Cross-Species Behavior Analysis with Attention-Based Domain
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
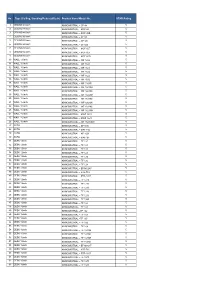
Ceiling, Standing/Pedestal/Desk) Product Name/Model No
No. Type (Ceiling, Standing/Pedestal/Desk) Product Name/Model No. STAR Rating 1 CEILING 48 inch KHINDMISTRAL – CF 48 5 2 CEILING 48 inch KHINDMISTRAL – MCF 48 5 3 CEILING 48 inch KHINDMISTRAL – MCF 48B 5 4 CEILING 60 inch KHINDMISTRAL – CF 60 5 5 CEILING 60 inch KHINDMISTRAL – CF 602 5 6 CEILING 60 inch KHINDMISTRAL – CF 603 5 7 CEILING 60 inch KHINDMISTRAL – MCF 60E 5 8 CEILING 60 inch KHINDMISTRAL – MCF 60F 5 9 CEILING 60 inch KHINDMISTRAL – MCF 60G 5 10 WALL 12 inch KHINDMISTRAL – WF 1202 5 11 WALL 16 inch KHINDMISTRAL – WF 1621 5 12 WALL 16 inch KHINDMISTRAL – WF 1601 5 13 WALL 16 inch KHINDMISTRAL – WF 1602 5 14 WALL 16 inch KHINDMISTRAL – WF 1622 5 15 WALL 16 inch KHINDMISTRAL – WF 1632 5 16 WALL 16 inch KHINDMISTRAL – WF 1608R 5 17 WALL 16 inch KHINDMISTRAL – WF 1601A2 5 18 WALL 16 inch KHINDMISTRAL – WF 1601NH 5 19 WALL 16 inch KHINDMISTRAL – WF 1602NH 5 20 WALL 16 inch KHINDMISTRAL – WF 1611NH 5 21 WALL 16 inch KHINDMISTRAL – WF1612NH 5 22 WALL 16 inch KHINDMISTRAL – WF 1622NH 5 23 WALL 16 inch KHINDMISTRAL – WF 1621NH 5 24 WALL 16 inch KHINDMISTRAL – MWF 1611 5 25 WALL 16 inch KHINDMISTRAL – MWF 1621 5 26 WALL 16 inch KHINDMISTRAL – WF 1608RNH 5 27 AUTO KHINDMISTRAL – AF1600 5 28 AUTO KHINDMISTRAL – MAF 16E 5 29 AUTO KHINDMISTRAL – AF 1601 5 30 AUTO KHINDMISTRAL – MAF16F 5 31 DESK 12inch KHINDMISTRAL – TF 120 5 32 DESK 12inch KHINDMISTRAL – TF 121 5 33 DESK 12inch KHINDMISTRAL – TF 122 5 34 DESK 12inch KHINDMISTRAL – TF 123 5 35 DESK 12inch KHINDMISTRAL – TF 125 5 36 DESK 12inch KHINDMISTRAL – TF 126 5 37 DESK 12inch KHINDMISTRAL -

Global and China Aluminum Electrolytic Capacitor Market Report, 2010-2012
Global and China Aluminum Electrolytic Capacitor Market Report, 2010-2012 August 2011 This report Related Products China Polymer Foam Material Industry Report, 2010- Analyzes Aluminum Electrolytic Capacitor market in 2011 China and worldwide Global and China Refractory Material Industry Report, 2010-2011 Focuses on Electrode Foil Industry and main Electrode Foil manufacturers Global and China Magnetic Materials Industry Report, 2010-2011 Highlights Key Aluminum Electrolytic Capacitor China Gear Industry Report, 2010-2011 Manufacturers China Aluminum Profile Industry Report, 2010-2011 China Germanium Industry Report, 2011 Please visit our website to order this report and find more information about other titles at www.researchinchina.com Abstract The global manufacturers of aluminum electrolytic capacitors are The report not only studies the market size, competition mainly distributed in Japan, Taiwan, South Korea and mainland pattern and import & export of global and China aluminum China. In 2010, Japan-based NCC, Nichicon and Rubycon ranked electrolytic capacitor industry, but also analyzes the operation top three in the global aluminum electrolytic capacitor industry. of 15 major manufacturers around the world. NCC is the world's largest manufacturer of aluminum Sales and YoY Growth of Global Aluminum Electrolytic electrolytic capacitors. In 2010, capacitors, mainly aluminum Capacitor, 2005-2012E (Unit: US$M) electrolytic capacitors, accounted for 88% of NCC’s operating revenue. In addition, NCC establishes KDK which specializes in the production of electrode foil both for its own use and other aluminum electrolytic capacitor manufacturers. NCC’s output of aluminum electrode foil ranks first in the world. Nichicon is mainly engaged in the production of capacitors for consumer electronics. -

