Universidad De Murcia
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Effect of Formica Aserva Forel (Hymenoptera: Formicidae) on Ground Dwelling Arthropods in Central British Columbia
EFFECT OF FORMICA ASERVA FOREL (HYMENOPTERA: FORMICIDAE) ON GROUND DWELLING ARTHROPODS IN CENTRAL BRITISH COLUMBIA by Kendra Gail Schotzko B.S., University of Idaho, 2008 THESIS SUBMITTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF MASTER OF SCIENCE IN NATURAL RESOURCES AND ENVIRONMENTAL STUDIES (BIOLOGY) UNIVERSITY OF NORTHERN BRITISH COLUMBIA June 2012 © Kendra G. Schotzko, 2012 Library and Archives Bibliotheque et Canada Archives Canada Published Heritage Direction du 1+1 Branch Patrimoine de I'edition 395 Wellington Street 395, rue Wellington Ottawa ON K1A0N4 Ottawa ON K1A 0N4 Canada Canada Your file Votre reference ISBN: 978-0-494-94131-7 Our file Notre reference ISBN: 978-0-494-94131-7 NOTICE: AVIS: The author has granted a non L'auteur a accorde une licence non exclusive exclusive license allowing Library and permettant a la Bibliotheque et Archives Archives Canada to reproduce, Canada de reproduire, publier, archiver, publish, archive, preserve, conserve, sauvegarder, conserver, transmettre au public communicate to the public by par telecommunication ou par I'lnternet, preter, telecommunication or on the Internet, distribuer et vendre des theses partout dans le loan, distrbute and sell theses monde, a des fins commerciales ou autres, sur worldwide, for commercial or non support microforme, papier, electronique et/ou commercial purposes, in microform, autres formats. paper, electronic and/or any other formats. The author retains copyright L'auteur conserve la propriete du droit d'auteur ownership and moral rights in this et des droits moraux qui protege cette these. Ni thesis. Neither the thesis nor la these ni des extraits substantiels de celle-ci substantial extracts from it may be ne doivent etre imprimes ou autrement printed or otherwise reproduced reproduits sans son autorisation. -
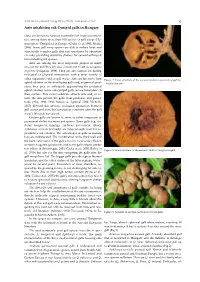
Ants Inhabiting Oak Cynipid Galls in Hungary
North-Western Journal of Zoology 2020, vol.16 (1) - Correspondence: Notes 95 Ants inhabiting oak Cynipid galls in Hungary Oaks are known to harbour extremely rich insect communi- ties, among them more than 100 species of gall wasps (Hy- menoptera: Cynipidae) in Europe (Csóka et al. 2005, Melika 2006). Some gall wasp species are able to induce large and structurally complex galls that can sometimes be abundant on oaks, providing attractive shelters for several arthropod taxa including ant species. Ants are among the most important players in many ecosystems and they are also considered to act as ecosystem engineers (Folgarait, 1998). They are also famous for having ecological or physical interactions with a great variety of other organisms, such as gall wasps. Ants are known to tend Figure 1. Inner structure of the asexual Andricus quercustozae gall in- aphid colonies on the developing galls and, as general pred- habited by ants. ators, they prey on arthropods approaching the protected aphid colonies. Some oak cynipid galls secrete honeydew on their surface. This sweet substrate attracts ants and, in re- turn, the ants protect the galls from predators and parasi- toids (Abe, 1988, 1992; Inouye & Agrawal 2004; Nicholls, 2017). Beyond this obvious ecological interaction between gall wasps and ants, this association continues after the gall wasp’s life cycle has ceased. Certain galls are known to serve as either temporary or permanent shelter for many ant species. Some galls (e.g. An- dricus hungaricus (Hartig), Andricus quercustozae (Bosc), Aphelonyx cerricola (Giraud)) are large enough even for re- productive ant colonies. The advantages of galls as nesting logs are multifaceted. -

