D-Sedoheptulose-7-Phosphate Is a Common Precursor for the Heptoses of Septacidin and Hygromycin B
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Postulated Physiological Roles of the Seven-Carbon Sugars, Mannoheptulose, and Perseitol in Avocado
J. AMER. SOC. HORT. SCI. 127(1):108–114. 2002. Postulated Physiological Roles of the Seven-carbon Sugars, Mannoheptulose, and Perseitol in Avocado Xuan Liu,1 James Sievert, Mary Lu Arpaia, and Monica A. Madore2 Department of Botany and Plant Sciences, University of California, Riverside, CA 92521 ADDITIONAL INDEX WORDS. ‘Hass’ avocado on ‘Duke 7’ rootstock, phloem transport, ripening, Lauraceae ABSTRACT. Avocado (Persea americana Mill.) tissues contain high levels of the seven-carbon (C7) ketosugar mannoheptulose and its polyol form, perseitol. Radiolabeling of intact leaves of ‘Hass’ avocado on ‘Duke 7’ rootstock indicated that both perseitol and mannoheptulose are not only primary products of photosynthetic CO2 fixation but are also exported in the phloem. In cell-free extracts from mature source leaves, formation of the C7 backbone occurred by condensation of a three-carbon metabolite (dihydroxyacetone-P) with a four-carbon metabolite (erythrose-4-P) to form sedoheptulose-1,7- bis-P, followed by isomerization to a phosphorylated D-mannoheptulose derivative. A transketolase reaction was also observed which converted five-carbon metabolites (ribose-5-P and xylulose-5-P) to form the C7 metabolite, sedoheptu- lose-7-P, but this compound was not metabolized further to mannoheptulose. This suggests that C7 sugars are formed from the Calvin Cycle, not oxidative pentose phosphate pathway, reactions in avocado leaves. In avocado fruit, C7 sugars were present in substantial quantities and the normal ripening processes (fruit softening, ethylene production, and climacteric respiration rise), which occurs several days after the fruit is picked, did not occur until levels of C7 sugars dropped below an apparent threshold concentration of ≈20 mg·g–1 fresh weight. -

A by Fluorous-Tag Assistance Th
Angewandte Chemie DOI: 10.1002/ange.200704262 Carbohydrate Microarrays Synthesis and Quantitative Evaluation of Glycero-d-manno-heptose Binding to Concanavalin A by Fluorous-Tag Assistance** Firoz A. Jaipuri, Beatrice Y. M. Collet, and Nicola L. Pohl* Herein we report the first use of a quantitative fluorous approach has proven valuable for the probing of other classes microarray strategy to show that the mannose-binding lectin of small molecules.[2] In the case of histone deacetylase concanavalin A (conA), contrary to prevailing belief, actually inhibitors with dissocation constants of less than 0.1s À1, the can accept modifications of the mannose at the C-6 position in hits found by fluorous microarrays were comparable to those the form of glycero-manno-heptoses found on pathogenic found by techniques such as surface plasmon resonance bacteria (Figure 1). The well-known mannose–conA interac- (SPR) and solution-based biochemical assays.[2a] Ideally, of course, the relative quantification of these binding interac- tions could also be carried out within the same fluorous microarray screening format. ConA is a plant lectin that is widely used like antibodies as research tools and diagnostics to identify the presence of specific sugars, such as mannose, on cells;[3] however, in reality the sugar specificities of lectins have not been tested broadly, especially against less readily available carbohydrates. ConA is the most-studied lectin and is usually considered to bind terminal alpha-linked mannose, glucose, and N-acetylglucos- amine. Earlier inhibition data suggest that modifications at the C-3, C-4, and C-6 positions of the d-mannopyranose deter binding to conA.[4] In particular, the loss of the hydroxy group in the C-6 position as in 6-deoxy-d-mannose and 1,6-anhydro- b-d-manno-pyranose result in complete loss of activity. -