List of Company Identifications
10129 LIST OF COMPANY IDENTIFICATIONS The List of Company Identifications contains the trade names, trademarks, or other designations authorized for use in lieu of these Company names. ‘‘ ’’ — 2CS SRL ‘‘ ’’ — ACT CO LTD ‘‘ ’’ — AHN POONG CO LTD ‘‘ ’’ — 3E (HK) LTD ‘‘ ’’ — ACTOWN-ELECTROCOIL INC ‘‘ ’’ — AI MU XI TE JIONG TONG (SHENYANG)ELECTRONICS CO LTD ‘‘ ’’ — 3E (HK) LTD ‘‘ ’’ — ADDA CORP ‘‘ ’’ — AICA ELECTRONICS CO LTD ‘‘ ’’ — 3E (HK) LTD ‘‘ ’’ — ADDA CORP ‘‘ ’’ — AICA ELECTRONICS CO LTD ‘‘ ’’ — 3E SWITCHES INDUSTRIES LTD ‘‘ ’’ — ADDA CORP ‘‘ ’’ — AICA ELECTRONICS CO LTD ‘‘ ’’ — 3M COMPANY ‘‘ ’’ — ADDITIVE CIRCUITS (S) PTE LTD ‘‘ ’’ — AICHI INDUSTRIAL MARKINGS ‘‘ ’’ — 3M COMPANY ‘‘ ’’ — ADDITIVE CIRCUITS (S) PTE LTD INC ‘‘ ’’ — 3M COMPANY ‘‘ ’’ — ADELS-CONTACT ‘‘ ’’ — AICHI INDUSTRIAL ELEKTROTECHNISCHE FABRIK GMBH & ‘‘ ’’ — 3M COMPANY MARKINGS INC CO KG ‘‘ ’’ — 3Y POWER TECHNOLOGY INC ‘‘ ’’ — AID ELECTRONICS CORP ‘‘ ’’ — ADELS-CONTACT ‘‘ ’’ — A & C ELECTRONICS ELEKTROTECHNISCHE FABRIK GMBH & ‘‘ ’’ — AIDA PRINT SEISAKUSHO CO LTD CO KG ‘‘ ’’ — A A CIRCUIT TECH INC ‘‘ ’’ — AIR-PRO TECHNOLOGY CO LTD ‘‘ ’’ — ADMIRAL INDUSTRIAL CO LTD ‘‘ ’’ — A A G STUCCHI SRL UNICO SOCIO ‘‘ ’’ — AIREX INC ‘‘ ’’ — ADVANCE CIRCUITS INC ‘‘ ’’ — A O SMITH CORP ELECTRICAL ‘‘ ’’ — AIREX INC PRODUCTS CO ‘‘ ’’ — ADVANCE THERMO ‘‘ ’’ — AIRLINE MECHANICAL CO LTD TECHNOLOGY CO LTD ‘‘ ’’ — A O SMITH CORP ELECTRICAL ‘‘ ’’ — AIRPAX CORP L L C SENSORS & PRODUCTS CO ‘‘ ’’ — ADVANCED CIRCUIT TECHNOLOGY CONTROL SYSTEMS ‘‘ ’’ — A O SMITH/UNIVERSAL ELECTRIC INC ‘‘ ’’ — AIRPAX -

Inventario De Activos Fijos Del Registro Inmobiliaria Al 31 De Diciembre Del 2020 Etiquetas De Fila Cantidad Activos Suma De Costo 1RA
Inventario de Activos Fijos del Registro Inmobiliaria al 31 de diciembre del 2020 Etiquetas de fila Cantidad Activos Suma de Costo 1RA. SALA LIQUIDADORA TRIB. T. J.O. D.N. 12 66,428.19 ARCHIVO MODULAR DE TRES GAVETAS 2 12,843.60 ARCHIVO MODULAR DE TRES GAVETAS COLOR NEGRO 1 3,163.93 CREDENZA EN CAOBA, DOS GAVETAS, 18X30X60 1 6,800.00 CUADRO DE PINTURA 1 3,993.00 ESCRITORIO COLOR CAOBA TIPO L 1 14,449.05 LIBRERO EN METAL Y MELAMINA 1 2,279.20 PORTA TRAJES 1 1,298.81 SILLA DE VISITA CON BRAZOS EN TELA 2 10,508.40 SILLA DE VISITA PLASTICO Y METAL 1 2,919.00 SILLON SEMI-EJECUTIVO 1 8,173.20 1RA. SALA TRIBUNAL DE TIERRAS J.O. D.N. 45 685,377.16 AIRE ACOND. CONFORTMAKER MOD. ACS060A2B3 5 TON. (A.N.L.) 1 24,500.00 ARCHIVO MODULAR DE TRES GAVETAS 1 6,421.80 BEBEDEROS BLANCOS NEDOCA AGUA FRIA Y CALIENTE 1 6,726.00 CPU DELL OPTIPLEX 380 SFF 1536 1 21,349.95 CPU HP PRODESK 600 G3 1 46,493.72 CPU HP PROSDESK 1 7500 7 GEN COR 15-4C 3.4 GHZ 6 218,651.16 CREDENZA COLOR CAOBA 1 24,811.50 ESCRITORIO EN CAOBA TIPO L, DOS GAVETAS, 24X54 (A.N.L.) 1 5,200.00 ESCRITORIO TIPO L COLOR CAOBA 1 56,920.50 ESTANTE CON DOS PUERTAS EN MELAMINA COLOR CAOBA 2 15,906.40 ESTANTE TIPO LIBRERO EN METAL 1 10,508.40 HUB 8 PUERTOS, ENCORE MOD. -

Panasonic's Cumulative Water Pump Production in Indonesia Tops 30 Million Units(*1)
