How Do We Know That There Are Atoms
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Biochemical Thermodynamics
Biochemical Thermodynamics Biochemical Thermodynamics By Juan S. Jiménez Biochemical Thermodynamics By Juan S. Jiménez This book first published 2020 Cambridge Scholars Publishing Lady Stephenson Library, Newcastle upon Tyne, NE6 2PA, UK British Library Cataloguing in Publication Data A catalogue record for this book is available from the British Library Copyright © 2020 by Juan S. Jiménez All rights for this book reserved. No part of this book may be reproduced, stored in a retrieval system, or transmitted, in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, without the prior permission of the copyright owner. ISBN (10): 1-5275-5359-0 ISBN (13): 978-1-5275-5359-0 To the memory of Brígida and Francisco Jiménez CONTENTS PREFACE ..................................................................................................... x CHAPTER 1 .................................................................................................. 1 INTRODUCTION 1.1 The Atomic Theory of John Dalton and the Hypothesis of Amedeo Avogadro 1.2 The Mole and Avogadro’s Number 1.3 The ideal gas model 1.4 The Periodic Table and Initial Atomic Theories 1.5 The Hydrogen Atom and the Schrödinger Equation 1.6 Atomic Structure 1.7 Molecules CHAPTER 2 ................................................................................................ 35 ENTROPY 2.1 Systems, Properties and States 2.2 The First Law of Thermodynamics 2.3 Enthalpy 2.4 Reversible changes 2.5 The Second Law of Thermodynamics 2.6 A Particle in a One-dimensional Box 2.7 Quantum States 2.8 The Boltzmann Equation CHAPTER 3 ................................................................................................ 63 THE CHEMICAL EQUILIBRIUM 3.1 The Gibbs Function 3.2 The Chemical Potential 3.3 Chemical Equilibrium 3.4 Model Systems 3.5 The Equilibrium Constant for Chemical Reactions between Gases. -

Amedeo Avogadro
Amedeo Avogadro ALSO LISTED IN Physicists FAMOUS AS Chemist and Physicist NATIONALITY Italian Famous Italian Men RELIGION Roman Catholic BORN ON 09 August 1776 AD Famous 9th August Birthdays ZODIAC SIGN Leo Leo Men BORN IN Turin, Italy DIED ON 09 July 1856 AD PLACE OF DEATH Turin, Italy FATHER Filippo Avogadro MOTHER Anna Maria Vercellone SPOUSE: Felicita Mazzé Lorenzo Romano Amedeo Carlo Avogadro de Quaregna e di Cerreto, more popularly known as Amedeo Avogadro was born on August 9, 1776, in Turin, Italy. He was a gifted physicist and chemist who proposed the molecular theory, which is more popularly known as ‘Avogadro’s Law’. Although he earned a doctorate in ecclesiastical law, he developed a passion for studying mathematics and physics. He then gave up his career in law and pursued a career teaching natural physics at the Royal College of Vercelli. Years later, he was offered the chair of mathematical physics at the University of Turin. Avogadro conducted experiments in both physics and chemistry using mathematics as a basis for his findings. His hypothesis, known as the ‘Avogadro’s Law’ is recognized all over the world. He also published many works during his lifetime. The number 6.02214199 x 10^23 is named as Avogadro’s number to honor him for his contribution in molecular theory. Read on to know more about this great physicist and chemist. Read more at http://www.thefamouspeople.com/profiles/amadeo-avogadro- 532.php#09s5lE6QjEOdfSro.99 Career After studying philosophy in 1789, Amedeo Avogadro graduated in jurisprudence in 1792 and earned his doctorate in ecclesiastical law in 1796. -