(12) United States Patent (10) Patent No.: US 6,713,116 B1 Aldrich Et Al
USOO6713116B1 (12) United States Patent (10) Patent No.: US 6,713,116 B1 Aldrich et al. (45) Date of Patent: Mar. 30, 2004 (54) SWEET-STABLE ACIDIFIED BEVERAGES 4,957,763 A 9/1990 Saita et al. ................. 426/548 5,169,671. A 12/1992 Harada et al. .............. 426/658 (75) Inventors: Jessica A. Aldrich, Hazlet, NJ (US); 5,380,541 A 1/1995 Beyts et al. ................ 426/548 Lisa Y. Hanger, Basking Ridge, NJ 5.431,929 A 7/1995 Yatka et al. ................... 426/3 5,731,025 A 3/1998 Mitchell ..................... 426/548 (US); Guido Ritter, Laer (DE) 6,322,835 B1 * 11/2001 De Soete et al. ........... 426/453 (73) Assignee: Nutrinova Inc., Somerset, NJ (US) 6,372.277 B1 * 4/2002 Admiraal et al. ........... 426/548 FOREIGN PATENT DOCUMENTS (*) Notice: Subject to any disclaimer, the term of this patent is extended or adjusted under 35 W WO as: : 3.1. - - - - - - - - - - - A23L/1/236 U.S.C. 154(b) by 0 days. WO WO 98/19564 5/1998 ............. A23L/2/60 (21) Appl. No.: 09/675,825 OTHER PUBLICATIONS (22) Filed: Sep. 29, 2000 Widemann et al., “Synergistic Sweeteners”, Food Ingred. and Analysis Int., 19(6):51-52, 55-56 (abstract only), Dec. Related U.S. Application Data 1997.* (63) Continuation-in-part of application No. 09/186,275, filed on sk cited- by examiner Nov. 5, 1998, now abandoned. Primary Examiner Keith Hendricks (60) Pisional application No. 60/079,408, filed on Mar. 26, (74) Attorney, Agent, or Firm-ProPat, L.L.C. (51) Int. Cl. -

Carbohydrate Composition of Endotoxins from R-Type Isogenic Mutants of Shigella Sonnei Studied by Capillary Electrophoresis and GC-MS†
CROATICA CHEMICA ACTA CCACAA, ISSN 0011-1643, e-ISSN 1334-417X Croat. Chem. Acta 84 (3) (2011) 393–398. CCA-3487 Original Scientific Article Carbohydrate Composition of Endotoxins from R-type Isogenic Mutants of Shigella sonnei Studied by Capillary Electrophoresis and GC-MS† Annamária Bui,a Anikó Kilár,b,c Ágnes Dörnyei,a,c Viktória Poór,a Krisztina Kovács,b Béla Kocsis,b and Ferenc Kilára,c,* aInstitute of Bioanalysis, Faculty of Medicine, University of Pécs, Szigeti út 12., H-7624 Pécs bDepartment of Medical Microbiology and Immunology, Faculty of Medicine, University of Pécs, Szigeti út 12., H-7624 Pécs, Hungary cDepartment of Analytical and Environmental Chemistry, Faculty of Sciences, University of Pécs, Ifjúság útja 6., H-7624 Pécs, Hungary RECEIVED NOVEMBER 9, 2010; REVISED FEBRUARY 5, 2011; ACCEPTED MAY 13, 2011 Abstract. The carbohydrate composition of the rough-type endotoxins (lipopolysaccharides, LPSs) from Shigella sonnei mutant strains (Shigella sonnei phase II - 4303, 562H, R41 and 4350) was investigated by capillary electrophoresis and GC-MS. The monosaccharides obtained by hydrolysis were determined by capillary electrophoresis combined with laser induced fluorescence detection (CE-LIF) after labeling with 8-aminopyrene-1,3,6-trisulfonic acid (APTS) and by GC-MS as alditol-acetate derivatives. It was ob- tained that the lipopolysaccharides of the isogenic rough mutants are formed in a step-like manner, con- taining no heptose (4350), one D-glycero-D-mannoheptose (562H), or two or three L-glycero-D- mannoheptoses (R41, 4303, respectively) in the deep core region. Besides the heptoses, the longest LPS from the mutant Shigella sonnei 4303 contains hexoses, such as glucoses and galactoses, in a pro- portion of approximately 3:2. -

