A Companion to Xxth Century Physics
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

James Chadwick: Ahead of His Time
July 15, 2020 James Chadwick: ahead of his time Gerhard Ecker University of Vienna, Faculty of Physics Boltzmanngasse 5, A-1090 Wien, Austria Abstract James Chadwick is known for his discovery of the neutron. Many of his earlier findings and ideas in the context of weak and strong nuclear forces are much less known. This biographical sketch attempts to highlight the achievements of a scientist who paved the way for contemporary subatomic physics. arXiv:2007.06926v1 [physics.hist-ph] 14 Jul 2020 1 Early years James Chadwick was born on Oct. 20, 1891 in Bollington, Cheshire in the northwest of England, as the eldest son of John Joseph Chadwick and his wife Anne Mary. His father was a cotton spinner while his mother worked as a domestic servant. In 1895 the parents left Bollington to seek a better life in Manchester. James was left behind in the care of his grandparents, a parallel with his famous predecessor Isaac Newton who also grew up with his grandmother. It might be an interesting topic for sociologists of science to find out whether there is a correlation between children educated by their grandmothers and future scientific geniuses. James attended Bollington Cross School. He was very attached to his grandmother, much less to his parents. Nevertheless, he joined his parents in Manchester around 1902 but found it difficult to adjust to the new environment. The family felt they could not afford to send James to Manchester Grammar School although he had been offered a scholarship. Instead, he attended the less prestigious Central Grammar School where the teaching was actually very good, as Chadwick later emphasised. -

Chemists and Physicists Behaving Badly: the Shadow Side of Two Elemental Discoveries Volume 23, Issue 3 (2020), P
Comptes Rendus Chimie Marco Fontani, Mary Virginia Orna and Mariagrazia Costa Chemists and physicists behaving badly: The shadow side of two elemental discoveries Volume 23, issue 3 (2020), p. 231-241. <https://doi.org/10.5802/crchim.1> Part of the Thematic Issue: Variations around the Periodic Table Guest editors: Pierre Braunstein (Université de Strasbourg, Académie des sciences) and Robert Guillaumont (Académie des sciences) © Académie des sciences, Paris and the authors, 2020. Some rights reserved. This article is licensed under the Creative Commons Attribution 4.0 International License. http://creativecommons.org/licenses/by/4.0/ Les Comptes Rendus. Chimie sont membres du Centre Mersenne pour l’édition scientifique ouverte www.centre-mersenne.org Comptes Rendus Chimie 2020, 23, nO 3, p. 231-241 https://doi.org/10.5802/crchim.1 Variations around the Periodic Table/ Variations autour du tableau périodique Chemists and physicists behaving badly: The shadow side of two elemental discoveries Des chimistes, et leurs mauvaises habitudes , a b a Marco Fontani¤ , Mary Virginia Orna and Mariagrazia Costa a Dipartimento di Chimica “Ugo SchiV”, Università degli Studi di Firenze, Italy b College of New Rochelle, New Rochelle, NY, USA E-mails: marco.fontani@unifi.it (M. Fontani), [email protected] (M. V. Orna) Abstract. It is appropriate to recall that 2019 was the year dedicated to the Periodic Table. But when we speak about false elements – in the aftermath of the celebrations marking this year, – we are greeted most warmly, but with some puzzlement, as to how it came to mind to celebrate “Mendeleev’s creature” in such a peculiar way, that is, by commemorating elements that never existed. -
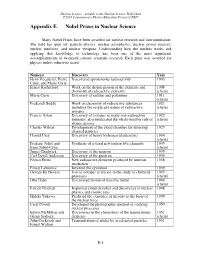
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