Succession in Ant Communities (Hymenoptera: Formicidae) in Deciduous Forest Clear-Cuts – an Eastern European Case Study
EUROPEAN JOURNAL OF ENTOMOLOGYENTOMOLOGY ISSN (online): 1802-8829 Eur. J. Entomol. 114: 92–100, 2017 http://www.eje.cz doi: 10.14411/eje.2017.013 ORIGINAL ARTICLE Succession in ant communities (Hymenoptera: Formicidae) in deciduous forest clear-cuts – an Eastern European case study IOAN TĂUŞAN 1, JENS DAUBER 2, MARIA R. TRICĂ1 and BÁLINT MARKÓ 3 1 Department of Environmental Sciences, Lucian Blaga University of Sibiu; Applied Ecology Research Centre, Dr. Raţiu 5-7, 550012 Sibiu, Romania; e-mails: [email protected], [email protected] 2 Thünen Institute of Biodiversity, Federal Research Institute for Rural Areas, Forestry and Fisheries, Bundesallee 50, D-38116 Braunschweig, Germany; e-mail: [email protected] 3 Hungarian Department of Biology and Ecology, Babeş-Bolyai University, Clinicilor 5-7, 400006 Cluj-Napoca, Romania; e-mails: [email protected], [email protected] Key words. Hymenoptera, Formicidae, ants, deciduous forests, secondary succession, clear-cutting, community structure, pitfall traps Abstract. Clear-cutting, the main method of harvesting in many forests in the world, causes a series of dramatic environmental changes to the forest habitat and removes habitat resources for arboreal and epigeal species. It results in considerable changes in the composition of both plant and animal communities. Ants have many critical roles in the maintenance and functioning of forest ecosystems. Therefore, the response of ants to clear-cutting and the time it takes for an ant community to recover after clear- cutting are important indicators of the effect of this harvesting technique on the forest ecosystem. We investigated ground-dwelling ant communities during secondary succession of deciduous forests in Transylvania, Romania. -

The Ants of Oklahoma Master of Science
THE ANTS OF OKLAHOMA By Jerry H. Young(I\" Bachelor of Science Oklahoma Agricultural and Mechanical College Stillwater, Oklahoma 1955 Submitted to the faculty of the Graduate School of the Oklahoma Agricultural and Mechanical College in partial fulfillment of the requirements for the degree of MASTER OF SCIENCE January 1 1956 tl<lAWMA AGCMCl«.f�Al L �Ci'!AlttCAl e&U.Ull LIBRARY JUL16195 6 THE ANTS OF OKLAHOMA Thesis Approved: Thesis Adviser }>JcMem��f � 't'" he Thesis ) Committee Member of the Thesis Committee 7'4'.��Member of the Thesis Committee Head of the Department ifean of the Graduate School 361565 ii PREFACE The study of the distribution of ants in the United States has been a long and continuous process with many contributors, but the State of Oklahoma has not received the attentions of these observers to any great extent. The only known list of ants of Oklahoma is one prepared by Mo Ro Smith (1935)0 Early in 1954 a survey of the state of Oklahoma was made to determine the species present and their distributiono The results of this survey, which blanketed the entire State, are given in this paper. The author wishes to express his appreciation to Dro Do E. Howell, chairman of the writer's thesis committee, for his valuable assistance and careful guidance in the preparation of this papero Also, much guidance on preparation of this manuscrip_t was received from Drs. Do Eo Bryan, William H. Irwin and F. A. Fenton. Many of the determin ations were made by M. R. Smith.. Vital infonnation was obtained from the museums at Oklahoma Agricultural and Mechanical College and the University of Oklahoma. -