PENTOSE PHOSPHATE PATHWAY — Restricted for Students Enrolled in MCB102, UC Berkeley, Spring 2008 ONLY
Metabolism Lecture 5 — PENTOSE PHOSPHATE PATHWAY — Restricted for students enrolled in MCB102, UC Berkeley, Spring 2008 ONLY Bryan Krantz: University of California, Berkeley MCB 102, Spring 2008, Metabolism Lecture 5 Reading: Ch. 14 of Principles of Biochemistry, “Glycolysis, Gluconeogenesis, & Pentose Phosphate Pathway.” PENTOSE PHOSPHATE PATHWAY This pathway produces ribose from glucose, and it also generates 2 NADPH. Two Phases: [1] Oxidative Phase & [2] Non-oxidative Phase + + Glucose 6-Phosphate + 2 NADP + H2O Ribose 5-Phosphate + 2 NADPH + CO2 + 2H ● What are pentoses? Why do we need them? ◦ DNA & RNA ◦ Cofactors in enzymes ● Where do we get them? Diet and from glucose (and other sugars) via the Pentose Phosphate Pathway. ● Is the Pentose Phosphate Pathway just about making ribose sugars from glucose? (1) Important for biosynthetic pathways using NADPH, and (2) a high cytosolic reducing potential from NADPH is sometimes required to advert oxidative damage by radicals, e.g., ● - ● O2 and H—O Metabolism Lecture 5 — PENTOSE PHOSPHATE PATHWAY — Restricted for students enrolled in MCB102, UC Berkeley, Spring 2008 ONLY Two Phases of the Pentose Pathway Metabolism Lecture 5 — PENTOSE PHOSPHATE PATHWAY — Restricted for students enrolled in MCB102, UC Berkeley, Spring 2008 ONLY NADPH vs. NADH Metabolism Lecture 5 — PENTOSE PHOSPHATE PATHWAY — Restricted for students enrolled in MCB102, UC Berkeley, Spring 2008 ONLY Oxidative Phase: Glucose-6-P Ribose-5-P Glucose 6-phosphate dehydrogenase. First enzymatic step in oxidative phase, converting NADP+ to NADPH. Glucose 6-phosphate + NADP+ 6-Phosphoglucono-δ-lactone + NADPH + H+ Mechanism. Oxidation reaction of C1 position. Hydride transfer to the NADP+, forming a lactone, which is an intra-molecular ester. -

The Isolation and Partial Characterization of the Lipopolysaccharides from Rhizobium Leguminosarum Biovar Trifolii Strains TB4, TB104, and TB112 (Llne)
Eastern Illinois University The Keep Masters Theses Student Theses & Publications 1990 The solI ation and Partial Characterization of the Lipopolysaccharides from Rhizobium leguminosarum biovar trifolii strains TB4, TB104, and TB112 Jill K. Miller Eastern Illinois University This research is a product of the graduate program in Zoology at Eastern Illinois University. Find out more about the program. Recommended Citation Miller, Jill K., "The sI olation and Partial Characterization of the Lipopolysaccharides from Rhizobium leguminosarum biovar trifolii strains TB4, TB104, and TB112" (1990). Masters Theses. 2302. https://thekeep.eiu.edu/theses/2302 This is brought to you for free and open access by the Student Theses & Publications at The Keep. It has been accepted for inclusion in Masters Theses by an authorized administrator of The Keep. For more information, please contact [email protected]. THESIS REPRODUCTION CERTIFICATE TO: Graduate Degree Candidates who have written formal theses. SUBJECT: Permission to reproduce theses. The University Library is receiving a number of requests from other institutions asking permission to reproduce dissertations for inclusion in their library holdings. Although no copyright laws are involved, we feel that professional courtesy demands that permission be obtained from the author before we allow theses to be copied. Please sign one of the following statements: Booth Library of Eastern Illinois University has my permission to lend my thesis to a reputable college or university for the purpose of copying it for inclusion in that institution's library or research holdings. l f ttJ9o I Date I respectfully request Booth Library of Eastern Illinois University not allow my thesis be reproduced because -------------- Date Author m The Isolation and Partial Characterization of the Lipopolysaccharides from Rhizobium leguminosarum biovar trifolii strains TB4, TB104, and TB112 (llnE) BY Jill K. -

