Transition Metal Coordination Chemistry What Is a Transition Metal?
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Powerful Framework for the Chemical Design of Molecular Nanomagnets Yan Duan†,1,2, Joana T
Data mining, dashboards and statistics: a powerful framework for the chemical design of molecular nanomagnets Yan Duan†,1,2, Joana T. Coutinho†,*,1,3, Lorena E. Rosaleny†,*,1, Salvador Cardona-Serra1, José J. Baldoví1, Alejandro Gaita-Ariño*,1 1 Instituto de Ciencia Molecular (ICMol), Universitat de València, C/ Catedrático José Beltrán 2, 46980 Paterna, Spain 2 Department of Chemistry and the Ilse Katz Institute for Nanoscale Science & Technology, Ben-Gurion University of the Negev, Beer Sheva 84105, Israel 3 Centre for Rapid and Sustainable Product Development, Polytechnic of Leiria, 2430-028 Marinha Grande, Portugal *e-mail: [email protected], [email protected], [email protected] Abstract Three decades of intensive research in molecular nanomagnets have brought the magnetic memory in molecules from liquid helium to liquid nitrogen temperature. The enhancement of this operational temperature relies on a wise choice of the magnetic ion and the coordination environment. However, serendipity, oversimplified theories and chemical intuition have played the main role. In order to establish a powerful framework for statistically driven chemical design, we collected chemical and physical data for lanthanide-based nanomagnets to create a catalogue of over 1400 published experiments, developed an interactive dashboard (SIMDAVIS) to visualise the dataset, and applied inferential statistical analysis to it. We found that the effective energy barrier derived from the Arrhenius equation displays an excellent correlation with the magnetic memory, and that among all chemical families studied, only terbium bis-phthalocyaninato sandwiches and dysprosium metallocenes consistently present magnetic memory up to high temperature, but that there are some promising strategies for improvement. -

And Cobalt(III): Syntheses, Structures, and Ligand Field Parameters
Inorg. Chem. 2005, 44, 8459−8468 Homoleptic Trimethylsilylacetylide Complexes of Chromium(III), Iron(II), and Cobalt(III): Syntheses, Structures, and Ligand Field Parameters Louise A. Berben and Jeffrey R. Long* Contribution from the Department of Chemistry, UniVersity of California, Berkeley California 94720-1460 Received September 10, 2005 A straightforward method for synthesizing soluble homoleptic trimethylsilylacetylide complexes of first-row transition metal ions is presented. Reaction of anhydrous CrCl2 with an excess of LiCCSiMe3 in THF at −25 °C affords orange Li3[Cr(CCSiMe3)6]‚6THF (1), while analogous reactions employing M(CF3SO3)2 (M ) Fe or Co) generate pale yellow Li4[Fe(CCSiMe3)6]‚4LiCCSiMe3‚4Et2O(2) and colorless Li3[Co(CCSiMe3)6]‚6THF (3). Slightly modified reaction conditions lead to Li8[Cr2O4(CCSiMe3)6]‚6LiCCSiMe3‚4glyme (4), featuring a bis-µ-oxo-bridged binuclear complex, and Li3[Co(CCSiMe3)5(CCH)]‚LiCF3SO3‚8THF (5). The crystal structures of 1−3 show the trimethylsilyl- acetylide complexes to display an octahedral coordination geometry, with M−C distances of 2.077(3), 1.917(7)− 1.935(7), and 1.908(3) Å for M ) CrIII,FeII, and CoIII, respectively, and nearly linear M−CtC angles. The UV− 3- 4 4 visible absorption spectrum of [Cr(CCSiMe3)6] in hexanes exhibits one spin-allowed d−d transition ( T2g r A1g) 4- 3- and three lower-energy spin-forbidden d−d transitions. The spectra of [Fe(CCSiMe3)6] and [Co(CCSiMe3)6] in acetonitrile display high-intensity charge-transfer bands, which obscure all d−d transitions except for the lowest- 1 1 energy spin-allowed band ( T1g r A1g) of the latter complex. -

