Sarcoptes Scabiei: Past, Present and Future Larry G
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Genetic Epidemiology and Pathology of Raccoon-Derived Sarcoptes Mites from Urban Areas of Germany
Medical and Veterinary Entomology (2014) 28 (Suppl. 1), 98–103 Genetic epidemiology and pathology of raccoon-derived Sarcoptes mites from urban areas of Germany Z. RENTERÍA-SOLÍS1,A.M.MIN2, S. ALASAAD3,4, K. MÜLLER5, F.-U. MICHLER6, R. SCHMÄSCHKE7, U. WITTSTATT8, L. ROSSI2 andG. WIBBELT1 1Department of Wildlife Diseases, Leibniz Institute for Zoo and Wildlife Research, Berlin, Germany, 2Department of Animal Production, Epidemiology and Ecology, University of Turin, Grugliasco, Italy, 3Doñana Biological Station, Spanish National Research Council (Consejo Superior de Investigaciones Científicas), Seville, Spain, 4Institute of Evolutionary Biology and Environmental Studies, University of Zurich, Zurich, Switzerland, 5Clinic for Small Animals, Faculty of Veterinary Medicine, Free University of Berlin, Berlin, Germany, 6Group for Wildlife Research, Institute of Forest Botany and Forest Zoology, Technical University of Dresden, Tharandt, Germany, 7Institute of Parasitology, Faculty of Veterinary Medicine, University of Leipzig, Leipzig, Germany and 8Department of Animal Diseases, Zoonoses and Infection Diagnostics, Landeslabor Berlin–Brandenburg, Berlin, Germany Abstract. The raccoon, Procyon lotor (Carnivora: Procyonidae), is an invasive species that is spreading throughout Europe, in which Germany represents its core area. Here, raccoons mostly live in rural regions, but some urban populations are already established, such as in the city of Kassel, or are starting to build up, such as in Berlin. The objective of this study was to investigate Sarcoptes (Sarcoptiformes: Sarcoptidae) infections in racoons in these two urban areas and to identify the putative origin of the parasite. Parasite morphology, and gross and histopathological examinations of diseased skin tissue were consistent with Sarcoptes scabiei infection. Using nine microsatellite markers, we genotyped individual mites from five raccoons and compared them with Sarcoptes mites derived from fox, wild boar and Northern chamois, originating from Italy and Switzerland. -

Transcriptomic Analysis of the Temporal Host Response to Skin
Burgess et al. BMC Genomics 2010, 11:624 http://www.biomedcentral.com/1471-2164/11/624 RESEARCH ARTICLE Open Access Transcriptomic analysis of the temporal host response to skin infestation with the ectoparasitic mite Psoroptes ovis Stewart TG Burgess*, David Frew, Francesca Nunn, Craig A Watkins, Tom N McNeilly, Alasdair J Nisbet, John F Huntley Abstract Background: Infestation of ovine skin with the ectoparasitic mite Psoroptes ovis results in a rapid cutaneous immune response, leading to the crusted skin lesions characteristic of sheep scab. Little is known regarding the mechanisms by which such a profound inflammatory response is instigated and to identify novel vaccine and drug targets a better understanding of the host-parasite relationship is essential. The main objective of this study was to perform a combined network and pathway analysis of the in vivo skin response to infestation with P. ovis to gain a clearer understanding of the mechanisms and signalling pathways involved. Results: Infestation with P. ovis resulted in differential expression of 1,552 genes over a 24 hour time course. Clustering by peak gene expression enabled classification of genes into temporally related groupings. Network and pathway analysis of clusters identified key signalling pathways involved in the host response to infestation. The analysis implicated a number of genes with roles in allergy and inflammation, including pro-inflammatory cytokines (IL1A, IL1B, IL6, IL8 and TNF) and factors involved in immune cell activation and recruitment (SELE, SELL, SELP, ICAM1, CSF2, CSF3, CCL2 and CXCL2). The analysis also highlighted the influence of the transcription factors NF-kB and AP-1 in the early pro-inflammatory response, and demonstrated a bias towards a Th2 type immune response. -

