Rutherford's Nuclear World: the Story of the Discovery of the Nuc
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Cathode Rays 89 Cathode Rays
Cathode Rays 89 Cathode Rays Theodore Arabatzis C The detection of cathode rays was a by-product of the investigation of the discharge of electricity through rarefied gases. The latter phenomenon had been studied since the early eighteenth century. By the middle of the nineteenth century it was known that the passage of electricity through a partly evacuated tube produced a glow in the gas, whose color depended on its chemical composition and its pressure. Below a certain pressure the glow assumed a stratified pattern of bright and dark bands. During the second half of the nineteenth century the discharge of electricity through gases became a topic of intense exploratory experimentation, primarily in Germany [21]. In 1855 the German instrument maker Heinrich Geißler (1815– 1879) manufactured improved vacuum tubes, which made possible the isolation and investigation of cathode rays [23]. In 1857 Geissler’s tubes were employed by Julius Pl¨ucker (1801–1868) to study the influence of a magnet on the electrical dis- charge. He observed various complex and striking phenomena associated with the discharge. Among those phenomena were a “light which appears about the negative electrode” and a fluorescence in the glass of the tube ([9], pp. 122, 130). The understanding of those phenomena was advanced by Pl¨ucker’s student and collaborator, Johann Wilhelm Hittorf (1824–1914), who observed that “if any ob- ject is interposed in the space filled with glow-light [emanating from the negative electrode], it throws a sharp shadow on the fluorescent side” ([5], p. 117). This effect implied that the “rays” emanating from the cathode followed a straight path. -

The Development of Military Nuclear Strategy And
The Development of Military Nuclear Strategy and Anglo-American Relations, 1939 – 1958 Submitted by: Geoffrey Charles Mallett Skinner to the University of Exeter as a thesis for the degree of Doctor of Philosophy in History, July 2018 This thesis is available for Library use on the understanding that it is copyright material and that no quotation from the thesis may be published without proper acknowledgement. I certify that all material in this thesis which is not my own work has been identified and that no material has previously been submitted and approved for the award of a degree by this or any other University. (Signature) ……………………………………………………………………………… 1 Abstract There was no special governmental partnership between Britain and America during the Second World War in atomic affairs. A recalibration is required that updates and amends the existing historiography in this respect. The wartime atomic relations of those countries were cooperative at the level of science and resources, but rarely that of the state. As soon as it became apparent that fission weaponry would be the main basis of future military power, America decided to gain exclusive control over the weapon. Britain could not replicate American resources and no assistance was offered to it by its conventional ally. America then created its own, closed, nuclear system and well before the 1946 Atomic Energy Act, the event which is typically seen by historians as the explanation of the fracturing of wartime atomic relations. Immediately after 1945 there was insufficient systemic force to create change in the consistent American policy of atomic monopoly. As fusion bombs introduced a new magnitude of risk, and as the nuclear world expanded and deepened, the systemic pressures grew. -

Proceedings of the Sir Mark Oliphant International Frontiers of Science and Technology Australian Geothermal Energy Conference Record 2008/18 Gurgenci, H
GEOSCIENCE AUSTRALIA Sir Mark Oliphant Conferences – International Frontiers of Science and Technology Proceedings of the Sir Mark Oliphant International Frontiers of Science and Technology Australian Geothermal Energy Conference Record 2008/18 Gurgenci, H. and Budd, A.R. APPLYING GEOSCIENCE TO AUSTRALIA’S MOST IMPORTANT CHALLENGES Proceedings of the Sir Mark Oliphant International Frontiers of Science and Technology Australian Geothermal Energy Conference GEOSCIENCE AUSTRALIA RECORD 2008/18 Edited by Hal Gurgenci 1 and Anthony Budd 2 1 Queensland Geothermal Energy Centre, The University of Queensland, St Lucia 4072 2 Geoscience Australia, GPO Box 378, Canberra, ACT 2601 Department of Resources, Energy and Tourism Minister for Resources and Energy: The Hon. Martin Ferguson, AM MP Secretary: Dr Peter Boxall Geoscience Australia Chief Executive Officer: Dr Neil Williams PSM © Commonwealth of Australia, 2008 This work is copyright. Apart from any fair dealings for the purpose of study, research, criticism, or review, as permitted under the Copyright Act 1968, no part may be reproduced by any process without written permission. Copyright is the responsibility of the Chief Executive Officer, Geoscience Australia. Requests and enquiries should be directed to the Chief Executive Officer, Geoscience Australia, GPO Box 378 Canberra ACT 2601. Geoscience Australia has tried to make the information in this product as accurate as possible. However, it does not guarantee that the information is totally accurate or complete. Therefore, you should not solely rely on this information when making a commercial decision. ISSN 1448-2177 ISBN 978 1 921498 19 0 GeoCat # 67255 Recommended bibliographic reference: Gurgenci, H. and Budd, A.R. (editors), 2008. -