The Use of Natural Product Substrates for the Synthesis of Libraries of Biologically Active, New Chemical Entities
University of Montana ScholarWorks at University of Montana Graduate Student Theses, Dissertations, & Graduate School Professional Papers 2010 The seU of Natural Product Substrates for the Synthesis of Libraries of Biologically Active, New Chemical Entities Joshua Bryant Phillips The University of Montana Let us know how access to this document benefits ouy . Follow this and additional works at: https://scholarworks.umt.edu/etd Recommended Citation Phillips, Joshua Bryant, "The sU e of Natural Product Substrates for the Synthesis of Libraries of Biologically Active, New Chemical Entities" (2010). Graduate Student Theses, Dissertations, & Professional Papers. 1100. https://scholarworks.umt.edu/etd/1100 This Dissertation is brought to you for free and open access by the Graduate School at ScholarWorks at University of Montana. It has been accepted for inclusion in Graduate Student Theses, Dissertations, & Professional Papers by an authorized administrator of ScholarWorks at University of Montana. For more information, please contact [email protected]. THE USE OF NATURAL PRODUCT SUBSTRATES FOR THE SYNTHESIS OF LIBRARIES OF BIOLOGICALLY ACTIVE, NEW CHEMICAL ENTITIES by Joshua Bryant Phillips B.S. Chemistry, Northern Arizona University, 2002 B.S. Microbiology (health pre-professional), Northern Arizona University, 2002 Presented in partial fulfillment of the requirements for the degree of Doctor of Philosophy Chemistry The University of Montana June 2010 Phillips, Joshua Bryant Ph.D., June 2010 Chemistry THE USE OF NATURAL PRODUCT SUBSTRATES FOR THE SYNTHESIS OF LIBRARIES OF BIOLOGICALLY ACTIVE, NEW CHEMICAL ENTITIES Advisor: Dr. Nigel D. Priestley Chairperson: Dr. Bruce Bowler ABSTRACT Since Alexander Fleming first noted the killing of a bacterial culture by a mold, antibiotics have revolutionized medicine, being able to treat, and often cure life-threatening illnesses and making surgical procedures possible by eliminating the possibility of opportunistic infection. -

Sedoheptulose in Photosynthesis by Plants
TWO-WEEK LOAN COPY This is a Library Circulating Copy which may be borrowed for two weeks. For a personal retention copy, call Tech. Info. Division, Ext. 5545 UCRW.268 Unclassified - Biology Distribution UNIVERSITY OF CALIFOmIA Radiation Laboratory Contract No. W-7405-eng-48 SEDOHEPT[JLOSE IN PHOTOSYNTHESIS BY PLANTS A. A. Benson, J. A. Bassham, and Me Calvin May 1, 1951 Berkeley, California A. A. Benson, 6. A, Bassham, and M, Calvin Radiation Laboratory and Department of Ohemistry University of Calif omia, Berkeley May 1, 1951 Although its function has not been ascertained, the general 1 occurrence of sedohep~ulose, D-altroheptuLose, in the succulent plants is well established. This sugar has not been identified in the majority of the members of the plat lringdom, but it now appears possibae that its phosphate esters may perform a vital function during photosynthesis. We haw isolated labeled sedoheptulose monopho sphate in cl% 2 photosynthesis products of all the plants thus far studied in this laboratory (~lorelh,Scenedesmus, Rhodospfr5.llyn rubn~,and the leaves of barley seedlings, soy bean, alf~lfa,sugar beet, spinach and geranium). It is invariably found as monophosphate esters. At least two such esters have been observed in radiograms of C1'-labeled Scenedesmus. The mjor one is associated with fructose monophosphztte while the minor one is inseparable, as yet, from glucose monophosphate. Sedoheptulose may be liberated e~zymticallyfrom its phosphates during the lrilling of the plan-i;,'but it has not been observed to accwnulate In amounts exceeding tbe steady state concentrations of -these phosphates. .--LL- * This work was sponsored bp the United States Atomic Energy Cormnission, -- This suggests its participation only as a phosphate in most plznts. -
