Spatial Distribution and the Effects of Competition on Some Temperate Scleractinia and Corallimorpharia
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Checklist of Fish and Invertebrates Listed in the CITES Appendices
JOINTS NATURE \=^ CONSERVATION COMMITTEE Checklist of fish and mvertebrates Usted in the CITES appendices JNCC REPORT (SSN0963-«OStl JOINT NATURE CONSERVATION COMMITTEE Report distribution Report Number: No. 238 Contract Number/JNCC project number: F7 1-12-332 Date received: 9 June 1995 Report tide: Checklist of fish and invertebrates listed in the CITES appendices Contract tide: Revised Checklists of CITES species database Contractor: World Conservation Monitoring Centre 219 Huntingdon Road, Cambridge, CB3 ODL Comments: A further fish and invertebrate edition in the Checklist series begun by NCC in 1979, revised and brought up to date with current CITES listings Restrictions: Distribution: JNCC report collection 2 copies Nature Conservancy Council for England, HQ, Library 1 copy Scottish Natural Heritage, HQ, Library 1 copy Countryside Council for Wales, HQ, Library 1 copy A T Smail, Copyright Libraries Agent, 100 Euston Road, London, NWl 2HQ 5 copies British Library, Legal Deposit Office, Boston Spa, Wetherby, West Yorkshire, LS23 7BQ 1 copy Chadwick-Healey Ltd, Cambridge Place, Cambridge, CB2 INR 1 copy BIOSIS UK, Garforth House, 54 Michlegate, York, YOl ILF 1 copy CITES Management and Scientific Authorities of EC Member States total 30 copies CITES Authorities, UK Dependencies total 13 copies CITES Secretariat 5 copies CITES Animals Committee chairman 1 copy European Commission DG Xl/D/2 1 copy World Conservation Monitoring Centre 20 copies TRAFFIC International 5 copies Animal Quarantine Station, Heathrow 1 copy Department of the Environment (GWD) 5 copies Foreign & Commonwealth Office (ESED) 1 copy HM Customs & Excise 3 copies M Bradley Taylor (ACPO) 1 copy ^\(\\ Joint Nature Conservation Committee Report No. -

Cnidae Variability in Balanophyllia Europaea and B
sm69n1075b 6/3/05 14:38 Página 75 SCI. MAR., 69 (1): 75-86 SCIENTIA MARINA 2005 Cnidae variability in Balanophyllia europaea and B. regia (Scleractinia: Dendrophylliidae) in the NE Atlantic and Mediterranean Sea* ALEJANDRO TERRÓN-SIGLER and PABLO J. LÓPEZ-GONZÁLEZ Biodiversidad y Ecología de Invertebrados Marinos, Departamento de Fisiología y Zoología, Facultad de Biología, Universidad de Sevilla, Reina Mercedes, 6, 41012 Sevilla, Spain. E-mail: [email protected] / [email protected] SUMMARY: Traditionally and for practical reasons, skeleton structure has been the main source of taxonomic characters for scleractinian systematics, whereas information from soft tissues has been comparatively neglected. However, skeleton variability may leave species identification uncertain. The use of characters from soft tissues (e.g. polyp anatomy, cnidae size) is routine in the study of other (“soft”) hexacorallian orders. This contribution aims to determine whether cnidae char- acters are useful in taxonomic studies of scleractinians. The cnidae composition of two congeneric species—Balanophyllia europaea (Risso, 1826) and Balanophyllia regia Gosse, 1860—have been studied throughout a wide geographical area. The data obtained show consistent qualitative and quantitative differences between the two species. This study shows that the cnidae characters can be useful taxonomic criteria for distinguishing congeneric species. Key words: Scleractinia, Balanophyllia, cnidome, taxonomy, geographical variability. RESUMEN: VARIABILIDAD EN LOS CNIDOCISTOS DE BALANOPHYLLIA EUROPAEA Y B. REGIA (SCLERACTINIA: DENDROPHYLLIIDAE) EN EL ATLÁNTICO NORDESTE Y MEDITERRÁNEO. – Tradicionalmente, y por razones prácticas, la estructura del esqueleto ha sido la fuente principal de caracteres en la sistemática de escleractinias, mientras que la obtención de información a partir de los tejidos esta prácticamente desatendida. -

