CYP3A4 inhibitors, and inducers or breast cancer resistant protein (BCRP) and P-glycoprotein (P-gp) inhibitors. In the case of monoclonal antibodies, the issue of pharmacodynamic interactions related to the modulation of the immune system functions was addressed. It also focuses on the effect of monoclonal antibodies on expression of class Fc gamma receptors (FcγR). Keywords: migraine; lasmiditan; gepants; monoclonal antibodies; drug–drug interactions 1. Introduction Migraine is a chronic neurological disorder characterized by a repetitive, usually unilateral, pulsating headache with attacks typically lasting from 4 to 72 h. -

Triptans Step Therapy/Quantity Limit Criteria
Triptans Step Therapy/Quantity Limit Criteria Program may be implemented with the following options 1) step therapy 2) quantity limits or 3) step therapy with quantity limits For Blue Cross and Blue Shield of Illinois Option 1 (step therapy only) will apply. Brand Generic Dosage Form Amerge® naratriptan tablets Axert® almotriptan tablets Frova® frovatriptan tablets Imitrex® sumatriptan injection*, nasal spray, tablets* Maxalt® rizatriptan tablets Maxalt-MLT® rizatriptan tablets Relpax® eletriptan tablets Treximet™ sumatriptan and naproxen tablets Zomig® zolmitriptan tablets, nasal spray Zomig-ZMT® zolmitriptan tablets * generic available and included as target agent in quantity limit edit FDA APPROVED INDICATIONS1-7 The following information is taken from individual drug prescribing information and is provided here as background information only. Not all FDA-approved indications may be considered medically necessary. All criteria are found in the section “Prior Authorization Criteria for Approval.” Amerge® Tablets1, Axert® Tablets2, Frova® Tablets3,Imitrex® injection4, Imitrex Nasal Spray5, Imitrex Tablets6, Maxalt® Tablets7, Maxalt-MLT® Tablets7, Relpax® Tablets8, Treximet™ Tablets9, Zomig® Tablets10, Zomig-ZMT® Tablets10, and Zomig® Nasal Spray11 Amerge (naratriptan), Axert (almotriptan), Frova (frovatriptan), Imitrex (sumatriptan), Maxalt (rizatriptan), Maxalt-MLT (rizatriptan orally disintegrating), Relpax (eletriptan), Treximet (sumatriptan/naproxen), Zomig (zolmitriptan), and Zomig-ZMT (zolmitriptan orally disintegrating) tablets, and Imitrex (sumatriptan) and Zomig (zolmitriptan) nasal spray are all indicated for the acute treatment of migraine attacks with or without aura in adults. They are not intended for the prophylactic therapy of migraine or for use in the management of hemiplegic or basilar migraine. Safety and effectiveness of all of these products have not been established for cluster headache, which is present in an older, predominantly male population. -