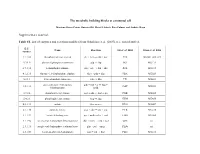
The Metabolic Building Blocks of a Minimal Cell Supplementary
The metabolic building blocks of a minimal cell Mariana Reyes-Prieto, Rosario Gil, Mercè Llabrés, Pere Palmer and Andrés Moya Supplementary material. Table S1. List of enzymes and reactions modified from Gabaldon et. al. (2007). n.i.: non identified. E.C. Name Reaction Gil et. al. 2004 Glass et. al. 2006 number 2.7.1.69 phosphotransferase system glc + pep → g6p + pyr PTS MG041, 069, 429 5.3.1.9 glucose-6-phosphate isomerase g6p ↔ f6p PGI MG111 2.7.1.11 6-phosphofructokinase f6p + atp → fbp + adp PFK MG215 4.1.2.13 fructose-1,6-bisphosphate aldolase fbp ↔ gdp + dhp FBA MG023 5.3.1.1 triose-phosphate isomerase gdp ↔ dhp TPI MG431 glyceraldehyde-3-phosphate gdp + nad + p ↔ bpg + 1.2.1.12 GAP MG301 dehydrogenase nadh 2.7.2.3 phosphoglycerate kinase bpg + adp ↔ 3pg + atp PGK MG300 5.4.2.1 phosphoglycerate mutase 3pg ↔ 2pg GPM MG430 4.2.1.11 enolase 2pg ↔ pep ENO MG407 2.7.1.40 pyruvate kinase pep + adp → pyr + atp PYK MG216 1.1.1.27 lactate dehydrogenase pyr + nadh ↔ lac + nad LDH MG460 1.1.1.94 sn-glycerol-3-phosphate dehydrogenase dhp + nadh → g3p + nad GPS n.i. 2.3.1.15 sn-glycerol-3-phosphate acyltransferase g3p + pal → mag PLSb n.i. 2.3.1.51 1-acyl-sn-glycerol-3-phosphate mag + pal → dag PLSc MG212 acyltransferase 2.7.7.41 phosphatidate cytidyltransferase dag + ctp → cdp-dag + pp CDS MG437 cdp-dag + ser → pser + 2.7.8.8 phosphatidylserine synthase PSS n.i. cmp 4.1.1.65 phosphatidylserine decarboxylase pser → peta PSD n.i. -

Apramycin Sulfate Salt (A2024)
APRAMYCIN SULFATE ProductInformation Sigma Prod. No. A-2024 NH2 CAS NUMBER: 65710-07-8 X H2SO4 OH S PHYSICAL PROPERTIES: HO S S Absolute stereochemistry: Rotation (+) O R Appearance: Faint yellow powder R OH H H Molecular formula: C21H41N5O11 • x H2SO4 H N 2 NH2 H2N O O Molecular weight: 539.58 (free base) R S R S R S R S R S R HO R H O O N H H CH3 OH OH DESCRIPTION: Apramycin is an aminoglycoside antibiotic produced by a strain of Streptomyces tenebrarius. The compound is used as the sulfate in veterinary practice.1,2 STABILITY / STORAGE AS SUPPLIED: Store desiccated at 2-8EC. SOLUBILITY / SOLUTION STABILITY: Aqueous solutions at 25 mg/mL are faint yellow. No solution stability data are currently available. APPLICATIONS: 1. An oral composition of 20 g apramycin sulfate, 1 g pyridoxal HCl, 10 g sodium alginate, 10 g glycine and about 30 powdered apples were fed to calves and swine in order to treat diarrhea. Pyridoxal HCl was added to enhance the bactericidal activity of apramycin sulfate.3 2. Under the Federal Food, Drug and Cosmetics Act, feed containing 150 g apramycin per ton may be used to control colibacillosis.4 3. Apramycin is a potent inhibitor of protein synthesis in bacteria in vivo and in vitro. To a limited extent, it induces translation errors in cell-free systems from Escherichia coli supplemented with poly(U). Apramycin also inhibits the translocation step of protein synthesis in vivo (e.g., in protoplasts of Bacillus megaterium), and in vitro (e.g., in cell-free systems from E. -

7 | Carbohydrates and Glycobiology