Coordination Chemistry (See Chapter 20, H&S 3Rd Ed.)
Coordination Chemistry (see Chapter 20, H&S 3rd Ed.) Transition metal elements, particularly ions, have a strong tendency to form compounds with Lewis bases. Why? Lewis base: electron pair donor egs. H2O, NH3 n+ Lewis acid: electron pair acceptor egs. M , BH3 ‘low lying’ empty orbitals: lower in energy than HOMO of Lewis case Lewis acidity enhanced by positive charge so TM ions are good Lewis bases 2+ 3+ familiar examples: AlCl3, BF3 from main group and M , M in TM series Type of bonding in these compounds? ‘coordinative’ or ‘dative’ bonds to form Lewis base ‘adducts’ L→M TM-Lewis base ‘adducts’ are often referred to as (i) TM ‘complexes’ OR (ii) coordination complexes NB: still polar covalent bonds – this just defines where the pair of electrons came from Some more definitions: ligand: molecule or ion that can act as a Lewis base to a metal - egs. Cl , PR3, ROR (R = generic term for an alkyl) donor atoms: atoms that are in direct contact with the metal ion O in R2O→M denticity: number of donor atoms per ligand; range from 1 to >6 unidentate bidentate tridentate O H O H2N NH2 2 O O water en diglyme N O O O N N O O N porphin 15-C-5 tetradentate pentadentate chelate: multidentate ligands forms rings; M tend to be very stable, OO especially 5 and 6 member chelate rings acac ‘bite’ or ‘bite angle’: a measure of the span or size of a chelate ligand; depends on M-L distance M OO acac coordination complex MLn generic terms commonly used: L neutral Lewis base (electron pair donor) X anionic donor (eg. -
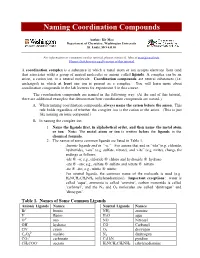
Naming Coordination Compounds
Naming Coordination Compounds Author: Kit Mao Department of Chemistry, Washington University St. Louis, MO 63130 For information or comments on this tutorial, please contact K. Mao at [email protected]. Please click here for a pdf version of this tutorial. A coordination complex is a substance in which a metal atom or ion accepts electrons from (and thus associates with) a group of neutral molecules or anions called ligands. A complex can be an anion, a cation ion, or a neutral molecule. Coordination compounds are neutral substances (i.e. uncharged) in which at least one ion is present as a complex. You will learn more about coordination compounds in the lab lectures for experiment 5 in this course. The coordination compounds are named in the following way. (At the end of this tutorial, there are additional examples that demonstrate how coordination compounds are named.) A. When naming coordination compounds, always name the cation before the anion. This rule holds regardless of whether the complex ion is the cation or the anion. (This is just like naming an ionic compound.) B. In naming the complex ion: 1. Name the ligands first, in alphabetical order, and then name the metal atom or ion. Note: The metal atom or ion is written before the ligands in the chemical formula. 2. The names of some common ligands are listed in Table 1. · Anionic ligands end in “-o.” For anions that end in “-ide”(e.g. chloride, hydroxide), “-ate” (e.g. sulfate, nitrate), and “-ite” (e.g. nirite), change the endings as follows: -ide ® -o; e.g., chloride ® chloro and hydroxide ® hydroxo -ate ® -ato; e.g., sulfate ® sulfato and nitrate ® nitrato -ite ® -ito; e.g., nitrite ® nitrito · For neutral ligands, the common name of the molecule is used (e.g. -

Bridging Ligands
Chapter 9 Coordination Chemistry I: Structure and Isomers 9-1 History 9-2 Nomenclature 9-3 Isomerism 9-4 Coordination Numbers and Structure History What is coordination compound? Coordniantion compounds include compound composed of a metal atom or ion and one or more ligands that formally donate electrons to the metal. More specifically, a transition metal surrounded by neutral molecules or anions with a definite geometry. What is ligand? Ligand can be a atom, ion, and molecules. History Prussian blue (German: Preußischblau or Berliner Blau, in English Berlin blue) is a dark blueWhatpigment is coordinationused in paints and compound? formerly in blueprints. Prussian blue wasCoordination discovered complexesby accident were by pai knownnter Heinrich - although Diesbach not understood in Berlin in 1704-5,in which any sense is why - it since is also the known beginning as Berlin of chemistry, blue. (Diesbach e.g. Prussian was attempting to createblue a paint, Aureolin with ,aand red copperhue.) It vitriolhas several. different chemical names, these being iron(III) ferrocyanide, ferric ferrocyanide, iron(III) hexacyanoferrate(II), and ferricThe hexacyanoferrate. key breakthrough Commonly occurred when and c Alfredonveniently Werner it isproposed, simply called "PB. inter alia, that Co(III) bears six ligands in an octahedral geometry. Aureolin (sometimes called Cobalt Yellow) is a pigment used in oil and watercolor painting. Its color index name is PY40 (40th entry on list of yellow pigments). It was first made in 1851 and its chemical composition is potassium cobaltinitrite. Copper(II) sulfate ("sulphate" in most Commonwealth nations) is the chemical compound with the formula CuSO4. This salt exists as a series of compounds that differ in their degree of hydration. -

