Methods in Reproductive Aquaculture Marine and Freshwater Species
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Aalseth Aaron Aarup Aasen Aasheim Abair Abanatha Abandschon Abarca Abarr Abate Abba Abbas Abbate Abbe Abbett Abbey Abbott Abbs
BUSCAPRONTA www.buscapronta.com ARQUIVO 35 DE PESQUISAS GENEALÓGICAS 306 PÁGINAS – MÉDIA DE 98.500 SOBRENOMES/OCORRÊNCIA Para pesquisar, utilize a ferramenta EDITAR/LOCALIZAR do WORD. A cada vez que você clicar ENTER e aparecer o sobrenome pesquisado GRIFADO (FUNDO PRETO) corresponderá um endereço Internet correspondente que foi pesquisado por nossa equipe. Ao solicitar seus endereços de acesso Internet, informe o SOBRENOME PESQUISADO, o número do ARQUIVO BUSCAPRONTA DIV ou BUSCAPRONTA GEN correspondente e o número de vezes em que encontrou o SOBRENOME PESQUISADO. Número eventualmente existente à direita do sobrenome (e na mesma linha) indica número de pessoas com aquele sobrenome cujas informações genealógicas são apresentadas. O valor de cada endereço Internet solicitado está em nosso site www.buscapronta.com . Para dados especificamente de registros gerais pesquise nos arquivos BUSCAPRONTA DIV. ATENÇÃO: Quando pesquisar em nossos arquivos, ao digitar o sobrenome procurado, faça- o, sempre que julgar necessário, COM E SEM os acentos agudo, grave, circunflexo, crase, til e trema. Sobrenomes com (ç) cedilha, digite também somente com (c) ou com dois esses (ss). Sobrenomes com dois esses (ss), digite com somente um esse (s) e com (ç). (ZZ) digite, também (Z) e vice-versa. (LL) digite, também (L) e vice-versa. Van Wolfgang – pesquise Wolfgang (faça o mesmo com outros complementos: Van der, De la etc) Sobrenomes compostos ( Mendes Caldeira) pesquise separadamente: MENDES e depois CALDEIRA. Tendo dificuldade com caracter Ø HAMMERSHØY – pesquise HAMMERSH HØJBJERG – pesquise JBJERG BUSCAPRONTA não reproduz dados genealógicos das pessoas, sendo necessário acessar os documentos Internet correspondentes para obter tais dados e informações. DESEJAMOS PLENO SUCESSO EM SUA PESQUISA. -

39400 13-11 Legala
Government Gazette Staatskoerant REPUBLIC OF SOUTH AFRICA REPUBLIEK VAN SUID-AFRIKA November Vol. 605 Pretoria, 13 2015 November No. 39400 PART 1 OF 2 LEGAL NOTICES A WETLIKE KENNISGEWINGS ISSN 1682-5843 N.B. The Government Printing Works will 39400 not be held responsible for the quality of “Hard Copies” or “Electronic Files” submitted for publication purposes 9 771682 584003 AIDS HELPLINE: 0800-0123-22 Prevention is the cure 2 No. 39400 GOVERNMENT GAZETTE, 13 NOVEMBER 2015 WARNING!!! To all suppliers and potential suppliers of goods to the Government Printing Works The Government Printing Works would like to warn members of the public against an organised syndicate(s) scamming unsuspecting members of the public and claiming to act on behalf of the Government Printing Works. One of the ways in which the syndicate operates is by requesting quotations for various goods and services on a quotation form with the logo of the Government Printing Works. Once the official order is placed the syndicate requesting upfront payment before delivery will take place. Once the upfront payment is done the syndicate do not deliver the goods and service provider then expect payment from Government Printing Works. Government Printing Works condemns such illegal activities and encourages service providers to confirm the legitimacy of purchase orders with GPW SCM, prior to processing and delivery of goods. To confirm the legitimacy of purchase orders, please contact: Renny Chetty (012) 748-6375 ([email protected]), Anna-Marie du Toit (012) 748-6292 ([email protected]) and Siraj Rizvi (012) 748-6380 ([email protected]) This gazette is also available free online at www.gpwonline.co.za STAATSKOERANT, 13 NOVEMBER 2015 No. -