How Basic Science Drives Technological Progress — and Vice Versa —
How Basic Science Drives Technological Progress | And Vice Versa | Sheldon Lee Glashow Metcalf Professor of Science & Mathematics Boston University Higgins Professor of Physics, Emeritus Harvard University February, 2017 BRIDGES Dialogues Toward a Culture of Peace Discoveries Can Be Intentional or Accidental Some technological advances, such as X-Rays and Penicillin, arose from research that was unplanned and not directed toward any specific goal. These discoveries were unexpected and stumbled upon by accident. Others, like Streptomycin and Nuclear Weapons, resulted from carefully planned and specifically targeted research. The History of Science proves both methods to be essential... ... a fact that must be borne in mind by aspiring scientists and by governmental, academic and industrial agencies seeking to foster scientific and technological progress. Immanuel Kant Versus The Princes of Serendip Some scientists focus on well-defined goals: first they lay careful plans, then they look. They follow the so-called scientific method. I call this the Kantian approach to discovery. Others have more fun: They look & listen to Nature with open minds, and sometimes discover amazing things. I call this the Serendipitous approach, like Columbus's `discovery' of America but unlike Magellan's successful plan to circumnavigate the globe. The two approaches often mix. Many Kantial efforts yield surprisimg discoveries. TNT was synthesized in 1863 and used as a yellow dye for 28 years until its value as an explosive was recognized. THALIDOMIDE, originally a sedative in the 1950s, led to a medical disaster. Much later it was found effective for treating cancer and leprosy! Pure Science Versus Applied Science Basic Research can proceed either by Kantian (intentional) or Serendipitous (accidental) means, but there is a second dichotomy. -

Lise Meitner 1878 – 1968
Discoveries that changed the world: 1932 – 1942 James Chadwick 1891 – 1974 Lise Meitner 1878 – 1968 I „The road to the neutron“ Staff and research students at the Cavendish Laboratory, Cambridge, 1923. (Names from left to right. Front row: J. Chadwick, G. Stead, F.W. Aston, Prof. Sir J. J. Thomson, Prof. Sir E. Rutherford, J.A. Crowther, Miss B. Trevelyan, G.I. Taylor, Second row: P. Kapitza, H. de W. Smyth, T. Alty, J.E. Crackston, H. Robinson, L.F. Curtiss, E.S. Bieler, A.G.D. West, P. Mercier. Back row: P.M.S. Blackett, R.E. Clay, H.W.B. Skinner, H.D. Griffith, A.W. Barton, L.F. Bates, J.S. Rogers, K.G. Emeleus.) The room which Rutherford and Chadwick used for their scattering experiments in the 1920s. The work was carried out in the dark, often to the accompaniment of Rutherford singing „Onward Christian Soldiers“. Rutherford had already proposed the neutron in 1920 in his Bakerian Lecture at the Royal Society. He talked about a “neutral doublet” (at that time considered a proton and electron) that could be difficult to detect and move easily through matter. Curie & Joliot published (incorrectly) in Jan. 1932 the observation: 9Be + 4He → 12C + 1n I. Curie and F. Joliot, C. R. Acad. Sci. Paris 194, 273 (1932) When the radiation was passed through wax the ionisation increased! This increase was due to knock-on protons. To explain this the Curie’s suggested that the emission was of a 55 MeV γ ray, an energy much greater than anything yet seen! Moreover, the radiation also passed through lead This experiment was first performed in 1930 by Walter Bothe and Herbet Becker at U. -