Chemistry in Italy During Late 18Th and 19Th Centuries
CHEMISTRY IN ITALY DURING LATE 18TH AND 19TH CENTURIES Ignazio Renato Bellobono, CSci, CChem, FRSC LASA, Department of Physics, University of Milan. e-mail add ress : i.bell obon o@ti scali.it LASA, Dept.Dept. ofPhysics, Physics, University of Milan The birth of Electrochemistry Luigi Galvani, Alessandro Volta, and Luigi Valentino Brugnatelli From Chemistry to Radiochemistry The birth of Chemistry and Periodic Table Amedeo Avogadro and Stanislao Cannizzaro Contributions to Organic Chemistry LASA, Dept.Dept. ofPhysics, Physics, University of Milan 1737 At the Faculty of Medicine of the Bologna University, the first chair of Chemistry is establishedestablished,,andandassigned to Jacopo Bartolomeo BECCARI (1692-1766). He studied phosphorescence and the action of light on silver halides 1776 In some marshes of the Lago Maggiore, near AngeraAngera,, Alessandro VOLTA ((17451745--18271827),),hi gh school teacher of physics in Como, individuates a flammable gas, which he calls aria infiammabile. Methane is thus discovereddiscovered.. Two years laterlater,,heheis assignedassigned,,asas professor of experimental phihysicscs,,toto the UiUniversi ty of PiPavia LASA, DtDept. of PhPhys icscs,, University of Milan 1778 In aletter a letter to Horace Bénédict de Saussure, aaSwissSwiss naturalist, VOLTA introduces, beneath that of electrical capacitycapacity,, the fundamental concept of tensione elettrica (electrical tension), exactly the name that CITCE recommended for the difference of potential in an electrochemical cell. 17901790--17911791 VOLTA anticipatesanticipates,,bybyabout 10 yearsyears,,thethe GAYGAY--LUSSACLUSSAC linear de ppyendency of gas volume on tem pp,erature, at constant pressurepressure,,andandafew a fewyears later ((17951795)) anticipatesanticipates,,byby about 6years 6 years,,thethe soso--calledcalled John Dalton’s rules ((18011801))ononvapour pressure LASA, Dept.Dept. -

2. Molecular Stucture/Basic Spectroscopy the Electromagnetic Spectrum
2. Molecular stucture/Basic spectroscopy The electromagnetic spectrum Spectral region fooatocadr atomic and molecular spectroscopy E. Hecht (2nd Ed.) Optics, Addison-Wesley Publishing Company,1987 Per-Erik Bengtsson Spectral regions Mo lecu lar spec troscopy o ften dea ls w ith ra dia tion in the ultraviolet (UV), visible, and infrared (IR) spectltral reg ions. • The visible region is from 400 nm – 700 nm • The ultraviolet region is below 400 nm • The infrared region is above 700 nm. 400 nm 500 nm 600 nm 700 nm Spectroscopy: That part of science which uses emission and/or absorption of radiation to deduce atomic/molecular properties Per-Erik Bengtsson Some basics about spectroscopy E = Energy difference = c /c h = Planck's constant, 6.63 10-34 Js ergy nn = Frequency E hn = h/hc /l E = h = hc / c = Velocity of light, 3.0 108 m/s = Wavelength 0 Often the wave number, , is used to express energy. The unit is cm-1. = E / hc = 1/ Example The energy difference between two states in the OH-molecule is 35714 cm-1. Which wavelength is needed to excite the molecule? Answer = 1/ =35714 cm -1 = 1/ = 280 nm. Other ways of expressing this energy: E = hc/ = 656.5 10-19 J E / h = c/ = 9.7 1014 Hz Per-Erik Bengtsson Species in combustion Combustion involves a large number of species Atoms oxygen (O), hydrogen (H), etc. formed by dissociation at high temperatures Diatomic molecules nitrogen (N2), oxygen (O2) carbon monoxide (CO), hydrogen (H2) nitr icoxide (NO), hy droxy l (OH), CH, e tc. -