Molecular Phylogeny of Echiuran Worms (Phylum: Annelida) Reveals Evolutionary Pattern of Feeding Mode and Sexual Dimorphism
Molecular Phylogeny of Echiuran Worms (Phylum: Annelida) Reveals Evolutionary Pattern of Feeding Mode and Sexual Dimorphism Ryutaro Goto1,2*, Tomoko Okamoto2, Hiroshi Ishikawa3, Yoichi Hamamura4, Makoto Kato2 1 Department of Marine Ecosystem Dynamics, Atmosphere and Ocean Research Institute, The University of Tokyo, Kashiwa, Chiba, Japan, 2 Graduate School of Human and Environmental Studies, Kyoto University, Kyoto, Japan, 3 Uwajima, Ehime, Japan, 4 Kure, Hiroshima, Japan Abstract The Echiura, or spoon worms, are a group of marine worms, most of which live in burrows in soft sediments. This annelid- like animal group was once considered as a separate phylum because of the absence of segmentation, although recent molecular analyses have placed it within the annelids. In this study, we elucidate the interfamily relationships of echiuran worms and their evolutionary pattern of feeding mode and sexual dimorphism, by performing molecular phylogenetic analyses using four genes (18S, 28S, H3, and COI) of representatives of all extant echiuran families. Our results suggest that Echiura is monophyletic and comprises two unexpected groups: [Echiuridae+Urechidae+Thalassematidae] and [Bone- lliidae+Ikedidae]. This grouping agrees with the presence/absence of marked sexual dimorphism involving dwarf males and the paired/non-paired configuration of the gonoducts (genital sacs). Furthermore, the data supports the sister group relationship of Echiuridae and Urechidae. These two families share the character of having anal chaetae rings around the posterior trunk as a synapomorphy. The analyses also suggest that deposit feeding is a basal feeding mode in echiurans and that filter feeding originated once in the common ancestor of Urechidae. Overall, our results contradict the currently accepted order-level classification, especially in that Echiuroinea is polyphyletic, and provide novel insights into the evolution of echiuran worms. -

Continuous Light on Tomato
CONTINUOUS LIGHT ON TOMATO From Gene to Yield Aarón I. Vélez-Ramírez i Thesis committee Promotor Prof. Dr Harro J. Bouwmeester Professor of Plant Physiology Wageningen University Co-promotors Dr Wim van Ieperen Assistant professor, Horticulture and Product Physiology Wageningen University Dr Dick Vreugdenhil Associate professor, Laboratory of Plant Physiology Wageningen University Other members Prof. Dr Ton Bisseling, Wageningen University Prof. Dr Roberta Croce, VU University Amsterdam, The Netherlands Prof. Dr Gerrit T.S. Beemster, University of Antwerp, Belgium Dr. Ronald Pierik, Utrecht University, The Netherlands This research was conducted under the auspices of the Graduate School of Experimental Plant Sciences (EPS). ii CONTINUOUS LIGHT ON TOMATO From Gene to Yield Aarón I. Vélez-Ramírez Thesis submitted in fulfillment of the requirements for the degree of doctor at Wageningen University by the authority of the Rector Magnificus Prof. Dr M.J. Kropff, in the presence of the Thesis Committee appointed by the Academic Board to be defended in public on Friday 19 September 2014 at 11 a.m. in the Aula. iii Aarón I. Vélez Ramírez Continuous Light on Tomato. From Gene to Yield 214 pages PhD thesis, Wageningen University , Wageningen, NL (2014) With references, summaries in English and Dutch ISBN 978-94-6257-078-8 iv Contents Chapter 1 | 1 General Introduction. Continuous-light-induced injury in tomato: An old enigma 13 Chapter 2 | Plants under continuous light Chapter 3 | 29 Continuous light as a way to increase greenhouse tomato production: -

