Calcium Atpase and Respiratory Muscle Function
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Aetiology of Skeletal Muscle 'Cramps' During Exercise: a Novel Hypothesis
Journal of Sports Sciences ISSN: 0264-0414 (Print) 1466-447X (Online) Journal homepage: http://www.tandfonline.com/loi/rjsp20 Aetiology of skeletal muscle ‘cramps’ during exercise: A novel hypothesis M. P. Schwellnus , E. W. Derman & T. D. Noakes To cite this article: M. P. Schwellnus , E. W. Derman & T. D. Noakes (1997) Aetiology of skeletal muscle ‘cramps’ during exercise: A novel hypothesis, Journal of Sports Sciences, 15:3, 277-285, DOI: 10.1080/026404197367281 To link to this article: http://dx.doi.org/10.1080/026404197367281 Published online: 01 Dec 2010. Submit your article to this journal Article views: 942 View related articles Citing articles: 68 View citing articles Full Terms & Conditions of access and use can be found at http://www.tandfonline.com/action/journalInformation?journalCode=rjsp20 Download by: [Australian Catholic University] Date: 24 September 2017, At: 18:51 Journal of Sports Sciences, 1997, 15, 277-285 Aetiology of skeletal muscle `cramps’ during exercise: A novel hypothesis M .P. SCH WELLN US,* E.W. D ERM AN and T.D. N OAKES M RC/UCT B ioenergetics of Exercise Research Unit, University of Cape Town M edical School, Sports Science Institute of South Africa, PO B ox 115, Newlands 7725, South Africa Accepted 3 September 1996 The aetiology of exercise-associated muscle cramps (EAMC), de® ned as `painful, spasmodic, involuntary contractions of skeletal muscle during or immediately after physical exercise’ , has not been well investigated and is therefore not well understood. This review focuses on the physiological basis for skeletal muscle relaxation, a historical perspective and analysis of the commonly postulated causes of EAMC, and known facts about EAMC from recent clinical studies. -

Plasma Membrane Ca2+–Atpase in Rat and Human Odontoblasts Mediates Dentin Mineralization
biomolecules Article Plasma Membrane Ca2+–ATPase in Rat and Human Odontoblasts Mediates Dentin Mineralization Maki Kimura 1,†, Hiroyuki Mochizuki 1,†, Ryouichi Satou 2, Miyu Iwasaki 2, Eitoyo Kokubu 3, Kyosuke Kono 1, Sachie Nomura 1, Takeshi Sakurai 1, Hidetaka Kuroda 1,4,† and Yoshiyuki Shibukawa 1,*,† 1 Department of Physiology, Tokyo Dental College, 2-9-18, Kanda-Misaki-cho, Chiyoda-ku, Tokyo 101-0061, Japan; [email protected] (M.K.); [email protected] (H.M.); [email protected] (K.K.); [email protected] (S.N.); [email protected] (T.S.); [email protected] (H.K.) 2 Department of Epidemiology and Public Health, Tokyo Dental College, Chiyodaku, Tokyo 101-0061, Japan; [email protected] (R.S.); [email protected] (M.I.) 3 Department of Microbiology, Tokyo Dental College, Chiyodaku, Tokyo 101-0061, Japan; [email protected] 4 Department of Dental Anesthesiology, Kanagawa Dental University, 1-23, Ogawacho, Kanagawa, Yokosuka-shi 238-8570, Japan * Correspondence: [email protected] † These authors contributed equally to this study. Abstract: Intracellular Ca2+ signaling engendered by Ca2+ influx and mobilization in odontoblasts is critical for dentinogenesis induced by multiple stimuli at the dentin surface. Increased Ca2+ is exported by the Na+–Ca2+ exchanger (NCX) and plasma membrane Ca2+–ATPase (PMCA) to Citation: Kimura, M.; Mochizuki, H.; maintain Ca2+ homeostasis. We previously demonstrated a functional coupling between Ca2+ Satou, R.; Iwasaki, M.; Kokubu, E.; extrusion by NCX and its influx through transient receptor potential channels in odontoblasts. Kono, K.; Nomura, S.; Sakurai, T.; Although the presence of PMCA in odontoblasts has been previously described, steady-state levels of Kuroda, H.; Shibukawa, Y. -