A Selected Bibliography of Publications By, and About, J
A Selected Bibliography of Publications by, and about, J. Robert Oppenheimer Nelson H. F. Beebe University of Utah Department of Mathematics, 110 LCB 155 S 1400 E RM 233 Salt Lake City, UT 84112-0090 USA Tel: +1 801 581 5254 FAX: +1 801 581 4148 E-mail: [email protected], [email protected], [email protected] (Internet) WWW URL: http://www.math.utah.edu/~beebe/ 17 March 2021 Version 1.47 Title word cross-reference $1 [Duf46]. $12.95 [Edg91]. $13.50 [Tho03]. $14.00 [Hug07]. $15.95 [Hen81]. $16.00 [RS06]. $16.95 [RS06]. $17.50 [Hen81]. $2.50 [Opp28g]. $20.00 [Hen81, Jor80]. $24.95 [Fra01]. $25.00 [Ger06]. $26.95 [Wol05]. $27.95 [Ger06]. $29.95 [Goo09]. $30.00 [Kev03, Kle07]. $32.50 [Edg91]. $35 [Wol05]. $35.00 [Bed06]. $37.50 [Hug09, Pol07, Dys13]. $39.50 [Edg91]. $39.95 [Bad95]. $8.95 [Edg91]. α [Opp27a, Rut27]. γ [LO34]. -particles [Opp27a]. -rays [Rut27]. -Teilchen [Opp27a]. 0-226-79845-3 [Guy07, Hug09]. 0-8014-8661-0 [Tho03]. 0-8047-1713-3 [Edg91]. 0-8047-1714-1 [Edg91]. 0-8047-1721-4 [Edg91]. 0-8047-1722-2 [Edg91]. 0-9672617-3-2 [Bro06, Hug07]. 1 [Opp57f]. 109 [Con05, Mur05, Nas07, Sap05a, Wol05, Kru07]. 112 [FW07]. 1 2 14.99/$25.00 [Ber04a]. 16 [GHK+96]. 1890-1960 [McG02]. 1911 [Meh75]. 1945 [GHK+96, Gow81, Haw61, Bad95, Gol95a, Hew66, She82, HBP94]. 1945-47 [Hew66]. 1950 [Ano50]. 1954 [Ano01b, GM54, SZC54]. 1960s [Sch08a]. 1963 [Kuh63]. 1967 [Bet67a, Bet97, Pun67, RB67]. 1976 [Sag79a, Sag79b]. 1981 [Ano81]. 20 [Goe88]. 2005 [Dre07]. 20th [Opp65a, Anoxx, Kai02]. -