Anti-BACE1 and Antimicrobial Activities of Steroidal Compounds Isolated from Marine Urechis Unicinctus
marine drugs Article Anti-BACE1 and Antimicrobial Activities of Steroidal Compounds Isolated from Marine Urechis unicinctus Yong-Zhe Zhu 1, Jing-Wen Liu 1, Xue Wang 2, In-Hong Jeong 3, Young-Joon Ahn 4 and Chuan-Jie Zhang 5,* ID 1 College of Chemistry and Pharmaceutical Science, Qingdao Agricultural University, Changcheng Rd, Chengyang district, Qingdao 266109, China; [email protected] (Y.-Z.Z.); [email protected] (J.-W.L.) 2 School of Pharmaceutical Sciences, Wenzhou Medical University, Wenzhou 325035, China; [email protected] 3 Division of Crop Protection, National Institute of Agricultural Science, Rural Development Administration, Jeollabuk-do 55365, Korea; [email protected] 4 Department of Agricultural Biotechnology, Seoul National University, 599 Gwanak-ro, Silim-dong, Gwanak-Gu, Seoul 151742, Korea; [email protected] 5 Department of Plant Science, University of Connecticut, 1376 Storrs Road, U-4163, Storrs, CT 06269, USA * Correspondence: [email protected]; Tel.: +1-860-486-2924 Received: 27 December 2017; Accepted: 12 March 2018; Published: 14 March 2018 Abstract: The human β-site amyloid cleaving enzyme (BACE1) has been considered as an effective drug target for treatment of Alzheimer’s disease (AD). In this study, Urechis unicinctus (U. unicinctus), which is a Far East specialty food known as innkeeper worm, ethanol extract was studied by bioassay-directed fractionation and isolation to examine its potential β-site amyloid cleaving enzyme inhibitory and antimicrobial activity. The following compounds were characterized: hecogenin, cholest-4-en-3-one, cholesta-4,6-dien-3-ol, and hurgadacin. These compounds were identified by their mass spectrometry, 1H, and 13C NMR spectral data, comparing those data with NIST/EPA/NIH Mass spectral database (NIST11) and published values. -
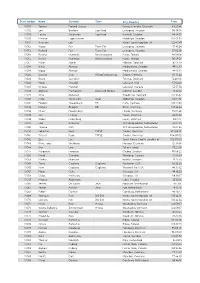
Start Number Name Surname Team City, Country Time 11001 Tommy
Start number Name Surname Team City, Country Time 11001 Tommy Frølund Jensen Kongens lyngby, Denmark 89:20:46 11002 Emil Boström Lag Nord Linköping, Sweden 98:19:04 11003 Lovisa Johansson Lag Nord Burträsk, Sweden 98:19:02 11005 Maurice Lagershausen Huddinge, Sweden 102:01:54 11011 Hyun jun Lee Korea, (south) republic of 122:40:49 11012 Viggo Fält Team Fält Linköping, Sweden 57:45:24 11013 Richard Fält Team Fält Linköping, Sweden 57:45:28 11014 Rasmus Nummela Rackmannarna Pjelax, Finland 98:59:06 11015 Rafael Nummela Rackmannarna Pjelax, Finland 98:59:06 11017 Marie Zølner Hillerød, Denmark 82:11:49 11018 Niklas Åkesson Mellbystrand, Sweden 99:45:22 11019 Viggo Åkesson Mellbystrand, Sweden 99:45:22 11020 Damian Sulik #StopComplaining Siegen, Germany 101:13:24 11023 Bruno Svendsen Tylstrup, Denmark 74:48:08 11024 Philip Oswald Lakeland, USA 55:52:02 11025 Virginia Marshall Lakefield, Canada 55:57:54 11026 Andreas Mathiasson Backyard Heroes Ödsmål, Sweden 75:12:02 11028 Stine Andersen Brædstrup, Denmark 104:35:37 11030 Patte Johansson Tiger Södertälje, Sweden 80:24:51 11031 Frederic Wiesenbach BB Dahn, Germany 101:21:50 11032 Romea Brugger BB Berlin, Germany 101:22:44 11034 Oliver Freudenberg Heyda, Germany 75:07:48 11035 Jan Hennig Farum, Denmark 82:11:50 11036 Robert Falkenberg Farum, Denmark 82:11:51 11037 Edo Boorsma Sint annaparochie, Netherlands 123:17:34 11038 Trijneke Stuit Sint annaparochie, Netherlands 123:17:35 11040 Sebastian Keck TATSE Gieäen, Germany 104:08:01 11041 Tatjana Kage TATSE Gieäen, Germany 104:08:02 11042 Eun Lee -

Function of the Anal Sacs and Mid-Gut in Mitochondrial Sulphide