Molecular Regulation and Physiology of the H ,K -Atpases in Kidney
Molecular Regulation and Physiology of the H؉,K؉-ATPases in Kidney Juan Codina and Thomas D. DuBose Jr Two H؉,K؉-adenosine triphosphatase (ATPase) proteins participate in K؉ absorption and ؉ ؉ ؉ H secretion in the renal medulla. Both the gastric (HK␣1) and colonic (HK␣2)H,K - ATPases have been localized and characterized by a number of techniques, and are known to be highly regulated in response to acid-base and electrolyte disturbances. Both ATPases are dimers of composition ␣/ that localize to the apical membrane and both interact with the tetraspanin protein CD63. Although CD63 interacts with the carboxy-terminus of the subunit of the colonic H؉,K؉-ATPase, it interacts with the -subunit of the gastric-␣ H؉,K؉-ATPase. Pharmacologically, both ATPases are distinct; for example, the gastric H؉,K؉-ATPase is inhibited by Sch-28080, but the colonic H؉,K؉-ATPase is inhibited by .ouabain (a classic inhibitor of the Na؉-pump) and is completely insensitive to Sch-28080 The ␣-subunit of the colonic H؉,K؉-ATPase is the only subunit of the X؉,K؉-ATPase superfamily that has 3 different splice variants that emerge by deletion or elongation of the amino-terminus. The messenger RNA and protein of one of these splice variants (HK␣2C)is specifically up-regulated in newborn rats and becomes undetectable in adult rats. There- fore, HK␣2, in addition to its role in potassium and acid-base homeostasis, appears to play a significant role in early growth and development. Finally, because chronic hypokalemia appears to be the most potent stimulus for upregulation of HK␣2, we propose that the HK␣2 participates importantly in the maintenance of chronic metabolic alkalosis. -

Na+/K+-Atpase) and Its Isoforms Kaur R1,2, Sodhi M2, Sharma VL1, Sharma A2, Mann S2 and Mukesh M 2
TRJ VOL. 2 ISSUE 2 MAR-APR 2016 ISSN: 2454-7301 (PRINT) | ISSN: 2454-4930 (ONLINE) A Review: Sodium-Potassium Adenosine Triphosphatase (Na+/K+-ATPase) and its Isoforms Kaur R1,2, Sodhi M2, Sharma VL1, Sharma A2, Mann S2 and Mukesh M 2. 1Department of Zoology, Panjab University, Chandigarh 2National Bureau of Animal Genetic Resources, Karnal *Email: [email protected] these enzymes use the hydrolysis of ATP to drive the transport Abstract - Na+/K+-ATPase, found in all mammalian cell of cations against an electrochemical potential. membranes, is necessary for proper cellular function since it The characteristic feature of P type ATPase is the helps to preserve the ionic gradients across the cell membrane formation of the phosphorylated intermediate state during the and thus the membrane potential and osmotic equilibrium of reaction cycle [5]. Hence this features differentiates it from the cell. The sodium-potassium ATPase (Na+/K+-ATPase) is other types of ATPases i.e. F-ATPases (present in an important ion transporter which pumps sodium out of cells mitochondria, chloroplasts and bacterial plasma membranes) in exchange for potassium, thereby generating large gradients and V-ATPases (present in eukaryotic vacuoles). The presence of these ions across the plasma membrane. The isoforms α (αl, of conserved sequence motifs in the cytoplasmic domains α2, α3 and α4) and β (β1, β2 and β3) combine to form a defines the primary structure of P-type ATPases. Nucleotide- number of Na+/K+-ATPase isozymes. So in present study binding (N), phosphorylation (P) and an actuator (A) domain paper information regarding molecular regulation of the are the three domains present in the cytoplasmic domain [6]. -

The SERCA Residue Glu340 Mediates Interdomain Communication That Guides Ca2+ Transport
The SERCA residue Glu340 mediates interdomain communication that guides Ca2+ transport Maxwell M. G. Geurtsa,1, Johannes D. Clausenb,c,1, Bertrand Arnoub,d,1, Cédric Montignyd, Guillaume Lenoird, Robin A. Coreya, Christine Jaxeld, Jesper V. Møllerb, Poul Nissenc,e, Jens Peter Andersenb, Marc le Maired, and Maike Bublitza,2 aDepartment of Biochemistry, University of Oxford, OX1 3QU Oxford, United Kingdom; bDepartment of Biomedicine, Aarhus University, 8000 Aarhus C, Denmark; cDepartment of Molecular Biology and Genetics, Aarhus University, 8000 Aarhus C, Denmark; dInstitute for Integrative Biology of the Cell (I2BC), Commissariat à l’Energie Atomique et aux Energies Alternatives, CNRS, Université Paris-Saclay, 91198 Gif-sur-Yvette, France; and eDanish Research Institute of Translational Neuroscience-DANDRITE, Nordic European Molecular Biology Laboratory Partnership for Molecular Medicine, Aarhus University, 8000 Aarhus C, Denmark Edited by Ivet Bahar, University of Pittsburgh School of Medicine, Pittsburgh, PA, and approved October 21, 2020 (received for review July 15, 2020) The sarco(endo)plasmic reticulum Ca2+-ATPase (SERCA) is a P-type A number of SERCA1a crystal structures have shed light on ATPase that transports Ca2+ from the cytosol into the sarco(endo) the nature of the conformational changes associated with Ca2+ plasmic reticulum (SR/ER) lumen, driven by ATP. This primary transport (reviewed in refs. 3–5). There are two Ca2+-binding transport activity depends on tight coupling between movements sites within the TM domain of SERCA, denoted sites I and II of the transmembrane helices forming the two Ca2+-binding sites based on a proven sequential order of Ca2+ binding (6). The and the cytosolic headpiece mediating ATP hydrolysis. -