Gamasid Mites
NATIONAL RESEARCH TOMSK STATE UNIVERSITY BIOLOGICAL INSTITUTE RUSSIAN ACADEMY OF SCIENCE ZOOLOGICAL INSTITUTE M.V. Orlova, M.K. Stanyukovich, O.L. Orlov GAMASID MITES (MESOSTIGMATA: GAMASINA) PARASITIZING BATS (CHIROPTERA: RHINOLOPHIDAE, VESPERTILIONIDAE, MOLOSSIDAE) OF PALAEARCTIC BOREAL ZONE (RUSSIA AND ADJACENT COUNTRIES) Scientific editor Andrey S. Babenko, Doctor of Science, professor, National Research Tomsk State University Tomsk Publishing House of Tomsk State University 2015 UDK 576.89:599.4 BBK E693.36+E083 Orlova M.V., Stanyukovich M.K., Orlov O.L. Gamasid mites (Mesostigmata: Gamasina) associated with bats (Chiroptera: Vespertilionidae, Rhinolophidae, Molossidae) of boreal Palaearctic zone (Russia and adjacent countries) / Scientific editor A.S. Babenko. – Tomsk : Publishing House of Tomsk State University, 2015. – 150 р. ISBN 978-5-94621-523-7 Bat gamasid mites is a highly specialized ectoparasite group which is of great interest due to strong isolation and other unique features of their hosts (the ability to fly, long distance migration, long-term hibernation). The book summarizes the results of almost 60 years of research and is the most complete summary of data on bat gamasid mites taxonomy, biology, ecol- ogy. It contains the first detailed description of bat wintering experience in sev- eral regions of the boreal Palaearctic. The book is addressed to zoologists, ecologists, experts in environmental protection and biodiversity conservation, students and teachers of biology, vet- erinary science and medicine. UDK 576.89:599.4 -

Sarcoptes Scabiei, Psoroptes Ovis
Mounsey et al. Parasites & Vectors 2012, 5:3 http://www.parasitesandvectors.com/content/5/1/3 RESEARCH Open Access Quantitative PCR-based genome size estimation of the astigmatid mites Sarcoptes scabiei, Psoroptes ovis and Dermatophagoides pteronyssinus Kate E Mounsey1,2, Charlene Willis1, Stewart TG Burgess3, Deborah C Holt4, James McCarthy1,5 and Katja Fischer1* Abstract Background: The lack of genomic data available for mites limits our understanding of their biology. Evolving high- throughput sequencing technologies promise to deliver rapid advances in this area, however, estimates of genome size are initially required to ensure sufficient coverage. Methods: Quantitative real-time PCR was used to estimate the genome sizes of the burrowing ectoparasitic mite Sarcoptes scabiei, the non-burrowing ectoparasitic mite Psoroptes ovis, and the free-living house dust mite Dermatophagoides pteronyssinus. Additionally, the chromosome number of S. scabiei was determined by chromosomal spreads of embryonic cells derived from single eggs. Results: S. scabiei cells were shown to contain 17 or 18 small (< 2 μM) chromosomes, suggesting an XO sex- determination mechanism. The average estimated genome sizes of S. scabiei and P. ovis were 96 (± 7) Mb and 86 (± 2) Mb respectively, among the smallest arthropod genomes reported to date. The D. pteronyssinus genome was estimated to be larger than its parasitic counterparts, at 151 Mb in female mites and 218 Mb in male mites. Conclusions: This data provides a starting point for understanding the genetic organisation and evolution of these astigmatid mites, informing future sequencing projects. A comparitive genomic approach including these three closely related mites is likely to reveal key insights on mite biology, parasitic adaptations and immune evasion. -
![Sarcoptes Scabiei] Genbank: AWM31286.1 Identical Proteins FASTA Graphics](https://docslib.b-cdn.net/cover/0198/sarcoptes-scabiei-genbank-awm31286-1-identical-proteins-fasta-graphics-170198.webp)
Sarcoptes Scabiei] Genbank: AWM31286.1 Identical Proteins FASTA Graphics
11/5/2020 cytochrome c oxidase subunit 1, partial (mitochondrion) [Sarcoptes sca - Protein - NCBI Protein COVID-19 is an emerging, rapidly evolving situation. Get the latest public health information from CDC: https://www.coronavirus.gov . Get the latest research from NIH: https://www.nih.gov/coronavirus. GenPept cytochrome c oxidase subunit 1, partial (mitochondrion) [Sarcoptes scabiei] GenBank: AWM31286.1 Identical Proteins FASTA Graphics Go to: LOCUS AWM31286 96 aa linear INV 28-MAY-2018 DEFINITION cytochrome c oxidase subunit 1, partial (mitochondrion) [Sarcoptes scabiei]. ACCESSION AWM31286 VERSION AWM31286.1 DBSOURCE accession MH077557.1 KEYWORDS . SOURCE mitochondrion Sarcoptes scabiei ORGANISM Sarcoptes scabiei Eukaryota; Metazoa; Ecdysozoa; Arthropoda; Chelicerata; Arachnida; Acari; Acariformes; Sarcoptiformes; Astigmata; Psoroptidia; Sarcoptoidea; Sarcoptidae; Sarcoptinae; Sarcoptes. REFERENCE 1 (residues 1 to 96) AUTHORS Lastuti,N.D.R., Rohman,A., Hastutiek,P., Desiandura,K. and Handijatno,D. TITLE Sequence analysis of cytochrome c oxidase subunit 1 gene of Sarcoptes scabiei isolated from goat and rabbit in East Java, Indonesia JOURNAL Unpublished REFERENCE 2 (residues 1 to 96) AUTHORS Lastuti,N.D.R., Rohman,A., Hastutiek,P., Desiandura,K. and Handijatno,D. TITLE Direct Submission JOURNAL Submitted (15-MAR-2018) Department of Veterinary Parasitology, Faculty of Veterinary Medicine, Universitas Airlangga, Kampus C Unair, Jl. Mulyorejo, Surabaya, East Java 60115, Indonesia FEATURES Location/Qualifiers source 1..96 /organism="Sarcoptes scabiei" /organelle="mitochondrion" /host="goat" /db_xref="taxon:52283" /country="Indonesia: Lamongan, East Java" Protein <1..>96 /product="cytochrome c oxidase subunit 1" Region 1..>75 /region_name="Heme_Cu_Oxidase_I" /note="Heme-copper oxidase subunit I. Heme-copper oxidases are transmembrane protein complexes in the respiratory chains of prokaryotes and mitochondria which catalyze the reduction of O2 and simultaneously pump protons across the membrane. -