Patrick Blackett: Sailor, Scientist, Socialist, Chris Eldridge
Naval War College Review Volume 57 Article 31 Number 1 Winter 2004 Patrick Blackett: Sailor, Scientist, Socialist, Chris Eldridge Follow this and additional works at: https://digital-commons.usnwc.edu/nwc-review Recommended Citation Eldridge, Chris (2004) "Patrick Blackett: Sailor, Scientist, Socialist,," Naval War College Review: Vol. 57 : No. 1 , Article 31. Available at: https://digital-commons.usnwc.edu/nwc-review/vol57/iss1/31 This Book Review is brought to you for free and open access by the Journals at U.S. Naval War College Digital Commons. It has been accepted for inclusion in Naval War College Review by an authorized editor of U.S. Naval War College Digital Commons. For more information, please contact [email protected]. 156 NAVAL WAR COLLEGE REVIEW Eldridge: Patrick Blackett: Sailor, Scientist, Socialist, understanding with the Soviet Union (a he was the heart and soul of the Cold position favored by some influential War military-academic-industrial com- opinions in the United States) nor to cre- plex. In this book, sixteen authors at- ate trouble for the French government, tempt to shed light on Blackett’s role in seemingly both dependent on and threat- that story. The collection includes pa- ened by the French Communist Party. pers presented at a 1998 conference The Soviet-menace card was played on commemorating Blackett at Cambridge several occasions in the unfolding de- University, as well as other recent writ- bate, but in general it was subordinated ings about him. to more abstract arguments of enlight- Not surprisingly, the compendium of- ened self-interest. Moreover, it was fers a range of perspectives on events clear to many in the administration that and issues with which Blackett was as- too great an emphasis on the immi- sociated, rather than a comprehensive nence of war with Russia would scuttle examination of his life and work. -

Front Matter (PDF)
PROCEEDINGS OF THE OYAL SOCIETY OF LONDON Series A CONTAINING PAPERS OF A MATHEMATICAL AND PHYSICAL CHARACTER. VOL. LXXXV. LONDON: P rinted for THE ROYAL SOCIETY and S old by HARRISON AND SONS, ST. MARTIN’S LANE, PRINTERS IN ORDINARY TO HIS MAJESTY. N ovember, 1911. LONDON: HARRISON AND SONS, PRINTERS IN ORDINARY TO HTS MAJESTY, ST. MARTIN’S LANE. CONTENTS --- 0 0 ^ 0 0 ----- SERIES A. VOL. LXXXV. No. A 575.—March 14, 1911. P AG-E The Chemical Physics involved in the Precipitation of Free Carbon from the Alloys of the Iron-Carbon System. By W. H. Hatfield, B. Met. (Sheffield University). Communicated by Prof. W. M. Hicks, F.R.S. (Plates 1—5) ................... .......... 1 On the Fourier Constants of a Function. By W. H. Young, Se.D., F.R.S................. 14 The Charges on Ions in Gases, and some Effects that Influence the Motion of Negative Ions. By Prof. John S. Townsend, F.R.S............................................... 25 On the Energy and Distribution of Scattered Rontgen Radiation. By J. A. Crowtlier, M.A., Fellow of St. John's College, Cambridge. Com municated by Prof. Sir J. J. Thomson, F.R.S...........................*.............................. 29 The Origin of Magnetic Storms. By Arthur Schuster, F.R.S...................................... 44 On the Periodicity of Sun-spots. By Arthur Schuster, F.R.S..................................... 50 The Absorption Spectx*a of Lithium and Caesium. By Prof. P. V. Bevan, M.A., Royal Holloway College. Communicated by Sir J. J. Thomson, F.R.S.............. 54 Dispersion in Vapours of the Alkali Metals. By Prof. P. V. Bevan, M.A., Royal Holloway College. -