7 | Carbohydrates and Glycobiology Aldoses and Ketoses; Representative monosaccharides. (a)Two trioses, an aldose and a ketose. The carbonyl group in each is shaded. • An aldose contains an aldehyde functionality • A ketose contains a ketone functionality The Glycosidic Bond • Two sugar molecules can be joined via a glycosidic bond between an anomeric carbon and a hydroxyl carbon • The glycosidic bond (an acetal) between monomers is less reactive than the hemiacetal at the second monomer – Second monomer, with the hemiacetal, is reducing – Anomeric carbon involved in the glycosidic linkage is nonreducing • The disaccharide formed upon condensation of two glucose molecules via 1 4 bond is called maltose • Formation of maltose. A disaccharide is formed from two monosaccharides (here, two molecules of D-glucose) when an —OH (alcohol) of one glucose molecule (right) condenses with the intramolecular hemiacetal of the other glucose molecule (left), with elimination of H2O and formation of a glycosidic bond. The reversal of this reaction is hydrolysis—attack by H2O on the glycosidic bond. The maltose molecule, shown here as an illustration, retains a reducing hemiacetal at the C-1 not involved in the glycosidic bond. Because mutarotation interconverts the α and β forms of the hemiacetal, the bonds at this position are sometimes depicted with wavy lines, as shown here, to indicate that the structure may be either α or β. Nonreducing Disaccharides • Two sugar molecules can be also joined via a glycosidic bond between two anomeric carbons • The product has two acetal groups and no hemiacetals • There are no reducing ends, this is a nonreducing sugar • Trehalose is a constituent of hemolymph of insects • Two common disaccharides. -

Nucleotide Sugars in Chemistry and Biology
molecules Review Nucleotide Sugars in Chemistry and Biology Satu Mikkola Department of Chemistry, University of Turku, 20014 Turku, Finland; satu.mikkola@utu.fi Academic Editor: David R. W. Hodgson Received: 15 November 2020; Accepted: 4 December 2020; Published: 6 December 2020 Abstract: Nucleotide sugars have essential roles in every living creature. They are the building blocks of the biosynthesis of carbohydrates and their conjugates. They are involved in processes that are targets for drug development, and their analogs are potential inhibitors of these processes. Drug development requires efficient methods for the synthesis of oligosaccharides and nucleotide sugar building blocks as well as of modified structures as potential inhibitors. It requires also understanding the details of biological and chemical processes as well as the reactivity and reactions under different conditions. This article addresses all these issues by giving a broad overview on nucleotide sugars in biological and chemical reactions. As the background for the topic, glycosylation reactions in mammalian and bacterial cells are briefly discussed. In the following sections, structures and biosynthetic routes for nucleotide sugars, as well as the mechanisms of action of nucleotide sugar-utilizing enzymes, are discussed. Chemical topics include the reactivity and chemical synthesis methods. Finally, the enzymatic in vitro synthesis of nucleotide sugars and the utilization of enzyme cascades in the synthesis of nucleotide sugars and oligosaccharides are briefly discussed. Keywords: nucleotide sugar; glycosylation; glycoconjugate; mechanism; reactivity; synthesis; chemoenzymatic synthesis 1. Introduction Nucleotide sugars consist of a monosaccharide and a nucleoside mono- or diphosphate moiety. The term often refers specifically to structures where the nucleotide is attached to the anomeric carbon of the sugar component.