Targeting Oncogenic Notch Signaling with SERCA Inhibitors Luca Pagliaro, Matteo Marchesini and Giovanni Roti*
Pagliaro et al. J Hematol Oncol (2021) 14:8 https://doi.org/10.1186/s13045-020-01015-9 REVIEW Open Access Targeting oncogenic Notch signaling with SERCA inhibitors Luca Pagliaro, Matteo Marchesini and Giovanni Roti* Abstract P-type ATPase inhibitors are among the most successful and widely prescribed therapeutics in modern pharmacol- ogy. Clinical transition has been safely achieved for H+/K+ ATPase inhibitors such as omeprazole and Na+/K+-ATPase 2 inhibitors like digoxin. However, this is more challenging for Ca +-ATPase modulators due to the physiological role of 2 2 Ca + in cardiac dynamics. Over the past two decades, sarco-endoplasmic reticulum Ca +-ATPase (SERCA) modula- 2 tors have been studied as potential chemotherapy agents because of their Ca +-mediated pan-cancer lethal efects. Instead, recent evidence suggests that SERCA inhibition suppresses oncogenic Notch1 signaling emerging as an alternative to γ-secretase modulators that showed limited clinical activity due to severe side efects. In this review, we focus on how SERCA inhibitors alter Notch1 signaling and show that Notch on-target-mediated antileukemia proper- 2 ties of these molecules can be achieved without causing overt Ca + cellular overload. Keywords: SERCA , T cell acute lymphoblastic leukemia, Thapsigargin, Notch signaling, NOTCH1, CAD204520, T-ALL Background metalloprotease (ADAM-10 or TACE/ADAM-17). Te NOTCH receptors are transmembrane cell-surface pro- resulting short-lived protein fragments are substrates teins that control cell to cell communication, embryo- -

SERCA in Genesis of Arrhythmias: What We Already Know and What Is New?
Review 43 SERCA in genesis of arrhythmias: what we already know and what is new? Nilüfer Erkasap Department of Physiology, Medical Faculty, Eskiflehir Osmangazi University, Eskiflehir, Turkey ABSTRACT This review mainly focuses on the structure, function of the sarco(endo)plasmic reticulum calcium pump (SERCA) and its role in genesis of arrhythmias. SERCA is a membrane protein that belongs to the family of P-type ion translocating ATPases and pumps free cytosolic calcium into intracellular stores. Active transport of Ca2+ is achieved, according to the E1-E2 model, changing of SERCA structure by Ca2+. The affinity of Ca2+ -binding sites varies from high (E1) to low (E2). Three different SERCA genes were identified-SERCA1, SERCA2, and SERCA3. SERCA is mainly represented by the SERCA2a isoform in the heart. In heart muscle, during systole, depolarization triggers the release of Ca2+ from the sarcoplasmic reticulum (SR) and starts contraction. During diastole, muscle relaxation occurs as Ca2+ is again removed from cytosol, predominantly by accumulation into SR via the action of SERCA2a. The main regulator of SERCA2a is phospholamban and another regulator proteolipid of SERCA is sarcolipin. There are a lot of studies on the effect of decreased and/or increased SERCA activity in genesis of arrhythmia. Actually both decrease and increase of SERCA activity in the heart result in some pathological mechanisms such as heart failure and arrhythmia. (Anadolu Kardiyol Derg 2007: 7 Suppl 1; 43-6) Key words: sarco(endo)plasmic reticulum, SERCA, arrhythmia, calcium channels Introduction from cytosol, predominantly by accumulation into sarcoplasmic reticulum via the action of sarco(endo)plasmic reticulum Cardiac physiology is a major area of research in basic and Ca ATPase (SERCA). -