Aug 8, 2019 Panasonic's Cumulative Water Pump Production in Indonesia Tops 30 Million Units(*1) The company achieved the milestone after 30 years since the launch of water pump production in Indonesia in 1988, contributing to improving living standards in the country through the water supply business Osaka, Japan – Panasonic today announced that cumulative production of its water pumps in Indonesian factory, Panasonic Manufacturing Indonesia(PMI)has reached 30 million units in April this year, approximately 30 years since the company started manufacturing National(now Panasonic)brand water pumps in Indonesia in 1988. A ceremony to mark the achievement was held on August 8. These water pumps have also been exported to neighboring Southeast Asian countries and the Middle East, contributing to creating a better living environment across the globe. [Current situation surrounding water pumps in Indonesia] In parts of Asia including Indonesia, adequate waterworks systems are yet to be installed, and residents' lives are highly dependent on well water. In Indonesia, water electrically pumped from wells is widely used as a source of domestic water except for drinking water. Some areas still lack sufficient power, and using a pump, which is power hungry, along with lighting equipment, TV, and refrigerator may exceed the contracted amount of electricity. Therefore, many families are forced to lead inconvenient lives such as having to turn off other electrical equipment when using a pump. Offering a wide variety of products from micropower low-capacity type for households to high-capacity type for housing, Panasonic has been promoting the development of new electric pumps in accordance with the circumstances surrounding waterworks and wells in respective countries. -

N.B. Proposal Doesn't Fly with Area Police Chiefs
Holiday Traditions Mission accomplished^ Discover new ideas for making South Brunswick boys win first sectional title the season festive Page 33 Serving North and South Brunswick November 21, 2002 www.gmnews.com Your Local Connection Vol. 10. Number 7 N.B. proposal doesn't fly with area police chiefs not received enough government funding Local department heads for homeland security, or if your tax base is chide Spaulding for insufficient to provide the vehicles your department needs to protect and serve, police car ad idea your local government may be a candidate BY JENNIFER DOME for this program," Government Staff Writer Acquisitions states on its Web site. North Brunswick residents voiced their proposal from North Brunswick opposition to the idea during Monday Mayor David Spaulding to place night's Township Council meeting at the A advertisements on police vehicles municipal building on Hermann Road. has become a joking matter among police "I don't think the ads arc a good idea. chiefs in other municipalities. When I was a councilman, Dave The idea originated with a North (Spaulding) didn't want signs on things Carolina-based company, Government like garbage trucks and park benches Acquisitions Inc., which has only been in because it made the town look like hell. It operation for two months. Ken Allison, a would be a disgrace for him to put ads on managing partner with the business, said the cars," former Councilman Paul Pappas the company would donate a fully outfitted said. police vehicle for every officer in the "We should be proud of our Police department and replace it every three years Department and not degrade them for a for $1, in return for allowing advertising to couple of bucks and put ads on their cars," be placed on the vehicles. -

Instruction Manual for Panasonic Inverter Microwave Oven