Complexation
FACULTY OF PHARMACEUTICAL SCIENCES, RAMAUNIVERSITY, KANPUR B.PHARM 3rd SEM PHYSICAL PHARMACEUTICS-I BP302T MR. PEEYUSH Assistant professor Rama university, kanpur Complexation Overview Classification Introduction Metal ion complexes Organic Complexes Inclusion Complexes Methods of Analysis Method of Continuous Variation PH Titration Distribution Method Solubility Method Spectroscopy Learning Objectives 1. Define the three classes of complexes with pharmaceutically relevant examples. 2. Describe chelates, their physically properties, and what differentiates them from organic molecular complexes. 3. Describe the types of forces that hold together organic molecular complexes with examples. 4. Describe the forces in polymer–drug complexes used for drug delivery. 5. Discuss the pharmaceutical applications of cyclodextrins. 6. Describe the methods of analysis of complexes and determine their stoichiometric ratios and stability constants. Classification Introduction Metal ion complexes Organic Complexes Inclusion Complexes INTRODUCTION Complexes are compounds that result from donor–acceptor mechanisms between two or more chemical species. Complexes can be divided broadly into three classes depending the type of the acceptor substance: 1. Metal ion complexes 2. Organic molecular complexes 3. Inclusion complexes Intermolecular forces involved in the formation of complexes: 1. Van der Waals forces. 2. Hydrogen bonds (important in molecular complexes). 3. Coordinate covalence (important in metal complexes). 4. Charge transfer. 5. Hydrophobic interaction. Introduction Types of Complexes Metal Ion Complexes A. Inorganic type B. Chelates C. Olefin type D. Aromatic type II. Organic Molecular Complexes A. Quinhydrone type B. Picric acid type C. Caffeine and other drug complexes D. Polymer type III. Inclusion Compounds A. Channel lattice type B. Layer type C. Clathrates D. Monomolecular type E. -

Point Defects in Lithium Gallate and Gallium Oxide
Air Force Institute of Technology AFIT Scholar Theses and Dissertations Student Graduate Works 8-23-2019 Point Defects in Lithium Gallate and Gallium Oxide Christopher A. Lenyk Follow this and additional works at: https://scholar.afit.edu/etd Part of the Nuclear Engineering Commons Recommended Citation Lenyk, Christopher A., "Point Defects in Lithium Gallate and Gallium Oxide" (2019). Theses and Dissertations. 2369. https://scholar.afit.edu/etd/2369 This Dissertation is brought to you for free and open access by the Student Graduate Works at AFIT Scholar. It has been accepted for inclusion in Theses and Dissertations by an authorized administrator of AFIT Scholar. For more information, please contact [email protected]. POINT DEFECTS IN LITHIUM GALLATE AND GALLIUM OXIDE DISSERTATION Christopher A. Lenyk, Lt Col, USAF AFIT-ENP-DS-19-S-023 DEPARTMENT OF THE AIR FORCE AIR UNIVERSITY AIR FORCE INSTITUTE OF TECHNOLOGY Wright-Patterson Air Force Base, Ohio DISTRIBUTION STATEMENT A. APPROVED FOR PUBLIC RELEASE; DISTRIBUTION UNLIMITED. The views expressed in this document are those of the author and do not reflect the official policy or position of the United States Air Force, the United States Department of Defense or the United States Government. This material is declared a work of the U.S. Government and is not subject to copyright protection in the United States. AFIT-ENP-DS-19-S-023 POINT DEFECTS IN LITHIUM GALLATE AND GALLIUM OXIDE DISSERTATION Presented to the Faculty Graduate School of Engineering and Management Air Force Institute of Technology Air University Air Education and Training Command in Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy in Nuclear Engineering Christopher A. -