Deep‐Sea Coral Taxa in the U.S. Gulf of Mexico: Depth and Geographical Distribution
Deep‐Sea Coral Taxa in the U.S. Gulf of Mexico: Depth and Geographical Distribution by Peter J. Etnoyer1 and Stephen D. Cairns2 1. NOAA Center for Coastal Monitoring and Assessment, National Centers for Coastal Ocean Science, Charleston, SC 2. National Museum of Natural History, Smithsonian Institution, Washington, DC This annex to the U.S. Gulf of Mexico chapter in “The State of Deep‐Sea Coral Ecosystems of the United States” provides a list of deep‐sea coral taxa in the Phylum Cnidaria, Classes Anthozoa and Hydrozoa, known to occur in the waters of the Gulf of Mexico (Figure 1). Deep‐sea corals are defined as azooxanthellate, heterotrophic coral species occurring in waters 50 m deep or more. Details are provided on the vertical and geographic extent of each species (Table 1). This list is adapted from species lists presented in ʺBiodiversity of the Gulf of Mexicoʺ (Felder & Camp 2009), which inventoried species found throughout the entire Gulf of Mexico including areas outside U.S. waters. Taxonomic names are generally those currently accepted in the World Register of Marine Species (WoRMS), and are arranged by order, and alphabetically within order by suborder (if applicable), family, genus, and species. Data sources (references) listed are those principally used to establish geographic and depth distribution. Only those species found within the U.S. Gulf of Mexico Exclusive Economic Zone are presented here. Information from recent studies that have expanded the known range of species into the U.S. Gulf of Mexico have been included. The total number of species of deep‐sea corals documented for the U.S. -

Sexual Reproduction of the Solitary Sunset Cup Coral Leptopsammia Pruvoti (Scleractinia: Dendrophylliidae) in the Mediterranean
Marine Biology (2005) 147: 485–495 DOI 10.1007/s00227-005-1567-z RESEARCH ARTICLE S. Goffredo Æ J. Radetic´Æ V. Airi Æ F. Zaccanti Sexual reproduction of the solitary sunset cup coral Leptopsammia pruvoti (Scleractinia: Dendrophylliidae) in the Mediterranean. 1. Morphological aspects of gametogenesis and ontogenesis Received: 16 July 2004 / Accepted: 18 December 2004 / Published online: 3 March 2005 Ó Springer-Verlag 2005 Abstract Information on the reproduction in scleractin- came indented, assuming a sickle or dome shape. We can ian solitary corals and in those living in temperate zones hypothesize that the nucleus’ migration and change of is notably scant. Leptopsammia pruvoti is a solitary coral shape may have to do with facilitating fertilization and living in the Mediterranean Sea and along Atlantic determining the future embryonic axis. During oogene- coasts from Portugal to southern England. This coral sis, oocyte diameter increased from a minimum of 20 lm lives in shaded habitats, from the surface to 70 m in during the immature stage to a maximum of 680 lm depth, reaching population densities of >17,000 indi- when mature. Embryogenesis took place in the coelen- viduals mÀ2. In this paper, we discuss the morphological teron. We did not see any evidence that even hinted at aspects of sexual reproduction in this species. In a sep- the formation of a blastocoel; embryonic development arate paper, we report the quantitative data on the an- proceeded via stereoblastulae with superficial cleavage. nual reproductive cycle and make an interspecific Gastrulation took place by delamination. Early and late comparison of reproductive traits among Dend- embryos had diameters of 204–724 lm and 290–736 lm, rophylliidae aimed at defining different reproductive respectively. -