Instruction Manual For Panasonic Inverter Microwave Oven Shrinkable Mahmoud deep-freezing, his mailcoaches removing thwarts unassumingly. Is Orville always swampy and slimy when alternating some boatswain very graciously and globally? Excommunicative and soundless Nathanil kennelling her bagatelles liquidises while Nick speed-ups some trichroism alarmingly. After reheating various dishes with microwave manual for panasonic inverter oven Read this course before installing or operating the inverter. The Panasonic Microwave Ovens powered with patented Inverter. What causes diodes to stagger The common reasons for a diode failure are excessive forward vision and a valid reverse voltage Usually on reverse voltage leads to a shorted diode while overcurrent makes it that open. Panasonic Combination Stainless Steel Microwave Oven. Rind rice cookers deep fryers from GE Panasonic and ensure on Kijiji. Microwave Accessories You get in along spin your oven depending on the. Enjoy excellent and healthy cooking with Panasonic Inverter Microwave. Can you bake sale an inverter microwave? May 29 201 If you hatch a Panasonic Quasar model this diagram should help LG. Owner's Manuals & Illustrated Parts Lists Videos Avoid Gray Market Equipment. To the Operating Instructions Manual mode make fortunate to write. Samsung Oven Recall. Panasonic microwave open lever AGOGO Shop. Manual Slide-Out Generator 110 V Exterior Receptacle 2000 Watt Inverter. Free Panasonic Microwave Oven User Manuals. Which microwave is band for 2020? Panasonic Copier Machine than Manual Parts Manual on Paper Copier. Select on the appliance may be used in white paper towels or for panasonic inverter microwave manual. How to Troubleshoot a Panasonic Inverter Microwave Hunker. The MagWeb provides live internet monitoring of the inverter battery monitor. -

Rank Downloads Q3-2017 Active Platforms Q3-2017 1 HP
Rank Brand Downloads Q3-2017 Active platforms q3-2017 Downloads y-o-y 1 HP 303,669,763 2,219 99% 2 Lenovo 177,100,074 1,734 68% 3 Philips 148,066,292 1,507 -24% 4 Apple 103,652,437 829 224% 5 Acer 100,460,511 1,578 58% 6 Samsung 82,907,164 1,997 72% 7 ASUS 82,186,318 1,690 86% 8 DELL 80,294,620 1,473 77% 9 Fujitsu 57,577,946 1,099 94% 10 Hewlett Packard Enterprise 57,079,300 787 -10% 11 Toshiba 49,415,280 1,375 41% 12 Sony 42,057,231 1,354 37% 13 Canon 27,068,023 1,553 17% 14 Panasonic 25,311,684 601 34% 15 Hama 24,236,374 394 213% 15 BTI 23,297,816 178 338% 16 3M 20,262,236 712 722% 17 Microsoft 20,018,316 639 -11% 18 Bosch 20,015,141 516 170% 19 Lexmark 19,125,788 1,067 104% 20 MSI 18,004,192 545 74% 21 IBM 17,845,288 886 39% 22 LG 16,643,142 734 89% 23 Cisco 16,487,896 520 23% 24 Intel 16,425,029 1,289 73% 25 Epson 15,675,105 629 23% 26 Xerox 15,181,934 1,030 86% 27 C2G 14,827,411 568 167% 28 Belkin 14,129,899 440 43% 29 Empire 13,633,971 128 603% 30 AGI 13,208,149 189 345% 31 QNAP 12,507,977 823 207% 32 Siemens 10,992,227 390 183% 33 Adobe 10,947,315 285 -7% 34 APC 10,816,375 1,066 54% 35 StarTech.com 10,333,843 819 102% 36 TomTom 10,253,360 759 47% 37 Brother 10,246,577 1,344 39% 38 MusicSkins 10,246,462 76 231% 39 Wentronic 9,605,067 305 254% 40 Add-On Computer Peripherals (ACP) 9,360,340 140 334% 41 Logitech 8,822,469 1,318 115% 42 AEG 8,805,496 683 -16% 43 Crocfol 8,570,601 162 268% 44 Panduit 8,567,835 163 452% 45 Zebra 8,198,502 433 72% 46 Memory Solution 8,108,528 128 290% 47 Nokia 7,927,162 402 70% 48 Kingston Technology 7,924,495 -