Inorganic Chemistry , 6Th Ed
Chapter 1 8 Interpretation of Spectra In the previous chapter the topic of spectral studies on coordination compounds was introduced only briefl y in connection with ligand fi eld theory, and some of the attendant problems that are associ- ated with interpreting the spectra were described. In this chapter, a more complete description will be presented of the process of interpreting spectra of complexes. It is from the analysis of spectra that we obtain information about energies of spectroscopic states in metal ions and the effects produced by different ligands on the d orbitals. However, it is fi rst necessary to know what spectroscopic states are appropriate for various metal ions. The analysis then progresses to how the spectroscopic states for the metal ions are affected by the presence of the ligands and how ligand fi eld parameters are determined for spectral data. 18.1 SPLITTING OF SPECTROSCOPIC STATES As we have seen, an understanding of spin-orbit coupling is necessary to determine the spectroscopic states that exist for various electron confi gurations, d n (see Section 2.6). Because they will be needed frequently in this chapter, the spectroscopic states that result from spin-orbit coupling in d n ions that have degenerate d orbitals are summarized in Table 18.1 . The spectroscopic states shown in Table 18.1 are those that arise for the so-called free or gaseous ion. When a metal ion is surrounded by ligands in a coordination compound, those ligands generate an electrostatic fi eld that removes the degeneracy of the d orbitals. The result is that eg and t 2g subsets of orbitals are produced. -

The Intensity of Ligand Absorption
Western Kentucky University TopSCHOLAR® Masters Theses & Specialist Projects Graduate School 8-1-1973 The nI tensity of Ligand Absorption Shing-Bong Chen Western Kentucky University Follow this and additional works at: http://digitalcommons.wku.edu/theses Part of the Chemistry Commons Recommended Citation Chen, Shing-Bong, "The nI tensity of Ligand Absorption" (1973). Masters Theses & Specialist Projects. Paper 1018. http://digitalcommons.wku.edu/theses/1018 This Thesis is brought to you for free and open access by TopSCHOLAR®. It has been accepted for inclusion in Masters Theses & Specialist Projects by an authorized administrator of TopSCHOLAR®. For more information, please contact [email protected]. ny THE INTENSITY OF LIGAND ABSORPTION A Thesis Presented to the Faculty of the Department of Chemistry Western Kentucky University Bowling Green, Kentucky in Partial Fulfillment of the Requirements for the Degree Master of Science by Shing-Bong Chen August 1973 THE INTENSITY OP LIGAND ABSORPTION APPROVED (Date) Director of ' TheSIS —— r •Mb >„ fir t-73 :an of the Graduate School ACKNOWLEDGMENT I would like to express gratitude to my research advisor, Dr. Earl F. Pearson, who has given freely of his time and knowledge to show me the way to scientific research. His critical advice and encour- agement are especially appreciated. In addition, I wish to thank the members of the Department of Chemistry of VJestern Kentucky University for their invaluable discussions and suggestions. I also wish to thank Mrs. Linda Moore for typing this thesis. Shing-Bong Chen iii TABLE OP CONTENTS Page ACKNOVJIEDGEJ-'ENTS ±±± LIST OF TABLES v LIST OF FIGURES ; vl ABSTRACT vll INTRDDUCTICN 1 Chapter I. -

Rapid Colorimetric Detection of Cyanide
Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2012 Rapid Colorimetric Detection of Cyanide Männel-Croisé, Christine Posted at the Zurich Open Repository and Archive, University of Zurich ZORA URL: https://doi.org/10.5167/uzh-74766 Dissertation Published Version Originally published at: Männel-Croisé, Christine. Rapid Colorimetric Detection of Cyanide. 2012, University of Zurich, Faculty of Science. Rapid Colorimetric Detection of Cyanide Dissertation zur Erlangung der naturwissenschaftlichen Doktorwürde (Dr. sc. nat.) vorgelegt der Mathematisch-naturwissenschaftlichen Fakultät der Universität Zürich von Christine Männel-Croisé aus Deutschland Promotionskomitee Prof. Dr. Roger Alberto (Vorsitz) Dr. Felix Zelder (Leitung) Prof. Dr. Roland K. O. Sigel Zürich 2012 Summary iii New strategies for the rapid, straightforward colorimetric detection of cyanide in complex samples with corrin-based chemosensors have been developed. In principle, laboratory equipment is not required. The methods are, therefore, promising for applications by non-expert users for the detection of blood cyanide in emergency situations and for water and food analysis, particularly in tropical countries. Challenges in these situations are posed by the complex sample medium, the low levels of detection and the selectivity of the methods. This thesis presents the first method for the rapid visual detection of blood cyanide using immobilised corrinoids and solid-phase extraction. The result in form of a colour change is obtained within a few minutes using only 1 mL of blood. The semi- quantitative determination of blood cyanide content is achieved by means of a colour chart whereas quantitative determinations are possible by diffuse reflectance spectroscopy or a hand-held spectrophotometer. -