Endoplasmic Reticulum Potassium–Hydrogen Exchanger and Small
Research Article 625 Endoplasmic reticulum potassium–hydrogen exchanger and small conductance calcium-activated potassium channel activities are essential for ER calcium uptake in neurons and cardiomyocytes Malle Kuum1,2,3, Vladimir Veksler2,3, Joanna Liiv1, Renee Ventura-Clapier2,3 and Allen Kaasik1,* 1Department of Pharmacology, Centre of Excellence for Translational Medicine, University of Tartu, Ravila 19, Tartu EE-51014, Estonia 2INSERM, U-769, 5, rue Jean-Baptiste Clement, Chaˆtenay-Malabry F-92296, France 3Universite´ Paris-Sud, 5, rue Jean-Baptiste Clement, Chaˆtenay-Malabry F-92296, France *Author for correspondence ([email protected]) Accepted 12 September 2011 Journal of Cell Science 125, 625–633 ß 2012. Published by The Company of Biologists Ltd doi: 10.1242/jcs.090126 Summary Calcium pumping into the endoplasmic reticulum (ER) lumen is thought to be coupled to a countertransport of protons through sarcoplasmic/endoplasmic reticulum calcium ATPase (SERCA) and the members of the ClC family of chloride channels. However, pH in the ER lumen remains neutral, which suggests a mechanism responsible for proton re-entry. We studied whether cation–proton exchangers could act as routes for such a re-entry. ER Ca2+ uptake was measured in permeabilized immortalized hypothalamic neurons, primary rat cortical neurons and mouse cardiac fibers. Replacement of K+ in the uptake solution with Na+ or tetraethylammonium led to a strong inhibition of Ca2+ uptake in neurons and cardiomyocytes. Furthermore, inhibitors of the potassium–proton exchanger (quinine or propranolol) but not of the sodium–proton exchanger reduced ER Ca2+ uptake by 56–82%. Externally added nigericin, a potassium– + proton exchanger, attenuated the inhibitory effect of propranolol. -

1/05661 1 Al
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date _ . ... - 12 May 2011 (12.05.2011) W 2 11/05661 1 Al (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every C12Q 1/00 (2006.0 1) C12Q 1/48 (2006.0 1) kind of national protection available): AE, AG, AL, AM, C12Q 1/42 (2006.01) AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, (21) Number: International Application DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, PCT/US20 10/054171 HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, (22) International Filing Date: KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, 26 October 2010 (26.10.2010) ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, PE, PG, PH, PL, PT, RO, RS, RU, SC, SD, (25) Filing Language: English SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, (26) Publication Language: English TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: (84) Designated States (unless otherwise indicated, for every 61/255,068 26 October 2009 (26.10.2009) US kind of regional protection available): ARIPO (BW, GH, GM, KE, LR, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, (71) Applicant (for all designated States except US): ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, MYREXIS, INC. -

Fhit Proteins Can Also Recognize Substrates Other Than Dinucleoside Polyphosphates
FEBS Letters 582 (2008) 3152–3158 Fhit proteins can also recognize substrates other than dinucleoside polyphosphates Andrzej Guranowskia,*, Anna M. Wojdyłaa, Małgorzata Pietrowska-Borekb, Paweł Bieganowskic, Elena N. Khursd, Matthew J. Cliffe, G. Michael Blackburnf, Damian Błaziakg, Wojciech J. Stecg a Department of Biochemistry and Biotechnology, The University of Life Sciences, 35 Wołyn´ska Street, 60-637 Poznan´, Poland b Department of Plant Physiology, The University of Life Sciences, 60-637 Poznan´, Poland c International Institute of Molecular and Cell Biology, Warsaw, Poland d Engelhardt Institute of Molecular Biology, Russian Academy of Sciences, Moscow, Russia e Department of Molecular Biology and Biotechnology, Krebs Institute, University of Sheffield, Sheffield, UK f Department of Chemistry, Krebs Institute, University of Sheffield, Sheffield, UK g Center of Molecular and Macromolecular Studies, Polish Academy of Sciences, Ło´dz´, Poland Received 1 July 2008; revised 17 July 2008; accepted 31 July 2008 Available online 9 August 2008 Edited by Hans Eklund This work is dedicated to Professor Antonio Sillero on the occasion of his 70th birthday and to Professor Maria Antonia Gu¨nther Sillero. 1. Introduction Abstract We show here that Fhit proteins, in addition to their function as dinucleoside triphosphate hydrolases, act similarly to adenylylsulfatases and nucleoside phosphoramidases, liberat- Cells contain various minor nucleotides. Among these are 0 000 1 n 0 ing nucleoside 50-monophosphates from such natural metabolites the dinucleoside 5 ,5 -P ,P -polyphosphates (NpnN s, where as adenosine 50-phosphosulfate and adenosine 50-phosphorami- N and N0 are 50-O-nucleosides and n represents the number date. Moreover, Fhits recognize synthetic nucleotides, such as of phosphate residues in the polyphosphate chain that esterifies 0 0 0 0 0 adenosine 5 -O-phosphorofluoridate and adenosine 5 -O-(c-flu- N and N at their 5 position) [1].NpnN s accumulate as a re- orotriphosphate), and release AMP from them. -