European Patent Office of Opposition to That Patent, in Accordance with the Implementing Regulations
(19) TZZ _T (11) EP 2 666 772 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: C07D 409/04 (2006.01) C07D 209/16 (2006.01) 01.03.2017 Bulletin 2017/09 C07D 209/18 (2006.01) C07D 209/38 (2006.01) (21) Application number: 13173141.6 (22) Date of filing: 12.12.2003 (54) Synthesis of amines and intermediates for the synthesis thereof Synthese von Aminen und Zwischenverbindungen für die Synthese davon Synthèse d’amines et intermédiaires pour leur synthèse (84) Designated Contracting States: • DEMERSON C A ET AL: "ETODOLIC ACID AND AT BE BG CH CY CZ DE DK EE ES FI FR GB GR RELATED COMPOUNDS CHEMISTRY AND HU IE IT LI LU MC NL PT RO SE SI SK TR ANTIINFLAMMATORY ACTIONS OF SOME POTENT DI-AND TRISUBSTITUTED (30) Priority: 20.12.2002 EP 02406128 1,3,4,9-TETRAHYDROPYRANO not 3,4-B 3/4 INDOLE-I-ACETIC ACIDS", JOURNAL OF (43) Date of publication of application: MEDICINALCHEMISTRY, AMERICAN CHEMICAL 27.11.2013 Bulletin 2013/48 SOCIETY, vol. 19, no. 3, 1 March 1976 (1976-03-01), pages391-395, XP000940626, ISSN: (62) Document number(s) of the earlier application(s) in 0022-2623, DOI: 10.1021/JM00225A010 accordance with Art. 76 EPC: • HARUYASU ET ALL: "Possible metabolic 03799560.2 / 1 572 647 Intermediatesfrom IAA to beta-acid in Rice Bran", AGR. BIOL. CHEM., vol. 40, no. 12, 1976, pages (73) Proprietor: BASF SE 2465-2470, XP002714693, 67056 Ludwigshafen am Rhein (DE) • SOLL R M ET AL: "Multigram preparation of 1,8-diethyl-7-hydroxy-1,3,4,9-tetrahydropy (72) Inventors: rano[3,4-b]indole-1-acetic acid, a phenolic • Berens, Ulrich metabolite of the analgesic and antiinflammatory 79589 Binzen (DE) agent etodolac", THE JOURNAL OF ORGANIC • Dosenbach, Oliver CHEMISTRY, AMERICAN CHEMICAL SOCIETY 79415 Bad Bellingen (DE) [NOT]ETC. -

Patient Information Leaflet
Package Leaflet: Information for the user Sumatriptan 50 mg film-coated tablets Sumatriptan 100 mg film-coated tablets sumatriptan Read all of this leaflet carefully before you start using this medicine because it contains important information for you. • Keep this leaflet. You may need to read it again. • If you have any further questions, ask your doctor or pharmacist. • This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours. • If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. See section 4. What is in this leaflet 1. What Sumatriptan tablet is and what it is used for 2. What you need to know before you use Sumatriptan tablet 3. How to use Sumatriptan tablet 4. Possible side effects 5. How to store Sumatriptan tablet 6. Contents of the pack and other information 1. What Sumatriptan tablet is and what it is used for Sumatriptan belongs to a group of medicines called serotonin receptor (5-HT1) agonists. Migraine headaches are thought to result from the dilatation of blood vessels. Sumatriptan constricts these blood vessels, thus relieving the migraine headache. Sumatriptan tablet is used to treat migraine attacks with or without aura (aura is a premonition usually connected with flashes of light, serrated images, stars or waves). 2. What you need to know before you use Sumatriptan tablet Do not use Sumatriptan tablet: - if you are allergic to sumatriptan or any of the other ingredients of this medicine (listed in section 6). -
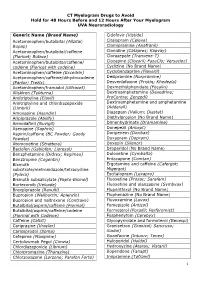
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology Generic Name (Brand Name) Cidofovir (Vistide) Acetaminophen/butalbital (Allzital; Citalopram (Celexa) Bupap) Clomipramine (Anafranil) Acetaminophen/butalbital/caffeine Clonidine (Catapres; Kapvay) (Fioricet; Butace) Clorazepate (Tranxene-T) Acetaminophen/butalbital/caffeine/ Clozapine (Clozaril; FazaClo; Versacloz) codeine (Fioricet with codeine) Cyclizine (No Brand Name) Acetaminophen/caffeine (Excedrin) Cyclobenzaprine (Flexeril) Acetaminophen/caffeine/dihydrocodeine Desipramine (Norpramine) (Panlor; Trezix) Desvenlafaxine (Pristiq; Khedezla) Acetaminophen/tramadol (Ultracet) Dexmethylphenidate (Focalin) Aliskiren (Tekturna) Dextroamphetamine (Dexedrine; Amitriptyline (Elavil) ProCentra; Zenzedi) Amitriptyline and chlordiazepoxide Dextroamphetamine and amphetamine (Limbril) (Adderall) Amoxapine (Asendin) Diazepam (Valium; Diastat) Aripiprazole (Abilify) Diethylpropion (No Brand Name) Armodafinil (Nuvigil) Dimenhydrinate (Dramamine) Asenapine (Saphris) Donepezil (Aricept) Aspirin/caffeine (BC Powder; Goody Doripenem (Doribax) Powder) Doxapram (Dopram) Atomoxetine (Strattera) Doxepin (Silenor) Baclofen (Gablofen; Lioresal) Droperidol (No Brand Name) Benzphetamine (Didrex; Regimex) Duloxetine (Cymbalta) Benztropine (Cogentin) Entacapone (Comtan) Bismuth Ergotamine and caffeine (Cafergot; subcitrate/metronidazole/tetracycline Migergot) (Pylera) Escitalopram (Lexapro) Bismuth subsalicylate (Pepto-Bismol) Fluoxetine (Prozac; Sarafem) -