Ijmp.Jor.Br V
INDEPENDENT JOURNAL OF MANAGEMENT & PRODUCTION (IJM&P) http://www.ijmp.jor.br v. 10, n. 8, Special Edition Seng 2019 ISSN: 2236-269X DOI: 10.14807/ijmp.v10i8.1046 A NEW HYPOTHESIS ABOUT THE NUCLEAR HYDROGEN STRUCTURE Relly Victoria Virgil Petrescu IFToMM, Romania E-mail: [email protected] Raffaella Aversa University of Naples, Italy E-mail: [email protected] Antonio Apicella University of Naples, Italy E-mail: [email protected] Taher M. Abu-Lebdeh North Carolina A and T State Univesity, United States E-mail: [email protected] Florian Ion Tiberiu Petrescu IFToMM, Romania E-mail: [email protected] Submission: 5/3/2019 Accept: 5/20/2019 ABSTRACT In other papers already presented on the structure and dimensions of elemental hydrogen, the elementary particle dynamics was taken into account in order to be able to determine the size of the hydrogen. This new work, one comes back with a new dynamic hypothesis designed to fundamentally change again the dynamic particle size due to the impulse influence of the particle. Until now it has been assumed that the impulse of an elementary particle is equal to the mass of the particle multiplied by its velocity, but in reality, the impulse definition is different, which is derived from the translational kinetic energy in a rapport of its velocity. This produces an additional condensation of matter in its elemental form. Keywords: Particle structure; Impulse; Condensed matter. [http://creativecommons.org/licenses/by/3.0/us/] Licensed under a Creative Commons Attribution 3.0 United States License 1749 INDEPENDENT JOURNAL OF MANAGEMENT & PRODUCTION (IJM&P) http://www.ijmp.jor.br v. -

Avogadro's Constant
Avogadro's Constant Nancy Eisenmenger Question Artem, 8th grade How \Avogadro constant" was invented and how scientists calculated it for the first time? Answer What is Avogadro's Constant? • Avogadros constant, NA: the number of particles in a mole • mole: The number of Carbon-12 atoms in 0.012 kg of Carbon-12 23 −1 • NA = 6:02214129(27) × 10 mol How was Avogadro's Constant Invented? Avogadro's constant was invented because scientists were learning about measuring matter and wanted a way to relate the microscopic to the macroscopic (e.g. how many particles (atoms, molecules, etc.) were in a sample of matter). The first scientists to see a need for Avogadro's constant were studying gases and how they behave under different temperatures and pressures. Amedeo Avogadro did not invent the constant, but he did state that all gases with the same volume, pressure, and temperature contained the same number of gas particles, which turned out to be very important to the development of our understanding of the principles of chemistry and physics. The constant was first calculated by Johann Josef Loschmidt, a German scientist, in 1865. He actually calculated the Loschmidt number, a constant that measures the same thing as Avogadro's number, but in different units (ideal gas particles per cubic meter at 0◦C and 1 atm). When converted to the same units, his number was off by about a factor of 10 from Avogadro's number. That may sound like a lot, but given the tools he had to work with for both theory and experiment and the magnitude of the number (∼ 1023), that is an impressively close estimate. -

Chemical Formulas the Elements
Chemical Formulas A chemical formula gives the numbers and types of atoms that are found in a substance. When the substance is a discrete molecule, then the chemical formula is also its molecular formula. Fe (iron) is a chemical formula Fe2O3 is a molecular formula The Elements The chemical formulas of most of the elements are simply their elemental symbol: Na (sodium) Fe (iron) He (helium) U (uranium) These chemical formulas are said to be monatomic—only an atom in chemical formula 1 The Elements There are seven elements that occur naturally as diatomic molecules—molecules that contain two atoms: H2 (hydrogen) N2 (nitrogen) O2 (oxygen) F2 (fluorine) Cl2 (chlorine) Br2 (bromine) I2 (iodine) The last four elements in this list are in the same family of the Periodic Table Binary Compounds A binary compound is one composed of only two different types of atoms. Rules for binary compound formulas 1. Element to left in Periodic Table comes first except for hydrogen: KCl PCl3 Al2S3 Fe3O4 2 Binary Compounds 2. Hydrogen comes last unless other element is from group 16 or 17: LiH, NH3, B2H4, CH4 3. If both elements are from the same group, the lower element comes first: SiC, BrF3 Other Compounds For compounds with three or more elements that are not ionic, if it contains carbon, this comes first followed by hydrogen. Other elements are then listed in alphabetical order: C2H6O C4H9BrO CH3Cl C8H10N4O2 3 Other Compounds However, the preceding rule is often ignored when writing organic formulas (molecules containing carbon, hydrogen, and maybe other elements) in order to give a better idea of how the atoms are connected: C2H6O is the molecular formula for ethanol, but nobody ever writes it this way—instead the formula is written C2H5OH to indicate one H atom is connected to the O atom. -