This article was downloaded by: [Qingdao Institute of Biomass Energy and Bioprocess Technology] On: 28 November 2012, At: 01:39 Publisher: Taylor & Francis Informa Ltd Registered in England and Wales Registered Number: 1072954 Registered office: Mortimer House, 37-41 Mortimer Street, London W1T 3JH, UK Marine Biology Research Publication details, including instructions for authors and subscription information: http://www.tandfonline.com/loi/smar20 Function of the anal sacs and mid-gut in mitochondrial sulphide metabolism in the echiuran worm Urechis unicinctus Yu-Bin Ma a b , Zhi-Feng Zhang a , Ming-Yu Shao a , Kyoung-Ho Kang c , Li-Tao Zhang a , Xiao-Li Shi a & Ying-Ping Dong a a Key Laboratory of Marine Genetics and Breeding of Ministry of Education, Ocean University of China, Qingdao, China b Shandong Provincial Key Laboratory of Energy Genetics, Key Laboratory of Biofuels, Qingdao Institute of Bioenergy and Bioprocess Technology, Chinese Academy of Sciences, Qingdao, China c Department of Aquaculture, Chonnam National University, Yeosu, South Korea Version of record first published: 26 Sep 2012. To cite this article: Yu-Bin Ma, Zhi-Feng Zhang, Ming-Yu Shao, Kyoung-Ho Kang, Li-Tao Zhang, Xiao-Li Shi & Ying-Ping Dong (2012): Function of the anal sacs and mid-gut in mitochondrial sulphide metabolism in the echiuran worm Urechis unicinctus , Marine Biology Research, 8:10, 1026-1031 To link to this article: http://dx.doi.org/10.1080/17451000.2012.707320 PLEASE SCROLL DOWN FOR ARTICLE Full terms and conditions of use: http://www.tandfonline.com/page/terms-and-conditions This article may be used for research, teaching, and private study purposes. -
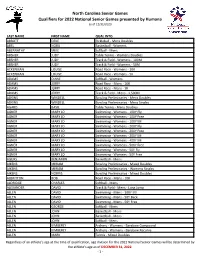
2022 NC National Qualifiers with Events As Of
North Carolina Senior Games Qualifiers for 2022 National Senior Games presented by Humana as of 12/31/2020 GFL LAST NAME FIRST NAME QUAL INTO ABBOTT DEKE Pickleball - Mens Doubles ABEL NORA Basketball - Womens ABERNATHY MIKE Softball - Mens ABSHER JUDY Table Tennis - Womens Doubles ABSHER JUDY Track & Field - Womens - 100M ABSHER JUDY Track & Field - Womens - 50M ACKERMAN LOUISE Road Race - Womens - 10K ACKERMAN LOUISE Road Race - Womens - 5K ADAMS DIANE Softball - Womens ADAMS JERRY Road Race - Mens - 10K ADAMS JERRY Road Race - Mens - 5K ADAMS JERRY Track & Field - Mens - 1,500M ADKINS WINDELL Bowling Preliminaries - Mens Doubles ADKINS WINDELL Bowling Preliminaries - Mens Singles AGARD BASIL Table Tennis - Mens Doubles AGNER MARY JO Swimming - Womens - 100Y Fly AGNER MARY JO Swimming - Womens - 100Y Free AGNER MARY JO Swimming - Womens - 100Y IM AGNER MARY JO Swimming - Womens - 200Y Fly AGNER MARY JO Swimming - Womens - 200Y Free AGNER MARY JO Swimming - Womens - 200Y IM AGNER MARY JO Swimming - Womens - 400Y IM AGNER MARY JO Swimming - Womens - 500Y Free AGNER MARY JO Swimming - Womens - 50Y Fly AGNER MARY JO Swimming - Womens - 50Y Free AGURS BENJAMIN Basketball - Mens AIKENS MIRIAM Bowling Preliminaries - Mixed Doubles AIKENS MIRIAM Bowling Preliminaries - Womens Singles AIKENS NORRIS Bowling Preliminaries - Mixed Doubles ALBRITTON BOB Road Race - Mens - 10K ALDRIDGE CHARLES Softball - Mens ALEXANDER DAVID Track & Field - Mens - Long Jump ALLEN DAVID Swimming - Mens - 100Y IM ALLEN DAVID Swimming - Mens - 50Y Back ALLEN DAVID Swimming - Mens - 50Y Free ALLEN GEORGE Softball - Mens ALLEN JOHN Basketball - Mens ALLEN JOHN Basketball - Mens ALLEN JOHN Softball - Mens ALLEN KIMBERLY Archery - Womens - Barebow Compound ALLEN KIMBERLY Archery - Womens - Barebow Recurve ALLEN MARK Tennis - Mixed Doubles Regardless of an athlete's age at the time of qualification, age division for the 2022 National Senior Games will be determined by the athlete’s age as of DECEMBER 31, 2022. -