Arachnida, Solifugae) with Special Focus on Functional Analyses and Phylogenetic Interpretations
HISTOLOGY AND ULTRASTRUCTURE OF SOLIFUGES Comparative studies of organ systems of solifuges (Arachnida, Solifugae) with special focus on functional analyses and phylogenetic interpretations HISTOLOGIE UND ULTRASTRUKTUR DER SOLIFUGEN Vergleichende Studien an Organsystemen der Solifugen (Arachnida, Solifugae) mit Schwerpunkt auf funktionellen Analysen und phylogenetischen Interpretationen I N A U G U R A L D I S S E R T A T I O N zur Erlangung des akademischen Grades doctor rerum naturalium (Dr. rer. nat.) an der Mathematisch-Naturwissenschaftlichen Fakultät der Ernst-Moritz-Arndt-Universität Greifswald vorgelegt von Anja Elisabeth Klann geboren am 28.November 1976 in Bremen Greifswald, den 04.06.2009 Dekan ........................................................................................................Prof. Dr. Klaus Fesser Prof. Dr. Dr. h.c. Gerd Alberti Erster Gutachter .......................................................................................... Zweiter Gutachter ........................................................................................Prof. Dr. Romano Dallai Tag der Promotion ........................................................................................15.09.2009 Content Summary ..........................................................................................1 Zusammenfassung ..........................................................................5 Acknowledgments ..........................................................................9 1. Introduction ............................................................................ -

Arthropod Parasites in Domestic Animals
ARTHROPOD PARASITES IN DOMESTIC ANIMALS Abbreviations KINGDOM PHYLUM CLASS ORDER CODE Metazoa Arthropoda Insecta Siphonaptera INS:Sip Mallophaga INS:Mal Anoplura INS:Ano Diptera INS:Dip Arachnida Ixodida ARA:Ixo Mesostigmata ARA:Mes Prostigmata ARA:Pro Astigmata ARA:Ast Crustacea Pentastomata CRU:Pen References Ashford, R.W. & Crewe, W. 2003. The parasites of Homo sapiens: an annotated checklist of the protozoa, helminths and arthropods for which we are home. Taylor & Francis. Taylor, M.A., Coop, R.L. & Wall, R.L. 2007. Veterinary Parasitology. 3rd edition, Blackwell Pub. HOST-PARASITE CHECKLIST Class: MAMMALIA [mammals] Subclass: EUTHERIA [placental mammals] Order: PRIMATES [prosimians and simians] Suborder: SIMIAE [monkeys, apes, man] Family: HOMINIDAE [man] Homo sapiens Linnaeus, 1758 [man] ARA:Ast Sarcoptes bovis, ectoparasite (‘milker’s itch’)(mange mite) ARA:Ast Sarcoptes equi, ectoparasite (‘cavalryman’s itch’)(mange mite) ARA:Ast Sarcoptes scabiei, skin (mange mite) ARA:Ixo Ixodes cornuatus, ectoparasite (scrub tick) ARA:Ixo Ixodes holocyclus, ectoparasite (scrub tick, paralysis tick) ARA:Ixo Ornithodoros gurneyi, ectoparasite (kangaroo tick) ARA:Pro Cheyletiella blakei, ectoparasite (mite) ARA:Pro Cheyletiella parasitivorax, ectoparasite (rabbit fur mite) ARA:Pro Demodex brevis, sebacceous glands (mange mite) ARA:Pro Demodex folliculorum, hair follicles (mange mite) ARA:Pro Trombicula sarcina, ectoparasite (black soil itch mite) INS:Ano Pediculus capitis, ectoparasite (head louse) INS:Ano Pediculus humanus, ectoparasite (body -