1 Ethers, Religion and Politics In
ORE Open Research Exeter TITLE Ethers, religion and politics in late-Victorian physics: beyond the Wynne thesis AUTHORS Noakes, Richard JOURNAL History of Science DEPOSITED IN ORE 16 June 2008 This version available at http://hdl.handle.net/10036/30065 COPYRIGHT AND REUSE Open Research Exeter makes this work available in accordance with publisher policies. A NOTE ON VERSIONS The version presented here may differ from the published version. If citing, you are advised to consult the published version for pagination, volume/issue and date of publication ETHERS, RELIGION AND POLITICS IN LATE-VICTORIAN PHYSICS: BEYOND THE WYNNE THESIS RICHARD NOAKES 1. INTRODUCTION In the past thirty years historians have demonstrated that the ether of physics was one of the most flexible of all concepts in the natural sciences. Cantor and Hodge’s seminal collection of essays of 1981 showed how during the eighteenth and nineteenth centuries British and European natural philosophers invented a range of ethers to fulfil diverse functions from the chemical and physiological to the physical and theological.1 In religious discourse, for example, Cantor identified “animate” and spiritual ethers invented by neo-Platonists, mystics and some Anglicans to provide a mechanism for supporting their belief in Divine immanence in the cosmos; material, mechanistic and contact-action ethers which appealed to atheists and Low Churchmen because such media enabled activity in the universe without constant and direct Divine intervention; and semi-spiritual/semi-material ethers that appealed to dualists seeking a mechanism for understanding the interaction of mind and matter. 2 The third type proved especially attractive to Oliver Lodge and several other late-Victorian physicists who claimed that the extraordinary physical properties of the ether made it a possible mediator between matter and spirit, and a weapon in their fight against materialistic conceptions of the cosmos. -

Download Report 2010-12
RESEARCH REPORt 2010—2012 MAX-PLANCK-INSTITUT FÜR WISSENSCHAFTSGESCHICHTE Max Planck Institute for the History of Science Cover: Aurora borealis paintings by William Crowder, National Geographic (1947). The International Geophysical Year (1957–8) transformed research on the aurora, one of nature’s most elusive and intensely beautiful phenomena. Aurorae became the center of interest for the big science of powerful rockets, complex satellites and large group efforts to understand the magnetic and charged particle environment of the earth. The auroral visoplot displayed here provided guidance for recording observations in a standardized form, translating the sublime aesthetics of pictorial depictions of aurorae into the mechanical aesthetics of numbers and symbols. Most of the portait photographs were taken by Skúli Sigurdsson RESEARCH REPORT 2010—2012 MAX-PLANCK-INSTITUT FÜR WISSENSCHAFTSGESCHICHTE Max Planck Institute for the History of Science Introduction The Max Planck Institute for the History of Science (MPIWG) is made up of three Departments, each administered by a Director, and several Independent Research Groups, each led for five years by an outstanding junior scholar. Since its foundation in 1994 the MPIWG has investigated fundamental questions of the history of knowl- edge from the Neolithic to the present. The focus has been on the history of the natu- ral sciences, but recent projects have also integrated the history of technology and the history of the human sciences into a more panoramic view of the history of knowl- edge. Of central interest is the emergence of basic categories of scientific thinking and practice as well as their transformation over time: examples include experiment, ob- servation, normalcy, space, evidence, biodiversity or force. -
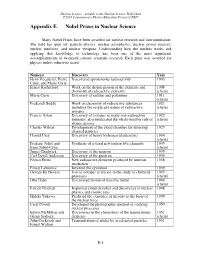
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

Front Matter (PDF)
PROCEEDINGS OF THE ROYAL SOCIETY OF LONDON Series A CONTAINING PAPERS OF A MATHEMATICAL AND PHYSICAL CHARACTER. VOL. G LONDON: P rinted for THE ROYAL SOCIETY and Sold by HARRISON AND SONS, Ltd., ST. MARTIN’S LANE,. PRINTERS in ordinary to h is m a jesty . March, 1922. LONDON : HARRISON AND SONS, LTD., PRINTERS IN ORDINARY TO HIS MAJESTY, ST. MARTIN^ LANE. CONTENTS ---------------- SERIES A. VOL. C. i PAGE Minutes of Meetings, November 3, 1021—February 23, 1922 .................................. i No. A 702.—October 4, 1921. On the Velocity of Sound in Gases at High Temperatures, and the Ratio of the Specific Heats. By Harold B. Dixon, C.B.E., F.R.S., Colin Campbell, D.Sc., and A. Parker, D.Sc....................................................................................................... 1 The Ratio of the Specific Heats of Air and of Carbon Dioxide. By J. R. Partington, D.Sc. Communicated by Dr. J. A. Harker, F.R.S............................ 27 On the Currents induced in a Conductor by the Passage of a Mass of Magnetic Material over it. By E. S. Bieler, M.Sc., 1851 Scholar of McGill University. Communicated by Sir E. Rutherford, F.R.S. (Plate 1 ) ............. ................ 1.......... 50 On some New Formuke for the Numerical Calculation of the Mutual Induction of Coaxial Circles. By Louis V. King, D.Sc., Macdonald Professor of Physics, McGill University, Montreal. Communicated by Prof. A. S. Eve, F.R.S......... 60 Gaseous Combustion at High Pressures. Part II.—The Explosion of Hydrogen-Air and Carbon Monoxide-Air Mixtures. By William A rthur Bone, D.Sc., Ph.D., F.R.S.,'and the late William Arthur Haward, M.Sc., Salters’Research Fellow.. -