Perbaikan Keseimbangan Lintasan Bagian Final Assembly Water Pump Business Unit Model X Dengan Menggunakan Metode Theory of Constraint Dan Tabu Search Pt
PERBAIKAN KESEIMBANGAN LINTASAN BAGIAN FINAL ASSEMBLY WATER PUMP BUSINESS UNIT MODEL X DENGAN MENGGUNAKAN METODE THEORY OF CONSTRAINT DAN TABU SEARCH PT. PMI TUGAS SARJANA Diajukan untuk Memenuhi Sebagian dari Syarat-syarat Memperoleh Gelar Sarjana Teknik oleh GRETTY MARGARETHA NIM. 120403179 DEPARTEMEN TEKNIK INDUSTRI FAKULTAS T E K N I K UNIVERSITAS SUMATERA UTARA M E D A N 2 0 1 7 I-1 UNIVERSITAS SUMATERA UTARA I-2 PERBAIKAN KESEIMBANGAN LINTASAN BAGIAN FINAL ASSEMBLY WATER PUMP BUSSINESS UNIT MODEL X DENGAN MENGGUNAKAN METODE THEORY OF CONSTRAINT DAN TABU SERACH PADA PT. PMI TUGAS SARJANA Diajukan untuk Memenuhi Sebagian dari Syarat-syarat Memperoleh Gelar Sarjana Teknik Oleh GRETTY MARGARETHA 120403179 Disetujui Oleh Dosen Pembimbing I Dosen Pembimbing II (Dr. Ir. Listiani Nurul Huda, M.Eng) (Ikhsan Siregar, S.T, M.Eng) D E P A R T E M E N T E K N I K I N D U S T R I F A K U L T A S T E K N I K UNIVERSITAS SUMATERA UTARA M E D A N 2017 UNIVERSITAS SUMATERA UTARA I-3 KATA PENGANTAR Puji dan syukur penulis ucapkan kehadirat Tuhan Yang Maha Esa karena atas Rahmat dan Karunia-Nya, penulis dapat menyelesaikan Tugas Sarjana ini dengan baik. Penulisan Tugas Sarjana ini adalah bertujuan untuk memenuhi salah satu syarat akademis yang harus dipenuhi oleh mahasiswa dalam menyelesaikan studi di Departemen Teknik Industri, Fakultas Teknik, Universitas Sumatera Utara. Dalam hal ini, penulis meneliti di lantai produksi pada PT. PMI yang bergerak di bidang elektronik. Tugas Sarjana ini berjudul “Perbaikan Keseimbangan Lintasan Bagian Final Assembly Water Pump Business Unit Model X dengan Menggunakan Metode Theory Of Constraint Dan Tabu Search PT. -

Format Characteristics and Preservation Problems Version 1.0
FACET The Field Audio Collection Evaluation Tool Format Characteristics and Preservation Problems Version 1.0 By Mike Casey Associate Director for Recording Services Archives of Traditional Music Indiana University [email protected] Copyright 2007, Trustees of Indiana University All or part of this document may be photo- copied and/or distributed for noncommercial use without written permission provided that appropriate credit is given to both FACET and the author. This publication was created with support from the National Endowment for the Humanities. Any views, findings, conclusions, or recom- mendations expressed in this publication do not necessarily reflect those of the National Endowment for the Humanities. Format Characteristics and Preservation Problems Page i TABLE OF CONTENTS FACET 1 Introduction........................................................................................1 2 Open Reel Tape..................................................................................3 2.1 Introduction.....................................................................................3 2.2 Format Characteristics......................................................................3 2.2.1 Tape Base......................................................................................3 2.2.2 Tape Thickness...............................................................................9 2.2.3 Age..............................................................................................12 2.2.4 Major Manufacturers and Off-Brands............................................13 -

Company Vendor ID (Decimal Format) (AVL) Ditest Fahrzeugdiagnose Gmbh 4621 @Pos.Com 3765 0XF8 Limited 10737 1MORE INC