Zachary Steel
MENTAL DISORDER AMONGST PEOPLE OF VIETNAMESE BACKGROUND: PREVALENCE, TRAUMA AND CULTURE Zachary Steel A thesis submitted in fulfilment of the requirements for the degree of Doctor of Philosophy School of Psychiatry Faculty of Medicine, University of New South Wales Sydney Australia June 2008 ii Abstract The role that culture and trauma plays in shaping mental health outcomes continues to dominate debate in the field of transcultural and post-conflict mental health. The broad aim of this thesis is to investigate key issues relevant to these two factors in relation to the Vietnamese. A meta-analysis of international epidemiological research indicated that countries of North and South East Asia appear to manifest low rates of mental disorder compared to English-speaking countries. A meta-regression analysis of research undertaken specifically with refugee and conflict-affected populations, confirmed a robust association between torture and general trauma and risk to mental disorder. The thesis then examines data from three population-based mental health surveys: 1,161 Vietnamese-Australian residents in the state of New South Wales; 3,039 Vietnamese resident in the Mekong Delta region of Vietnam; and 7,961 Australian-born persons drawn from a national survey. All surveys applied the Composite International Diagnostic Interview, with the Vietnamese surveys also applying the Phan Vietnamese Psychiatric Rating Scale, an indigenously-derived measure of mental disorder. The ICD-10 classification system yielded lowest rates amongst Vietnamese in the Mekong Delta, intermediate amongst Vietnamese in NSW; and highest rates amongst the Australian-born population. The Phan Vietnamese Psychiatric Rating Scale added a substantial number of cases in both Vietnamese samples. -

Full Text in Pdf Format
Vol. 13: 211–224, 2021 AQUACULTURE ENVIRONMENT INTERACTIONS Published May 27 https://doi.org/10.3354/aei00395 Aquacult Environ Interact OPEN ACCESS Intestinal microbial diversity and functional analysis of Urechis unicinctus from two different habitats: pond polycultured with Penaeus japonicus and coastal zone Yongzheng Tang1, Shuai Ma1, Yihao Liu2, Yongrui Pi1,*,Ying Liu1, Ye Zhao1 1School of Ocean, Yantai University, Yantai 264005, PR China 2Shandong Key Laboratory of Marine Ecological Restoration, Shandong Marine Source and Environment Research Institute, Yantai 264006, PR China ABSTRACT: Urechis unicinctus is an important commercial and ecological invertebrate that has potential applications in the study of marine invertebrate evolution and marine pharmaceutical development. Here we analyzed the intestinal microbial diversity of U. unicinctus from 2 different habitats using 16S rDNA 454 high-throughput sequencing. The dominant phyla were Proteo - bacteria, Bacterioidetes, Firmicutes, and Actinobacteria in gut samples of U. unicinctus, which significantly differed from those in its 2 habitats (i.e. intertidal mudflat and pond polyculture). Exceptions were Proteobacteria, Firmicutes and Bacterioidetes, which were the dominant phyla in the sediment and water samples. The top 15 genera in the gut samples did not show any signifi- cant differences between the 2 habitats. Functional analysis of the intestinal microbial community showed that metabolism, including carbohydrate and amino acid metabolism, was the most important function. Methane metabolism was one of the main components of energy metabolism. The gut microbes also played an important role in environmental and genetic information pro- cessing, cellular processes, etc. These findings provide an understanding of gut microbiome com- position and diversity in U. -