Wildlife Parasitology in Australia: Past, Present and Future
CSIRO PUBLISHING Australian Journal of Zoology, 2018, 66, 286–305 Review https://doi.org/10.1071/ZO19017 Wildlife parasitology in Australia: past, present and future David M. Spratt A,C and Ian Beveridge B AAustralian National Wildlife Collection, National Research Collections Australia, CSIRO, GPO Box 1700, Canberra, ACT 2601, Australia. BVeterinary Clinical Centre, Faculty of Veterinary and Agricultural Sciences, University of Melbourne, Werribee, Vic. 3030, Australia. CCorresponding author. Email: [email protected] Abstract. Wildlife parasitology is a highly diverse area of research encompassing many fields including taxonomy, ecology, pathology and epidemiology, and with participants from extremely disparate scientific fields. In addition, the organisms studied are highly dissimilar, ranging from platyhelminths, nematodes and acanthocephalans to insects, arachnids, crustaceans and protists. This review of the parasites of wildlife in Australia highlights the advances made to date, focussing on the work, interests and major findings of researchers over the years and identifies current significant gaps that exist in our understanding. The review is divided into three sections covering protist, helminth and arthropod parasites. The challenge to document the diversity of parasites in Australia continues at a traditional level but the advent of molecular methods has heightened the significance of this issue. Modern methods are providing an avenue for major advances in documenting and restructuring the phylogeny of protistan parasites in particular, while facilitating the recognition of species complexes in helminth taxa previously defined by traditional morphological methods. The life cycles, ecology and general biology of most parasites of wildlife in Australia are extremely poorly understood. While the phylogenetic origins of the Australian vertebrate fauna are complex, so too are the likely origins of their parasites, which do not necessarily mirror those of their hosts. -

(Acari: Oribatida: Galumnoidea) in Japan
Taxonomic studies on oribatid mites of the genera Neoribates, Trichogalumna and Cosmogalumna (Acari: Oribatida: Title Galumnoidea) in Japan Author(s) 萩野, 航 Citation 北海道大学. 博士(理学) 甲第13140号 Issue Date 2018-03-22 DOI 10.14943/doctoral.k13140 Doc URL http://hdl.handle.net/2115/72711 Type theses (doctoral) File Information Wataru_Hagino.pdf Instructions for use Hokkaido University Collection of Scholarly and Academic Papers : HUSCAP Doctoral Dissertation Taxonomic studies on oribatid mites of the genera Neoribates, Trichogalumna and Cosmogalumna (Acari: Oribatida: Galumnoidea) in Japan (, , ()) Wataru HAGINO Graduate School of Science, Hokkaido University Department of Natural History Sciences 2018 March Taxonomic studies of three genera of Oribatida in Japan Table of Contents General Introduction ...................................................................................................... 1 Materials and Methods ................................................................................................ 13 Taxonomy ...................................................................................................................... 17 Superfamily Galumnoidea Jacot, 1925 ........................................................................... 17 Family Parakalummidae Grandjean, 1936 ................................................................. 17 Genus Neoribates Berlese, 1915 .............................................................................. 17 Neoribates incisus Hagino, Shimano, and Aoki, 2016 ........................................ -