Advanced Inorganic Chemistry ADVANCED INORGANIC CHEMISTRY Diagrams Orgel a Splitting of the Weak Field Dn Ground State Terms in an Octahedral Ligand Field DVANCED
Advanced Inorganic Chemistry ADVANCED INORGANIC CHEMISTRY Diagrams Orgel A Splitting of the weak field dn ground state terms in an octahedral ligand field DVANCED Correlation of spectroscopic terms for dn configuration in O complexes I h NORGANIC om c um er Terms in O At i N b h Term of states Symmetry S 1 A1g P 3 T 1g D 5 T2g + Eg C HEMISTRY F 7 T1g + T2g + A2g Ground state determined by inspection of degeneracy of terms for given dn ADVANCED INORGANIC CHEMISTRY A Orgel Diagrams DVANCED 3 T1g(P) 4 T1g(P) I 3P 3A 4P NORGANIC 2 2g 5 Eg 4 T2g T1g 3 T2g 2 3 4 4 5 D F F T2g D C 2 3 5 HEMISTRY T2g T1g Eg 1 3 ∆o 4 ∆o 2 ∆ d 4 d ∆ d d o A2g o 2 2 T2g E 3 3 g T1g T2g 3 3 T1g A2g 3 3 T1g T1g(P) Ti3+ V2+ Cr3+ Mn3+ A DVANCED The d-d bands of the d2 ion [V(H O) ]3+ 2 6 I NORGANIC C HEMISTRY The Tanabe-Sugano diagram ADVANCED INORGANIC CHEMISTRY A DVANCED Correlation diagrams between energies of atomic and molecular terms can drawn as so-called I NORGANIC Tanabe-Sugano diagrams for each electron configuration of free ions. C HEMISTRY Y. Tanabe, s. Sugano; J. Phys. Soc, Jap., 9, 753 (1954) Energy values in a Tanabe-Sugano diagram are only given relative to the ground state (x-axis). A DVANCED The simple correlation diagram had multiples of B (the Racah parameter) on the energy axis to denote the relative energies of the atomic terms. -

Coordination Chemistry
P r i m e r Coordination Chemistry Bonds in introductory chemistry are typically classified Ligands can be monodentate or multidentate. according to whether they are ionic or covalent in Monodentate ligands like ammonia would coordinate to character. Coordinate covalent bonds are a third the metal via one coordination point while a mulitdentate classification. In this type of bond, a lone pair of electrons ligand would coordinate to the metal via two or more from one chemical species is donated to an empty orbital points. Ethylene diamine (NH2CH2CH2NH2) is an example on another chemical species to form the new bond. This of a bidentate ligand because it coordinates to the metal is different from a covalent bond because both electrons through each of the two amine groups (-NH2). come from one atom or molecule but are shared as in a typical covalent bond. Unlike an ionic bond, a coordinate The number of coordination points in a complex is covalent bond does not rely on formal electrostatic referred to as the coordination number. Coordination attraction between a cation and an anion to form. numbers of six and four are the most common and result Ammonia-borane complexes are an example of this type in geometries that are octahedral (6), tetrahedral (4), or of bonding (Figure 1). square planar (4) (Figure 3). Figure 1: Ammonia-borane complexes contain a Figure 3: Common geometries of coordination complexes coordinate covalent bond where the lone pair of with coordination numbers of 6 and 4. electrons on ammonia (NH ) are donated to an empty p 3 Coordination numbers and geometries vary dramatically, orbital on borane (BH ) to give NH BH .