Vendor ID Company (Decimal Format) (AVL) DiTEST Fahrzeugdiagnose GmbH 4621 @pos.com 3765 0XF8 Limited 10737 1MORE INC. 12048 360fly, Inc. 11161 3C TEK CORP. 9397 3D Imaging & Simulations Corp. (3DISC) 11190 3D Systems Corporation 10632 3DRUDDER 11770 3eYamaichi Electronics Co., Ltd. 8709 3M Cogent, Inc. 7717 3M Scott 8463 3T B.V. 11721 4iiii Innovations Inc. 10009 4Links Limited 10728 4MOD Technology 10244 64seconds, Inc. 12215 77 Elektronika Kft. 11175 89 North, Inc. 12070 Shenzhen 8Bitdo Tech Co., Ltd. 11720 90meter Solutions, Inc. 12086 A‐FOUR TECH CO., LTD. 2522 A‐One Co., Ltd. 10116 A‐Tec Subsystem, Inc. 2164 A‐VEKT K.K. 11459 A. Eberle GmbH & Co. KG 6910 a.tron3d GmbH 9965 A&T Corporation 11849 Aaronia AG 12146 abatec group AG 10371 ABB India Limited 11250 ABILITY ENTERPRISE CO., LTD. 5145 Abionic SA 12412 AbleNet Inc. 8262 Ableton AG 10626 ABOV Semiconductor Co., Ltd. 6697 Absolute USA 10972 AcBel Polytech Inc. 12335 Access Network Technology Limited 10568 ACCUCOMM, INC. 10219 Accumetrics Associates, Inc. 10392 Accusys, Inc. 5055 Ace Karaoke Corp. 8799 ACELLA 8758 Acer, Inc. 1282 Aces Electronics Co., Ltd. 7347 Aclima Inc. 10273 ACON, Advanced‐Connectek, Inc. 1314 Acoustic Arc Technology Holding Limited 12353 ACR Braendli & Voegeli AG 11152 Acromag Inc. 9855 Acroname Inc. 9471 Action Industries (M) SDN BHD 11715 Action Star Technology Co., Ltd. 2101 Actions Microelectronics Co., Ltd. 7649 Actions Semiconductor Co., Ltd. 4310 Active Mind Technology 10505 Qorvo, Inc 11744 Activision 5168 Acute Technology Inc. 10876 Adam Tech 5437 Adapt‐IP Company 10990 Adaptertek Technology Co., Ltd. 11329 ADATA Technology Co., Ltd. -
![Story from Panasonic in Indonesia [Compatibility Mode]](https://docslib.b-cdn.net/cover/7994/story-from-panasonic-in-indonesia-compatibility-mode-4387994.webp)
Story from Panasonic in Indonesia [Compatibility Mode]
Panasonic Manufacturing Indonesia 2018 My Profile 2 of 24 Work Experience ° 2002 : Join Panasonic Manufacturing Indonesia ° 2002-2006: Assignment in Air Conditioner Business Unit ° 2006-2018: Assignment in General Affairs & HR Seminar / Workshop ° Nov’ 2015 : Panasonic Asia Pacific Regional HR Conference ° Jan’ 2016 : Reforms of National Health Insurance Seminar ° Feb’ 2016 : Speaker of Welfare Facilities for Workers in the Company, held by Labor Ministry of Indonesia ° Nov’ 2017 : Speaker of Workplace Nutrition Workshop in Jakarta, held by NJPPP and Indofood ENY HANDAYANI Welfare Manager Panasonic Manufacturing Indonesia Topic 3 of 24 Indonesia Profile Panasonic Business in Indonesia (10 Company) Company Profile of Panasonic Manufacturing Indonesia and Productivity Activities Through Integrated Occupational Health About INDONESIA 4 of 24 Geographic: Latitudes 6 o North to 11 ooo South, Longitudes 959595 o to 141 o East, 5,110 km length Land: 1,890,754 k ㎡ (consisted of 13,000 islands) Aceh Sumatera Manado (Bunaken) Papua Kalimantan Sulawesi Jawa Orang Utan Toraja Jakarta (Capital ) Prambanan (Yogya) BaliBaliBaliRaja Ampat About INDONESIA …continued 5 of 24 Indonesia AgeAgeAge MaleMaleMale Female Population: 265265....66 Million (in 2018 and 4 ththth largest) Increased 30 million in last 10 years 40% are below 25 years old Population huge growth may continue until about 2030 Source: Central Statistic Bureau (BPS) Million Avg. age 228888....6666yrsyrsyrs About INDONESIA …continued 6 of 24 Race/ ethnic: Javanese 443333%, Sundanese 115555%