Syllabus of Biochemistry (Hons.)
Syllabus of Biochemistry (Hons.) for SEM-I & SEM-II under CBCS (to be effective from Academic Year: 2017-18) The University of Burdwan Burdwan, West Bengal 1 1. Introduction The syllabus for Biochemistry at undergraduate level using the Choice Based Credit system has been framed in compliance with model syllabus given by UGC. The main objective of framing this new syllabus is to give the students a holistic understanding of the subject giving substantial weight age to both the core content and techniques used in Biochemistry. The ultimate goal of the syllabus is that the students at the end are able to secure a job. Keeping in mind and in tune with the changing nature of the subject, adequate emphasis has been given on new techniques of mapping and understanding of the subject. The syllabus has also been framed in such a way that the basic skills of subject are taught to the students, and everyone might not need to go for higher studies and the scope of securing a job after graduation will increase. It is essential that Biochemistry students select their general electives courses from Chemistry, Physics, Mathematics and/or any branch of Life Sciences disciplines. While the syllabus is in compliance with UGC model curriculum, it is necessary that Biochemistry students should learn “Basic Microbiology” as one of the core courses rather than as elective while. Course on “Concept of Genetics” has been moved to electives. Also, it is recommended that two elective courses namely Nutritional Biochemistry and Advanced Biochemistry may be made compulsory. 2 Type of Courses Number of Courses Course type Description B. -

GANPAT UNIVERSITY FACULTY of SCIENCE TEACHING and EXAMINATION SCHEME Programme Bachelor of Science Branch/Spec
GANPAT UNIVERSITY FACULTY OF SCIENCE TEACHING AND EXAMINATION SCHEME Programme Bachelor of Science Branch/Spec. Microbiology Semester I Effective from Academic Year 2015- Effective for the batch Admitted in June 2015 16 Teaching scheme Examination scheme (Marks) Subject Subject Name Credit Hours (per week) Theory Practical Code Lecture(DT) Practical(Lab.) Lecture(DT) Practical(Lab.) CE SEE Total CE SEE Total L TU Total P TW Total L TU Total P TW Total UMBA101FOM FUNDAMENTALS 04 - 04 - - - 04 - 04 - - - 40 60 100 - - - OF MICROBIOLOGY UCHA101GCH GENERAL 04 - 04 - - - 04 - 04 - - - 40 60 100 - - - CHEMISTRY-I UPHA101GPH GENERAL 04 - 04 - - - 04 - 04 - - - 40 60 100 - - - PHYSICS-I UENA101ENG ENGLISH-I 02 - 02 - - - 02 - 02 - - - 40 60 100 - - - OPEN SUBJECT – 1 02 - 02 - - - 02 - 02 - - - 40 60 100 - - - UPMA101PRA PRACTICAL - - - 02 - 02 - - - 04 - 04 - - - - 50 50 MODULE-I UPCA101PRA PRACTICAL - - - 02 - 02 - - - 04 - 04 - - - - 50 50 MODULE-I UPPA101PRA PRACTICAL - - - 02 - 02 - - - 04 - 04 - - - - 50 50 MODULE-I Total 16 - 16 06 - 06 16 - 16 12 - 12 200 300 500 - 150 150 GANPAT UNIVERSITY FACULTY OF SCIENCE Programme Bachelor of Science Branch/Spec. Microbiology Semester I Version 1.0.1.0 Effective from Academic Year 2015-16 Effective for the batch Admitted in July 2015 Subject code UMBA 101 Subject Name FUNDAMENTALS OF MICROBIOLOGY FOM Teaching scheme Examination scheme (Marks) (Per week) Lecture(DT) Practical(Lab.) Total CE SEE Total L TU P TW Credit 04 -- -- -- 04 Theory 40 60 100 Hours 04 -- -- -- 04 Practical -- -- -- Pre-requisites: Students should have basic knowledge of Microorganisms and microscopy of 10+2 level. Learning Outcome: The course will help the student to understand basic fundamentals of Microbiology and history of Microbiology. -