James Chadwick and E.S
What is the Universe Made Of? Atoms - Electrons Nucleus - Nucleons Antiparticles And ... http://www.parentcompany.com/creation_explanation/cx6a.htm What Holds it Together? Gravitational Force Electromagnetic Force Strong Force Weak Force Timeline - Ancient 624-547 B.C. Thales of Miletus - water is the basic substance, knew attractive power of magnets and rubbed amber. 580-500 B.C. Pythagoras - Earth spherical, sought mathematical understanding of universe. 500-428 B.C. Anaxagoras changes in matter due to different orderings of indivisible particles (law of the conservation of matter) 484-424 B.C. Empedocles reduced indivisible particles into four elements: earth, air, fire, and water. 460-370 B.C. Democritus All matter is made of indivisible particles called atoms. 384-322 B.C. Aristotle formalized the gathering of scientific knowledge. 310-230 B.C. Aristarchus describes a cosmology identical to that of Copernicus. 287-212 B.C. Archimedes provided the foundations of hydrostatics. 70-147 AD Ptolemy of Alexandria collected the optical knowledge, theory of planetary motion. 1214-1294 AD Roger Bacon To learn the secrets of nature we must first observe. 1473-1543 AD Nicholaus Copernicus The earth revolves around the sun Timeline – Classical Physics 1564-1642 Galileo Galilei - scientifically deduced theories. 1546-1601, Tycho Brahe accurate celestial data to support Copernican system. 1571-1630, Johannes Kepler. theory of elliptical planetary motion 1642-1727 Sir Isaac Newton laws of mechanics explain motion, gravity . 1773-1829 Thomas Young - the wave theory of light and light interference. 1791-1867 Michael Faraday - the electric motor, and electromagnetic induction, electricity and magnetism are related. electrolysis, conservation of energy. -

Hitler's Uranium Club, the Secret Recordings at Farm Hall
HITLER’S URANIUM CLUB DER FARMHALLER NOBELPREIS-SONG (Melodie: Studio of seiner Reis) Detained since more than half a year Ein jeder weiss, das Unglueck kam Sind Hahn und wir in Farm Hall hier. Infolge splitting von Uran, Und fragt man wer is Schuld daran Und fragt man, wer ist Schuld daran, So ist die Antwort: Otto Hahn. So ist die Antwort: Otto Hahn. The real reason nebenbei Die energy macht alles waermer. Ist weil we worked on nuclei. Only die Schweden werden aermer. Und fragt man, wer ist Schuld daran, Und fragt man, wer ist Schuld daran, So ist die Antwort: Otto Hahn. So ist die Antwort: Otto Hahn. Die nuclei waren fuer den Krieg Auf akademisches Geheiss Und fuer den allgemeinen Sieg. Kriegt Deutschland einen Nobel-Preis. Und fragt man, wer ist Schuld daran, Und fragt man, wer ist Schuld daran, So ist die Antwort: Otto Hahn. So ist die Antwort: Otto Hahn. Wie ist das moeglich, fragt man sich, In Oxford Street, da lebt ein Wesen, The story seems wunderlich. Die wird das heut’ mit Thraenen lesen. Und fragt man, wer ist Schuld daran Und fragt man, wer ist Schuld daran, So ist die Antwort: Otto Hahn. So ist die Antwort: Otto Hahn. Die Feldherrn, Staatschefs, Zeitungsknaben, Es fehlte damals nur ein atom, Ihn everyday im Munde haben. Haett er gesagt: I marry you madam. Und fragt man, wer ist Schuld daran, Und fragt man, wer ist Schuld daran, So ist die Antwort: Otto Hahn. So ist die Antwort: Otto Hahn. Even the sweethearts in the world(s) Dies ist nur unsre-erste Feier, Sie nennen sich jetzt: “Atom-girls.” Ich glaub die Sache wird noch teuer, Und fragt man, wer ist Schuld daran, Und fragt man, wer ist Schuld daran, So ist die Antwort: Otto Hahn. -