Urechis Unicinctus
Hou et al. BMC Genomics (2020) 21:892 https://doi.org/10.1186/s12864-020-07312-4 RESEARCH ARTICLE Open Access Identification of the neuropeptide precursor genes potentially involved in the larval settlement in the Echiuran worm Urechis unicinctus Xitan Hou1, Zhenkui Qin1, Maokai Wei1, Zhong Fu3, Ruonan Liu4,LiLu1, Shumiao Bai1, Yubin Ma1* and Zhifeng Zhang1,2* Abstract Background: In marine invertebrate life cycles, which often consist of planktonic larval and benthonic adult stages, settlement of the free-swimming larva to the sea floor in response to environmental cues is a key life cycle transition. Settlement is regulated by a specialized sensory–neurosecretory system, the larval apical organ. The neuroendocrine mechanisms through which the apical organ transduces environmental cues into behavioral responses during settlement are not fully understood yet. Results: In this study, a total of 54 neuropeptide precursors (pNPs) were identified in the Urechis unicinctus larva and adult transcriptome databases using local BLAST and NpSearch prediction, of which 10 pNPs belonging to the ancient eumetazoa, 24 pNPs belonging to the ancient bilaterian, 3 pNPs belonging to the ancient protostome, 9 pNPs exclusive in lophotrochozoa, 3 pNPs exclusive in annelid, and 5 pNPs only found in U. unicinctus.Furthermore,four pNPs (MIP, FRWamide, FxFamide and FILamide) which may be associated with the settlement and metamorphosis of U. unicinctus larvae were analysed by qRT-PCR. Whole-mount in situ hybridization results showed that all the four pNPs were expressed in the region of the apical organ of the larva, and the positive signals were also detected in the ciliary band and abdomen chaetae. -

Urechis Unicinctus by Digital Gene Expression Analysis Xiaolong Liu†, Litao Zhang†, Zhifeng Zhang*, Xiaoyu Ma and Jianguo Liu
Liu et al. BMC Genomics (2015) 16:829 DOI 10.1186/s12864-015-2094-z RESEARCH ARTICLE Open Access Transcriptional response to sulfide in the Echiuran Worm Urechis unicinctus by digital gene expression analysis Xiaolong Liu†, Litao Zhang†, Zhifeng Zhang*, Xiaoyu Ma and Jianguo Liu Abstract Background: Urechis unicinctus, an echiuran worm inhabiting the U-shaped burrows in the coastal mud flats, is an important commercial and ecological invertebrate in Northeast Asian countries, which has potential applications in the study of animal evolution, coastal sediment improvement and marine drug development. Furthermore, the worm can tolerate and utilize well-known toxicant-sulfide. However, knowledge is limited on the molecular mechanism of U. unicinctus responding to sulfide due to deficiency of its genetic information. Methods: In this study, we performed Illumina sequencing to obtain the first Urechis unicinctus transcriptome data. Sequenced reads were assembled and then annotated using blast searches against Nr, Nt, Swiss-Prot, KEGG and COG. The clean tags from four digital gene expression (DGE) libraries were mapped to the U. unicinctus transcriptome. DGE analysis and functional annotation were then performed to reveal its response to sulfide. The expressions of 12 candidate genes were validated using quantitative real-time PCR. The results of qRT-PCR were regressed against the DGE analysis, with a correlation coefficient and p-value reported for each of them. Results: Here we first present a draft of U. unicinctus transcriptome using the Illumina HiSeqTM 2000 platform and 52,093 unique sequences were assembled with the average length of 738 bp and N50 of 1131 bp. About 51.6 % of the transcriptome were functionally annotated based on the databases of Nr, Nt, Swiss-Prot, KEGG and COG.