Comparative Genomics Reveals the Origins and Diversity of Arthropod Immune Systems
bioRxiv preprint doi: https://doi.org/10.1101/010942; this version posted October 30, 2014. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Comparative genomics reveals the origins and diversity of arthropod immune systems William J. Palmer* and Francis M. Jiggins Department of Genetics, University of Cambridge, Downing Street, Cambridge CB2 3EH UK * corresponding author; [email protected] 1 bioRxiv preprint doi: https://doi.org/10.1101/010942; this version posted October 30, 2014. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Abstract While the innate immune system of insects is well-studied, comparatively little is known about how other arthropods defend themselves against infection. We have characterised key immune components in the genomes of five chelicerates, a myriapod and a crustacean. We found clear traces of an ancient origin of innate immunity, with some arthropods having Toll- like receptors and C3-complement factors that are more closely related in sequence or structure to vertebrates than other arthropods. Across the arthropods some components of the immune system, like the Toll signalling pathway, are highly conserved. However, there is also remarkable diversity. The chelicerates apparently lack the Imd signalling pathway and BGRPs – a key class of pathogen recognition receptors. Many genes have large copy number variation across species, and this may sometimes be accompanied by changes in function. For example, peptidoglycan recognition proteins (PGRPs) have frequently lost their catalytic activity and switch between secreted and intracellular forms. -
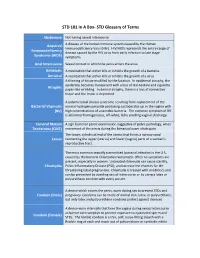
STD Glossary of Terms
STD 101 In A Box- STD Glossary of Terms Abstinence Not having sexual intercourse Acquired A disease of the human immune system caused by the Human Immunodeficiency Virus (HIV). HIV/AIDS represents the entire range of Immunodeficiency disease caused by the HIV virus from early infection to late stage Syndrome (AIDS) symptoms. Anal Intercourse Sexual contact in which the penis enters the anus. Antibiotic A medication that either kills or inhibits the growth of a bacteria. Antiviral A medication that either kills or inhibits the growth of a virus. A thinning of tissue modified by the location. In epidermal atrophy, the epidermis becomes transparent with a loss of skin texture and cigarette Atrophic paper-like wrinkling. In dermal atrophy, there is a loss of connective tissue and the lesion is depressed. A polymicrobial clinical syndrome resulting from replacement of the Bacterial Vaginosis normal hydrogen peroxide producing Lactobacillus sp. in the vagina with (BV) high concentrations of anaerobic bacteria. The common symptom of BV is abnormal homogeneous, off-white, fishy smelling vaginal discharge. Cervical Motion A sign found on pelvic examination suggestive of pelvic pathology; when Tenderness (CMT) movement of the cervix during the bimanual exam elicits pain. The lower, cylindrical end of the uterus that forms a narrow canal Cervix connecting the upper (uterus) and lower (vagina) parts of a woman's reproductive tract. The most common sexually transmitted bacterial infection in the U.S., caused by the bacteria Chlamydia trachomatis. Often no symptoms are present, especially in women. Untreated chlamydia can cause sterility, Chlamydia Pelvic Inflammatory Disease (PID), and increase the chances for life- threatening tubal pregnancies. -

The External Parasites of Birds: a Review
THE EXTERNAL PARASITES OF BIRDS: A REVIEW BY ELIZABETH M. BOYD Birds may harbor a great variety and numher of ectoparasites. Among the insects are biting lice (Mallophaga), fleas (Siphonaptera), and such Diptera as hippohoscid flies (Hippohoscidae) and the very transitory mosquitoes (Culicidae) and black flies (Simuliidae), which are rarely if every caught on animals since they fly off as soon as they have completed their blood-meal. One may also find, in birds ’ nests, bugs of the hemipterous family Cimicidae, and parasitic dipterous larvae that attack nestlings. Arachnida infesting birds comprise the hard ticks (Ixodidae), soft ticks (Argasidae), and certain mites. Most ectoparasites are blood-suckers; only the Ischnocera lice and some species of mites subsist on skin components. The distribution of ectoparasites on the host varies with the parasite concerned. Some show no habitat preference while others tend to confine themselves to, or even are restricted to, definite areas on the body. A list of 198 external parasites for 2.55 species and/or subspecies of birds east of the Mississippi has been compiled by Peters (1936) from files of the Bureau of Entomology and Plant Quarantine between 1928 and 1935. Fleas and dipterous larvae were omitted from this list. According to Peters, it is possible to collect three species of lice, one or two hippoboscids, and several types of mites on a single bird. He records as many as 15 species of ectoparasites each from the Bob-white (Co&us uirginianus), Song Sparrow (Melospiza melodia), and Robin (Turdus migratorius). The lice and plumicolous mites, however, are typically the most abundant forms present on avian hosts.