Some Famous Physicists Ing Weyl’S Book Space, Time, Matter, the Same Book That Heisenberg Had Read As a Young Man
Heisenberg’s first graduate student was Felix Bloch. One day, while walking together, they started to discuss the concepts of space and time. Bloch had just finished read- Some Famous Physicists ing Weyl’s book Space, Time, Matter, the same book that Heisenberg had read as a young man. Still very much Anton Z. Capri† under the influence of this scholarly work, Bloch declared that he now understood that space was simply the field of Sometimes we are influenced by teachers in ways that, affine transformations. Heisenberg paused, looked at him, although negative, lead to positive results. This happened and replied, “Nonsense, space is blue and birds fly through to Werner Heisenberg1 and Max Born,2 both of whom it.” started out to be mathematicians, but switched to physics There is another version of why Heisenberg switched to due to encounters with professors. physics. At an age of 19, Heisenberg went to the University As a young student at the University of Munich, Heisen- of G¨ottingen to hear lectures by Niels Bohr.4 These lec- berg wanted to attend the seminar of Professor F. von tures were attended by physicists and their students from Lindemann,3 famous for solving the ancient problem of various universities and were jokingly referred to as “the squaring the circle. Heisenberg had Bohr festival season.” Here, Bohr expounded on his latest read Weyl’s book Space, Time, Matter theories of atomic structure. The young Heisenberg in the and, both excited and disturbed by the audience did not hesitate to ask questions when Bohr’s abstract mathematical arguments, had explanations were less than clear. -

Father of the Shell Model
CENTENARY Father of the shell model Heidelberg University held a symposium on fundamental physics and the shell model this summer to celebrate the 100th anniversary of the birth of Hans Jensen, the German who created the nuclear shell model with Maria Goeppert-Mayer of Argonne. Hans Jensen (1907–1973) is the only theorist among the three winners from Heidelberg University of the Nobel Prize for Physics. He shared the award with Maria Goeppert-Mayer in 1963 for the development of the nuclear shell model, which they published independently in 1949. The model offered the first coherent expla- nation for the variety of properties and structures of atomic nuclei. In particular, the “magic numbers” of protons and neutrons, which had been determined experimentally from the stability properties and observed abundances of chemical elements, found a natural explanation in terms of the spin-orbit coupling of the nucleons. These numbers play a decisive role in the synthesis of the ele- ments in stars, as well as in the artificial synthesis of the heaviest elements at the borderline of the periodic table of elements. Hans Jensen was born in Hamburg on 25 June 1907. He studied physics, mathematics, chemistry and philosophy in Hamburg and Freiburg, obtaining his PhD in 1932. After a short period in the Hans Jensen in his study at 16 Philosophenweg, Heidelberg German army’s weather service, he became professor of theo- in 1963. (Courtesy Bettmann/UPI/Corbis.) retical physics in Hannover in 1940. Jensen then accepted a new chair for theoretical physics in Heidelberg in 1949 on the initiative Mayer 1949). -

Bruno Touschek in Germany After the War: 1945-46
LABORATORI NAZIONALI DI FRASCATI INFN–19-17/LNF October 10, 2019 MIT-CTP/5150 Bruno Touschek in Germany after the War: 1945-46 Luisa Bonolis1, Giulia Pancheri2;† 1)Max Planck Institute for the History of Science, Boltzmannstraße 22, 14195 Berlin, Germany 2)INFN, Laboratori Nazionali di Frascati, P.O. Box 13, I-00044 Frascati, Italy Abstract Bruno Touschek was an Austrian born theoretical physicist, who proposed and built the first electron-positron collider in 1960 in the Frascati National Laboratories in Italy. In this note we reconstruct a crucial period of Bruno Touschek’s life so far scarcely explored, which runs from Summer 1945 to the end of 1946. We shall describe his university studies in Gottingen,¨ placing them in the context of the reconstruction of German science after 1945. The influence of Werner Heisenberg and other prominent German physicists will be highlighted. In parallel, we shall show how the decisions of the Allied powers, towards restructuring science and technology in the UK after the war effort, determined Touschek’s move to the University of Glasgow in 1947. Make it a story of distances and starlight Robert Penn Warren, 1905-1989, c 1985 Robert Penn Warren arXiv:1910.09075v1 [physics.hist-ph] 20 Oct 2019 e-mail: [email protected], [email protected]. Authors’ ordering in this and related works alternates to reflect that this work is part of a joint collaboration project with no principal author. †) Also at Center for Theoretical Physics, Massachusetts Institute of Technology, USA. Contents 1 Introduction2 2 Hamburg 1945: from death rays to post-war science4 3 German science and the mission of the T-force6 3.1 Operation Epsilon . -