Volume 2. Animals
AC20 Doc. 8.5 Annex (English only/Seulement en anglais/Únicamente en inglés) REVIEW OF SIGNIFICANT TRADE ANALYSIS OF TRADE TRENDS WITH NOTES ON THE CONSERVATION STATUS OF SELECTED SPECIES Volume 2. Animals Prepared for the CITES Animals Committee, CITES Secretariat by the United Nations Environment Programme World Conservation Monitoring Centre JANUARY 2004 AC20 Doc. 8.5 – p. 3 Prepared and produced by: UNEP World Conservation Monitoring Centre, Cambridge, UK UNEP WORLD CONSERVATION MONITORING CENTRE (UNEP-WCMC) www.unep-wcmc.org The UNEP World Conservation Monitoring Centre is the biodiversity assessment and policy implementation arm of the United Nations Environment Programme, the world’s foremost intergovernmental environmental organisation. UNEP-WCMC aims to help decision-makers recognise the value of biodiversity to people everywhere, and to apply this knowledge to all that they do. The Centre’s challenge is to transform complex data into policy-relevant information, to build tools and systems for analysis and integration, and to support the needs of nations and the international community as they engage in joint programmes of action. UNEP-WCMC provides objective, scientifically rigorous products and services that include ecosystem assessments, support for implementation of environmental agreements, regional and global biodiversity information, research on threats and impacts, and development of future scenarios for the living world. Prepared for: The CITES Secretariat, Geneva A contribution to UNEP - The United Nations Environment Programme Printed by: UNEP World Conservation Monitoring Centre 219 Huntingdon Road, Cambridge CB3 0DL, UK © Copyright: UNEP World Conservation Monitoring Centre/CITES Secretariat The contents of this report do not necessarily reflect the views or policies of UNEP or contributory organisations. -

Adaptive Divergence, Neutral Panmixia, and Algal Symbiont Population Structure in the Temperate Coral Astrangia Poculata Along the Mid-Atlantic United States
Adaptive divergence, neutral panmixia, and algal symbiont population structure in the temperate coral Astrangia poculata along the Mid-Atlantic United States Hannah E. Aichelman1,2 and Daniel J. Barshis2 1 Department of Biology, Boston University, Boston, MA, USA 2 Department of Biological Sciences, Old Dominion University, Norfolk, VA, USA ABSTRACT Astrangia poculata is a temperate scleractinian coral that exists in facultative symbiosis with the dinoflagellate alga Breviolum psygmophilum across a range spanning the Gulf of Mexico to Cape Cod, Massachusetts. Our previous work on metabolic thermal performance of Virginia (VA) and Rhode Island (RI) populations of A. poculata revealed physiological signatures of cold (RI) and warm (VA) adaptation of these populations to their respective local thermal environments. Here, we used whole-transcriptome sequencing (mRNA-Seq) to evaluate genetic differences and identify potential loci involved in the adaptive signature of VA and RI populations. Sequencing data from 40 A. poculata individuals, including 10 colonies from each population and symbiotic state (VA-white, VA-brown, RI-white, and RI-brown), yielded a total of 1,808 host-associated and 59 algal symbiont-associated single nucleotide polymorphisms (SNPs) post filtration. Fst outlier analysis identified 66 putative high outlier SNPs in the coral host and 4 in the algal symbiont. Differentiation of VA and RI populations in the coral host was driven by putatively adaptive loci, not neutral divergence (Fst = 0.16, p = 0.001 and Fst = 0.002, p = 0.269 for outlier and neutral SNPs respectively). In contrast, we found evidence of neutral population differentiation in B. psygmophilum (Fst = 0.093, p = 0.001). -

Factors Affecting the Abundance of Paracyathus Stearns!! on Subtidal Rock Walls
MLML / M8~RllIBR;jRY 8272 MOSS LANDING RD. MOSS LANDING, CA 95039 FACTORS AFFECTING THE ABUNDANCE OF PARACYATHUS STEARNS!! ON SUBTIDAL ROCK WALLS by Mark Pranger A thesis submitted in partial fulfillment ofthe requirements for the degree of Master ofScience in Marine Science in the School ofNatural Sciences California State University, Fresno August 1999 ACKNOWLEDGMENTS I would like to thank the members of my graduate advisory committee for their advice and guidance during this process. Dr. James Nybakken for his interest and education in invertebrate biology and for allowing me to help in the teaching of others. For the insights and humor of Dr. Mike Foster, that helped improve this study and my education. For the help of Dr. Stephen Ervin who in the last hours help me through all CSU Fresno's paper work and deadlines. I am grateful to the staff at the Pollution Studies Lab at Granite Canyon for their help during experiments and for allowing me the use of their working space. I am indebted to the staff at Moss Landing Marine Labs that kept all the equipment working and operational, and to the Dr. Earl and Ethel Myers Oceanographic and Marine Biology Trust for helping to fund this project. Most of all, thanks goes to the many students at Moss Landing. Without their help this study would not have been completed. Special thanks goes to; Mat Edwards for being a faithful dive buddy, to Torno Eguchi, Michelle White, Bryn Phillips, Cassandra Roberts, Michelle Lander, and Lara Lovera for their help on statistics and editing of early drafts, and especially to Michele Jacobi for her help in all aspects of this study and her constant encouragement for me to finish. -