The Mole and Avogadro's Number
The Mole and Avogadro's Number The name mole (German Mol) is attributed to Wilhelm Ostwald who introduced the concept in the year 1902. It is an abbreviation for molecule (German Molekül), which is in turn derived from Latin moles "mass, massive structure". (From the Wikipedia article on the mole unit.) A good site that introduces the mole concept and includes sample calculations and practice problems can be found here, from John Park's excellent ChemTeam site. For some interesting background on Avogadro's number, see here. By T.A. Furtsch, Tennessee Technological University, Cookeville, TN. Don't miss the interview with Count Amedeo Avogadro and his wife the Countess Felicita, located here (thanks to Kory Tonouchi, Roosevelt High, Honolulu, HI). Avogadro's 1811 publication, "Essay on a Manner of Determining the Relative Masses of the Elementary Molecules of Bodies, and the Proportions in Which They Enter into These Compounds", may be found here (thanks to Carmen Giunta, Le Moyne College, Syracuse, NY). A fun "mole" page to visit is here. It is a collection of student projects from Carondelet High School. Back in '98 they had a mole mystery described here. Did you know we have a mole day? Find out about it here. And for some corny mole jokes, don't miss the Dictionary of Mole Day Terms & Jokes here! Introduction A mole of objects contains Avogadro's number, 6.022 X 1023, objects. Just as a dozen apples is 12 apples, a mole of apples is 6.022 X 1023 apples. A mole of iron atoms is 6.022 X 1023 iron atoms. -

Chemistry in One Dimension Pierre-Fran¸Coisloos,1, A) Caleb J
Chemistry in One Dimension Pierre-Fran¸coisLoos,1, a) Caleb J. Ball,1 and Peter M. W. Gill1, b) Research School of Chemistry, Australian National University, Canberra ACT 0200, Australia We report benchmark results for one-dimensional (1D) atomic and molecular sys- tems interacting via the Coulomb operator jxj−1. Using various wavefunction-type approaches, such as Hartree-Fock theory, second- and third-order Møller-Plesset per- turbation theory and explicitly correlated calculations, we study the ground state of atoms with up to ten electrons as well as small diatomic and triatomic molecules containing up to two electrons. A detailed analysis of the 1D helium-like ions is given and the expression of the high-density correlation energy is reported. We report the total energies, ionization energies, electron affinities and other interesting properties of the many-electron 1D atoms and, based on these results, we construct the 1D analog of Mendeleev's periodic table. We find that the 1D periodic table contains only two groups: the alkali metals and the noble gases. We also calculate the dissociation + 2+ 3+ curves of various 1D diatomics and study the chemical bond in H2 , HeH , He2 , + 2+ H2, HeH and He2 . We find that, unlike their 3D counterparts, 1D molecules are + primarily bound by one-electron bonds. Finally, we study the chemistry of H3 and we discuss the stability of the 1D polymer resulting from an infinite chain of hydrogen atoms. PACS numbers: 31.15.A-, 31.15.V-, 31.15.xt Keywords: one-dimensional atom; one-dimensional molecule; one-dimensional polymer; helium-like ions arXiv:1406.1343v1 [physics.chem-ph] 5 Jun 2014 a)Electronic mail: Corresponding author: [email protected] b)Electronic mail: [email protected] 1 I. -
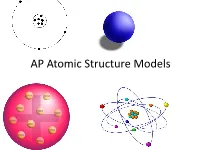
AP Atomic Structure Models What Is a Model?
AP Atomic Structure Models What is a Model? On a scrap piece of paper, write down your definition of a model with at least two examples. A model is a representation of an object, idea, action, or concept. Models A model in science is usually an approximation of the “real thing.” This means that all models have simplifications that may lead to omitted information. Models When a model is selected for a specific purpose, it should be able to help predict a “behavior.” The Four Historical Models of the Atom 1. Particle model 2. Plum Pudding model 3. Rutherford model 4. Bohr model Modern Model Quantum Mechanical Model Why do we have so many historical models? Are some of them still useful? When we initially began the process of determining the structure of the atom, we first had to overcome the prevailing scientific concept that matter was continuous. This idea of continuous matter was from Aristotle. (a Greek philosopher 384-322 B.C.E.) Particle Model John Dalton’s four ideas about the particle nature of matter helped push forward the acceptance of the first model. 1. All elements are composed of tiny indivisible particles called atoms. 2. Atoms of the same element are identical. The atoms of any one element are different from those of any other element. John Dalton 3. Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. 4. Chemical reactions occur when atoms are separated, joined, or rearranged. Atoms of one element, however, are never changed into atoms of another element as a result of a chemical reaction.