Actions of the 5-Hydroxytryptamine 1 Receptor Agonist Sumatriptan
36 Gut 1998;42:36–41 Actions of the 5-hydroxytryptamine 1 receptor agonist sumatriptan on interdigestive Gut: first published as 10.1136/gut.42.1.36 on 1 January 1998. Downloaded from gastrointestinal motility in man J Tack, B Coulie, A Wilmer, T Peeters, J Janssens Abstract Evidence has accumulated that enteric neu- Background—Pharmacological studies of ronal 5-hydroxytryptamine (5HT) receptors the enteric nervous system have shown the are involved in regulation of the MMC in ani- presence of several subtypes of 5-hydroxy- mals and in man.3–7 In vitro studies in the tryptamine (5HT) receptor, which might guinea pig showed that 5HT4 receptors, 5HT3 be involved in control of the migrating receptors, 5HT1A receptors, and 5HT1P recep- motor complex. tors are present on myenteric neurones in the Aims—To study the eVect of sumatriptan, stomach and in the small intestine.8–11 Using an agonist of enteric neuronal 5HT1P the selective 5HT3 receptor antagonist on- receptors, on interdigestive motility in dansetron, we showed that 5HT3 receptors are man. involved in the initiation of gastric phase 3 12 Subjects and methods—In 12 healthy sub- motor activity in man. Lack of suitable jects, interdigestive motility was recorded ligands precluded a similar study for 5HT1 manometrically in the upper gastrointes- receptors and for 5HT4 receptors. Recently, tinal tract. In seven subjects blood sam- however, we showed that sumatriptan, a 5HT1 ples were drawn every 15 minutes for receptor agonist which is used in the treatment 13 radioimmunoassay of motilin and soma- of migraine in man, is an agonist of 5HT1P tostatin. -

Guideline for Preoperative Medication Management
Guideline: Preoperative Medication Management Guideline for Preoperative Medication Management Purpose of Guideline: To provide guidance to physicians, advanced practice providers (APPs), pharmacists, and nurses regarding medication management in the preoperative setting. Background: Appropriate perioperative medication management is essential to ensure positive surgical outcomes and prevent medication misadventures.1 Results from a prospective analysis of 1,025 patients admitted to a general surgical unit concluded that patients on at least one medication for a chronic disease are 2.7 times more likely to experience surgical complications compared with those not taking any medications. As the aging population requires more medication use and the availability of various nonprescription medications continues to increase, so does the risk of polypharmacy and the need for perioperative medication guidance.2 There are no well-designed trials to support evidence-based recommendations for perioperative medication management; however, general principles and best practice approaches are available. General considerations for perioperative medication management include a thorough medication history, understanding of the medication pharmacokinetics and potential for withdrawal symptoms, understanding the risks associated with the surgical procedure and the risks of medication discontinuation based on the intended indication. Clinical judgement must be exercised, especially if medication pharmacokinetics are not predictable or there are significant risks associated with inappropriate medication withdrawal (eg, tolerance) or continuation (eg, postsurgical infection).2 Clinical Assessment: Prior to instructing the patient on preoperative medication management, completion of a thorough medication history is recommended – including all information on prescription medications, over-the-counter medications, “as needed” medications, vitamins, supplements, and herbal medications. Allergies should also be verified and documented. -