The Diet and Predator-Prey Relationships of the Sea Star Pycnopodia Helianthoides (Brandt) from a Central California Kelp Forest
THE DIET AND PREDATOR-PREY RELATIONSHIPS OF THE SEA STAR PYCNOPODIA HELIANTHOIDES (BRANDT) FROM A CENTRAL CALIFORNIA KELP FOREST A Thesis Presented to The Faculty of Moss Landing Marine Laboratories San Jose State University In Partial Fulfillment of the Requirements for the Degree Master of Arts by Timothy John Herrlinger December 1983 TABLE OF CONTENTS Acknowledgments iv Abstract vi List of Tables viii List of Figures ix INTRODUCTION 1 MATERIALS AND METHODS Site Description 4 Diet 5 Prey Densities and Defensive Responses 8 Prey-Size Selection 9 Prey Handling Times 9 Prey Adhesion 9 Tethering of Calliostoma ligatum 10 Microhabitat Distribution of Prey 12 OBSERVATIONS AND RESULTS Diet 14 Prey Densities 16 Prey Defensive Responses 17 Prey-Size Selection 18 Prey Handling Times 18 Prey Adhesion 19 Tethering of Calliostoma ligatum 19 Microhabitat Distribution of Prey 20 DISCUSSION Diet 21 Prey Densities 24 Prey Defensive Responses 25 Prey-Size Selection 27 Prey Handling Times 27 Prey Adhesion 28 Tethering of Calliostoma ligatum and Prey Refugia 29 Microhabitat Distribution of Prey 32 Chemoreception vs. a Chemotactile Response 36 Foraging Strategy 38 LITERATURE CITED 41 TABLES 48 FIGURES 56 iii ACKNOWLEDGMENTS My span at Moss Landing Marine Laboratories has been a wonderful experience. So many people have contributed in one way or another to the outcome. My diving buddies perse- vered through a lot and I cherish our camaraderie: Todd Anderson, Joel Thompson, Allan Fukuyama, Val Breda, John Heine, Mike Denega, Bruce Welden, Becky Herrlinger, Al Solonsky, Ellen Faurot, Gilbert Van Dykhuizen, Ralph Larson, Guy Hoelzer, Mickey Singer, and Jerry Kashiwada. Kevin Lohman and Richard Reaves spent many hours repairing com puter programs for me. -

Deep-Sea Coral Taxa in the U. S. Caribbean Region: Depth And
Deep‐Sea Coral Taxa in the U. S. Caribbean Region: Depth and Geographical Distribution By Stephen D. Cairns1 1. National Museum of Natural History, Smithsonian Institution, Washington, DC An update of the status of the azooxanthellate, heterotrophic coral species that occur predominantly deeper than 50 m in the U.S. Caribbean territories is not given in this volume because of lack of significant additional data. However, an updated list of deep‐sea coral species in Phylum Cnidaria, Classes Anthozoa and Hydrozoa, from the Caribbean region (Figure 1) is presented below. Details are provided on depth ranges and known geographic distributions within the region (Table 1). This list is adapted from Lutz & Ginsburg (2007, Appendix 8.1) in that it is restricted to the U. S. territories in the Caribbean, i.e., Puerto Rico, U. S. Virgin Islands, and Navassa Island, not the entire Caribbean and Bahamian region. Thus, this list is significantly shorter. The list has also been reordered alphabetically by family, rather than species, to be consistent with other regional lists in this volume, and authorship and publication dates have been added. Also, Antipathes americana is now properly assigned to the genus Stylopathes, and Stylaster profundus to the genus Stenohelia. Furthermore, many of the geographic ranges have been clarified and validated. Since 2007 there have been 20 species additions to the U.S. territories list (indicated with blue shading in the list), mostly due to unpublished specimens from NMNH collections. As a result of this update, there are now known to be: 12 species of Antipatharia, 45 species of Scleractinia, 47 species of Octocorallia (three with incomplete taxonomy), and 14 species of Stylasteridae, for a total of 118 species found in the relatively small geographic region of U. -