Regulation of Membrane Calcium Transport Proteins by the Surrounding Lipid Environment
Review Regulation of Membrane Calcium Transport Proteins by the Surrounding Lipid Environment Louise Conrard and Donatienne Tyteca * CELL Unit, de Duve Institute and Université catholique de Louvain, UCL B1.75.05, avenue Hippocrate, 75, B‐1200 Brussels, Belgium * Correspondence: [email protected]; Tel.: +32‐2‐764.75.91; Fax: +32‐2‐764.75.43 Received: 8 August 2019; Accepted: 10 September 2019; Published: 20 September 2019 Abstract: Calcium ions (Ca2+) are major messengers in cell signaling, impacting nearly every aspect of cellular life. Those signals are generated within a wide spatial and temporal range through a large variety of Ca2+ channels, pumps, and exchangers. More and more evidences suggest that Ca2+ exchanges are regulated by their surrounding lipid environment. In this review, we point out the technical challenges that are currently being overcome and those that still need to be defeated to analyze the Ca2+ transport protein–lipid interactions. We then provide evidences for the modulation of Ca2+ transport proteins by lipids, including cholesterol, acidic phospholipids, sphingolipids, and their metabolites. We also integrate documented mechanisms involved in the regulation of Ca2+ transport proteins by the lipid environment. Those include: (i) Direct interaction inside the protein with non‐annular lipids; (ii) close interaction with the first shell of annular lipids; (iii) regulation of membrane biophysical properties (e.g., membrane lipid packing, thickness, and curvature) directly around the protein through annular lipids; and (iv) gathering and downstream signaling of several proteins inside lipid domains. We finally discuss recent reports supporting the related alteration of Ca2+ and lipids in different pathophysiological events and the possibility to target lipids in Ca2+‐ related diseases. -
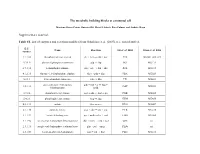
The Metabolic Building Blocks of a Minimal Cell Supplementary
The metabolic building blocks of a minimal cell Mariana Reyes-Prieto, Rosario Gil, Mercè Llabrés, Pere Palmer and Andrés Moya Supplementary material. Table S1. List of enzymes and reactions modified from Gabaldon et. al. (2007). n.i.: non identified. E.C. Name Reaction Gil et. al. 2004 Glass et. al. 2006 number 2.7.1.69 phosphotransferase system glc + pep → g6p + pyr PTS MG041, 069, 429 5.3.1.9 glucose-6-phosphate isomerase g6p ↔ f6p PGI MG111 2.7.1.11 6-phosphofructokinase f6p + atp → fbp + adp PFK MG215 4.1.2.13 fructose-1,6-bisphosphate aldolase fbp ↔ gdp + dhp FBA MG023 5.3.1.1 triose-phosphate isomerase gdp ↔ dhp TPI MG431 glyceraldehyde-3-phosphate gdp + nad + p ↔ bpg + 1.2.1.12 GAP MG301 dehydrogenase nadh 2.7.2.3 phosphoglycerate kinase bpg + adp ↔ 3pg + atp PGK MG300 5.4.2.1 phosphoglycerate mutase 3pg ↔ 2pg GPM MG430 4.2.1.11 enolase 2pg ↔ pep ENO MG407 2.7.1.40 pyruvate kinase pep + adp → pyr + atp PYK MG216 1.1.1.27 lactate dehydrogenase pyr + nadh ↔ lac + nad LDH MG460 1.1.1.94 sn-glycerol-3-phosphate dehydrogenase dhp + nadh → g3p + nad GPS n.i. 2.3.1.15 sn-glycerol-3-phosphate acyltransferase g3p + pal → mag PLSb n.i. 2.3.1.51 1-acyl-sn-glycerol-3-phosphate mag + pal → dag PLSc MG212 acyltransferase 2.7.7.41 phosphatidate cytidyltransferase dag + ctp → cdp-dag + pp CDS MG437 cdp-dag + ser → pser + 2.7.8.8 phosphatidylserine synthase PSS n.i. cmp 4.1.1.65 phosphatidylserine decarboxylase pser → peta PSD n.i.