The Beginning of the Nuclear Age
The Beginning of the Nuclear Age M. SHIFMAN 1 Theoretical Physics Institute, University of Minnesota 1 Introduction A few years ago I delivered a lecture course for pre-med freshmen students. It was a required calculus-based introductory course, with a huge class of nearly 200. The problem was that the majority of students had a limited exposure to physics, and, what was even worse, low interest in this subject. They had an impression that a physics course was a formal requirement and they would never need physics in their future lives. Besides, by the end of the week they were apparently tired. To remedy this problem I decided that each Friday I would break the standard succession of topics, and tell them of something physics-related but { simultaneously { entertaining. Three or four Friday lectures were devoted to why certain Hollywood movies contradict laws of Nature. After looking through fragments we discussed which particular laws were grossly violated and why. I remember that during one Friday lecture I captivated students with TV sci-fi miniseries on a catastrophic earthquake entitled 10.5, and then we talked about real-life earthquakes. Humans have been recording earthquakes for nearly 4,000 years. The deadliest one happened in China in 1556 A.D. On January 23 of that year, a powerful quake killed an estimated 830,000 people. By today's estimate its Richter scale magnitude was about 8.3. The strongest earthquake ever recorded was the 9.5-magnitude Valdivia earthquake in Chile which occurred in 1960. My remark that in passing from 9.5. -

Chemical Elements
D.N. Trifonov and V D. Trifonov CHEMICAL ELEMENTS HOW THEY WERE DISCOVERED Moscow Period* A I B MENDELEEV'S PERIODIC OF TH E ELE MEN 1 H A II B A III B A IV B A Vl 3 Li 4 Be sB eC 7 N " 6.94 9.01218 10.81 12.011 2s2 2pJ 14.0067 2 2l' 2p' ta Lithium Beryllium Boron Carbon Nitrogen 11 Na 12 Mg 13 Al 14 Si 15 P 1 22.98977 24.305 26 98,54 30.9737 » 3 3s1 3i* 3.'V Sodium Magnesium Aluminium Silicon Phoaphorui 19 K 20 Ca 21 Sc 22 Ti 4790 3 j 39.09, 4i, 40.08 , , 44.9559 Jd, W * 30 4S Potassium Calcium Scandium Titanium Van! 4 29 CU 30 Zn 31 Ga 32 Ge 33 AS J 63 54 65.38 69.72 , , 725» *.24p' 74.9216 | «. , • 3d*°4»2 Copper Zinc Gallium Germanium Arsenic ,] 37 Rb 38 Sr 39 Y 40 Zr 4| 85.467s 87.62 < 2 88.9059 9,22 5s1 j(> w Ss 4dw Rubidium Strontium Yttrium Zirconium 5 4 47 Ag 48 Cd 49 In so Sn 51 Sb i H2.40 mM H8.69 121.7 |j 5..5P- 5.' 5p2 S Silver Cadmium Indium Tin Antimony 55 CS 56 Ba 57 La* 72 Hf 132.9054 137.34 6V _ . 138.905s 5di«.» Hatnium Cesium Barium Lanthanum 6 79 AU so Hg 81 Tl 82 Pb 83 Bi I 20059 207.2 196.9665 «28p2 208.9804 J M'06*' Gold Mercury Thallium Lead Bismuth | 87 Fr 88 Ra 89 AC** 104 Ku 105 ff 7 (223J 226.0254 6dW 122') sa.7,2 [2611 Francium Radium Actinium Kurchatovium (NilStKH * LANTHANIDES 58Ce6>! 59 Pr 60 Nd 61 Pm 62 Sm 63 Eu 64& 140.12 so' 140.907 144.24 , , (145) 1504 .