Joseph Rotblat: the Nuclear Physicist John Finney 1515
Joseph Rotblat: The Nuclear Physicist John Finney ‘‘I don’t know if you have met Rotblat, a Pole who has been here about nine months. He is an extremely able man, one of the best I have come across for some years.’’1) So wrote the discoverer of the neutron, James Chadwick, to John Cockcroft at the Cavendish Laboratory, Cambridge, in 1940. Over the ten years or so that Rot- blat spent working with Chadwick and in the Liverpool University Physics Department, Chadwick’s initial assessment only grew. So much so that Chadwick tried hard to dissuade Rotblat from taking the Chair in Physics at St Bartholomew’s Hospital in London in 1950. Rotblat’s moving the focus of his work to medical applica- tions would, he said, be a great loss to nuclear physics. Moreover, it would mean, argued Chadwick, that Rotblat would never be elected a Fellow of the Royal Society. In the light of this very positive assessment of Rotblat’s work in nuclear physics from one of the ‘‘greats’’ of the field, it is perhaps a little surprising that, apart from his work on the atomic bomb in the early 1940s, his contributions to the early developments of nu- clear physics are little appreciated. Both his early nuclear physics and later medical physics achievements tend to be overshadowed by his tireless work aimed at ridding the world of nuclear weapons. But his achievements in nuclear physics were also major. They de- serve to be noted and recognized. What were his main achievements as an early nuclear physicist? 1) Letter from J. -

Historic Barriers to Anglo-American Nuclear Cooperation
3 HISTORIC BARRIERS TO ANGLO- AMERICAN NUCLEAR COOPERATION ANDREW BROWN Despite being the closest of allies, with shared values and language, at- tempts by the United Kingdom and the United States to reach accords on nuclear matters generated distrust and resentment but no durable arrangements until the Mutual Defense Agreement of 1958. There were times when the perceived national interests of the two countries were unsynchronized or at odds; periods when political leaders did not see eye to eye or made secret agreements that remained just that; and when espionage, propaganda, and public opinion caused addi- tional tensions. STATUS IMBALANCE The Magna Carta of the nuclear age is the two-part Frisch-Peierls mem- orandum. It was produced by two European émigrés, Otto Frisch and Rudolf Peierls, at Birmingham University in the spring of 1940. Un- like Einstein’s famous letter to President Franklin D. Roosevelt, with its vague warning that a powerful new bomb might be constructed from uranium, the Frisch-Peierls memorandum set out detailed technical arguments leading to the conclusion that “a moderate amount of U-235 [highly enriched uranium] would indeed constitute an extremely effi- cient explosive.” Like Einstein, Frisch and Peierls were worried that the Germans might already be working toward an atomic bomb against which there would be no defense. By suggesting “a counter-threat with a similar bomb,” they first enunciated the concept of mutual deterrence and recommended “start[ing] production as soon as possible, even if 36 Historic Barriers to Anglo-American Nuclear Cooperation 37 it is not intended to use the bomb as a means of attack.”1 Professor Mark Oliphant from Birmingham convinced the UK authorities that “the whole thing must be taken rather seriously,”2 and a small group of senior scientists came together as the Maud Committee.