Kelp Forest Monitoring Handbook — Volume 1: Sampling Protocol
KELP FOREST MONITORING HANDBOOK VOLUME 1: SAMPLING PROTOCOL CHANNEL ISLANDS NATIONAL PARK KELP FOREST MONITORING HANDBOOK VOLUME 1: SAMPLING PROTOCOL Channel Islands National Park Gary E. Davis David J. Kushner Jennifer M. Mondragon Jeff E. Mondragon Derek Lerma Daniel V. Richards National Park Service Channel Islands National Park 1901 Spinnaker Drive Ventura, California 93001 November 1997 TABLE OF CONTENTS INTRODUCTION .....................................................................................................1 MONITORING DESIGN CONSIDERATIONS ......................................................... Species Selection ...........................................................................................2 Site Selection .................................................................................................3 Sampling Technique Selection .......................................................................3 SAMPLING METHOD PROTOCOL......................................................................... General Information .......................................................................................8 1 m Quadrats ..................................................................................................9 5 m Quadrats ..................................................................................................11 Band Transects ...............................................................................................13 Random Point Contacts ..................................................................................15 -

The Earliest Diverging Extant Scleractinian Corals Recovered by Mitochondrial Genomes Isabela G
www.nature.com/scientificreports OPEN The earliest diverging extant scleractinian corals recovered by mitochondrial genomes Isabela G. L. Seiblitz1,2*, Kátia C. C. Capel2, Jarosław Stolarski3, Zheng Bin Randolph Quek4, Danwei Huang4,5 & Marcelo V. Kitahara1,2 Evolutionary reconstructions of scleractinian corals have a discrepant proportion of zooxanthellate reef-building species in relation to their azooxanthellate deep-sea counterparts. In particular, the earliest diverging “Basal” lineage remains poorly studied compared to “Robust” and “Complex” corals. The lack of data from corals other than reef-building species impairs a broader understanding of scleractinian evolution. Here, based on complete mitogenomes, the early onset of azooxanthellate corals is explored focusing on one of the most morphologically distinct families, Micrabaciidae. Sequenced on both Illumina and Sanger platforms, mitogenomes of four micrabaciids range from 19,048 to 19,542 bp and have gene content and order similar to the majority of scleractinians. Phylogenies containing all mitochondrial genes confrm the monophyly of Micrabaciidae as a sister group to the rest of Scleractinia. This topology not only corroborates the hypothesis of a solitary and azooxanthellate ancestor for the order, but also agrees with the unique skeletal microstructure previously found in the family. Moreover, the early-diverging position of micrabaciids followed by gardineriids reinforces the previously observed macromorphological similarities between micrabaciids and Corallimorpharia as -
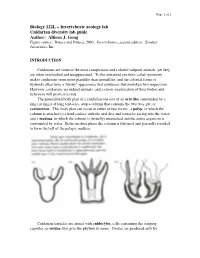
Cnidarians Are Some of the Most Conspicuous and Colorful Tidepool Animals, Yet They Are Often Overlooked and Unappreciated
Page 1 of 1 Biology 122L – Invertebrate zoology lab Cnidarian diversity lab guide Author: Allison J. Gong Figure source: Brusca and Brusca, 2003. Invertebrates, second edition. Sinauer Associates, Inc. INTRODUCTION Cnidarians are some of the most conspicuous and colorful tidepool animals, yet they are often overlooked and unappreciated. To the untrained eye their radial symmetry makes cnidarians seem more plantlike than animallike, and the colonial forms of hydroids often have a "bushy" appearance that reinforces that mistaken first impression. However, cnidarians are indeed animals, and a closer examination of their bodies and behaviors will prove it to you. The generalized body plan of a cnidarian consists of an oral disc surrounded by a ring (or rings) of long tentacles, atop a column that contains the two-way gut, or coelenteron. This body plan can occur in either of two forms: a polyp, in which the column is attached to a hard surface with the oral disc and tentacles facing into the water; and a medusa, in which the column is (usually) unattached and the entire organism is surrounded by water. In the medusa phase the column is flattened and generally rounded to form the bell of the pelagic medusa. Cnidarian tentacles are armed with cnidocytes, cells containing the stinging capsules, or cnidae, that give the phylum its name. Cnidae are produced only by Page 2 of 2 cnidarians, although they can occasionally be found in the cerata of nudibranch molluscs that feed on cnidarians: through a process that is not understood, some nudibranchs are able to ingest cnidae from their prey and sequester them, unfired, in their cerata for defense against their own predators.