United States Patent 19 11) Patent Number: 5,387,604 Mcdonald Et Al
US005387604A United States Patent 19 11) Patent Number: 5,387,604 McDonald et al. 45 Date of Patent: Feb. 7, 1995 (54) 14 BENZODIOXIN DERIVATIVES AND pp. 323-330, Role of 5HT1-like receptors in the reduc THEIR USE AS SEROTONIN 5HT tion of porcine cranial arteriovenous anastomotic shunt AGONSTS ing by Sumatriptan. 75 Inventors: Ian A. McDonald, Loveland; Ronald Saxena, et al, TiPS, May 1989, vol. 10, pp. 200-204; C. Bernotas, Cincinnati; Mark W. 5HT1-like receptor agonists and the pathophysiology Dudley, Somerville; Jeffrey S. of migraine. Sprouse, Cincinnati, all of Ohio Hamel et al., Br. J. Pharmacol, (1991), 102, pp. 227-233; Contractile 5HT1 receptors in human isolated pial arte 73) Assignee: Merrell Dow Pharmaceuticals Inc., rioles: correlation with 5-HT1D binding sites. Cincinnati, Ohio Edward E. Schwiezer, et al.; Psychopharmacology Bul 21 Appl. No.: 962,434 leting, vol. 22, No. 1, 1986, pp. 183-185; Open Trial of Buspirone in the Treatment of Major Depressive Disor 22 Filed: Oct. 16, 1992 der. Related U.S. Application Data European Journal of Pharmacology, 180 (1990) pp. 339-349, Dreteler, et al.; Comparison of the cardiovas 60 Division of Ser. No. 735,700, Jul. 30, 1991, Pat. No. cular effects of the 5-HT1A receptor agonist flexinoxan 5,189,179, which is a continuation-in-part of Ser. No. with that of 8-OH-DPAT in the rat. 574,710, Aug. 29, 1990, abandoned. European Journal of Pharmacology, 182 (1990) 63-72, 51) int. C.6 .................. C07D 319/20: A61K 31/335. Connor, et al.; Cardiovascular effects of 5HT1 receptor 52 U.S. -

Test Purchase of New Synthetic Tryptamines Via the Internet: Identity Check by GC-MS and Separation by HPLC
Journal of Applied Pharmaceutical Science Vol. 6 (01), pp. 028-034, January, 2016 Available online at http://www.japsonline.com DOI: 10.7324/JAPS.2016.600105 ISSN 2231-3354 Test purchase of new synthetic tryptamines via the Internet: Identity check by GC-MS and separation by HPLC Magdalena Taschwer, Edith Ebner, Martin G Schmid* Department of Pharmaceutical Chemistry, Institute of Pharmaceutical Sciences, University of Graz, Universitätsplatz 1, A-8010 Graz, Austria. ABSTRACT ARTICLE INFO Article history: Over the past few years, a continuous alteration of the recreational drug market took place. Among other novel Received on: 25/09/2015 psychoactive drugs, new synthetic tryptamine derivatives appeared on the market. These compounds are mainly Revised on: 18/10/2015 traded via the Internet, which has become an important marketplace for the sale of recreational drugs. The goal of Accepted on: 08/11/2015 our research was to check, if 13 new synthetic tryptamines obtained by test purchase via different online vendors Available online: 26/01/2016 meet the promised identity. Analysis was performed by GC-MS, using a common 30 m HP-5MS capillary column as stationary phase. Subsequently, a simple HPLC method for the separation of these tryptamines was Key words: developed. Therefore, the aim was to establish a method to separate a broad spectrum of trypamines Tryptamines, Legal highs, simultaneously within short time. Measurements were performed by a LiChrospher® RP-18e column and a Novel psychoactive drugs, mobile phase consisting of 0.1% triethylammonium acetate buffer, methanol and acetonitrile. Both presented HPLC, GC-MS. methods were found to be suitable for the identification as well as separation of tryptamines as the analysis times were short and the selectivity sufficient.