Short Term Response of Ants to the Removal of Ground Cover in Organic Olive Orchards
Eur. J. Entomol. 108: 417–423, 2011 http://www.eje.cz/scripts/viewabstract.php?abstract=1632 ISSN 1210-5759 (print), 1802-8829 (online) Short term response of ants to the removal of ground cover in organic olive orchards MERCEDES CAMPOS1, LUISA FERNÁNDEZ1, FRANCISCA RUANO3, BELÉN COTES1, MANUEL CÁRDENAS1 and JUAN CASTRO2 1Department of Environmental Protection, Estación Experimental del Zaidín, (CSIC) C/Profesor Albareda n° 1, 18008 – Granada, Spain; e-mail: [email protected] 2IFAPA Centro Camino de Purchil, CAP (Junta de Andalucia), P.O. Box 2027, 18080 – Granada, Spain 3Department of Animal Biology, University of Granada, 18071 – Granada, Spain Key words. Hymenoptera, Formicidae, disturbance, biodiversity, soil management Abstract. Ants are the most abundant group of soil arthropods in olive groves where they are involved in various trophic relation- ships of great importance for crops. The system of soil management is one agricultural practice that has a great effect on ants, so the objective of this study was to compare ant populations in organic olive orchards with a ground cover of natural vegetation and others where this natural vegetation is mechanically removed at the beginning of June. Ants were sampled using pitfall traps at 14, 30, 70 and 90 days after the removal of the ground vegetation. Overall, ant biodiversity did not change. However, changes were observed in the abundance of ant species, in particular, in those species that build shallow nests in the soil, both between the rows of trees and under the canopy of olive trees. In contrast, deep nesting species, such as Messor barbarus, were not affected. -

Food Collection by Cataglyphis Iberica (EM.) (Hymenoptera, Formicidae)
POLSKA AKADEMIA NAUK INSTYTUT ZOOLOGII ANNALES ZOOLOGICI Tom 41 Warszawa, 30 V 1988 Nr 18 Xim Cerd A Food collection byCałaglyphis iberica (Em.) (Hyrnenoptera, Formicidae) [With 3 figures and 3 tables im the text] A bstract. CataglypMs iberica is an ant that lives in hot dry places and forages individually. Its staple food are arthropods, mainly insects. The range of food items taken by the workers was determined using two methods: the analysis of the remains found in the middens of the nests and that of the prey brought by the foragers. Even though the main groups that make up the diet are the same in both cases (ants, Coleoptera, Hemiptera), there are some differences because the soft prey (Orthoptera, Diptera, larvae) is more scarce in the middens. INTRODUCTION CataglypMs iberica (Emery , 1906) is an endemic species of the Iberian Peninsula and its distribution area is limited to Spain and Portugal (C olling - wood and Y arrow 1969). So far studies have been carried out on the forag ing activity (de H aro 1982, 1983, C erda 1986) and on the social carrying between nests (de H aro 1981, 1983, de H aro and C erda 1984, C erda 1986). As a new step to the understanding of the ecology of this species, the present work deals with the dietary spectrum. CataglypMs iberica nests in very dry locations, characteristically very sunny and with scant vegetation. Usually the societies are monogynic and polycalic (with several nests related by social carrying and only one of wich has a queen) (de H aro and C erda 1984). -

Digging Deeper Into the Ecology of Subterranean Ants: Diversity and Niche Partitioning Across Two Continents
diversity Article Digging Deeper into the Ecology of Subterranean Ants: Diversity and Niche Partitioning across Two Continents Mickal Houadria * and Florian Menzel Institute of Organismic and Molecular Evolution, Johannes-Gutenberg-University Mainz, Hanns-Dieter-Hüsch-Weg 15, 55128 Mainz, Germany; [email protected] * Correspondence: [email protected] Abstract: Soil fauna is generally understudied compared to above-ground arthropods, and ants are no exception. Here, we compared a primary and a secondary forest each on two continents using four different sampling methods. Winkler sampling, pitfalls, and four types of above- and below-ground baits (dead, crushed insects; melezitose; living termites; living mealworms/grasshoppers) were applied on four plots (4 × 4 grid points) on each site. Although less diverse than Winkler samples and pitfalls, subterranean baits provided a remarkable ant community. Our baiting system provided a large dataset to systematically quantify strata and dietary specialisation in tropical rainforest ants. Compared to above-ground baits, 10–28% of the species at subterranean baits were overall more common (or unique to) below ground, indicating a fauna that was truly specialised to this stratum. Species turnover was particularly high in the primary forests, both concerning above-ground and subterranean baits and between grid points within a site. This suggests that secondary forests are more impoverished, especially concerning their subterranean fauna. Although subterranean ants rarely displayed specific preferences for a bait type, they were in general more specialised than above-ground ants; this was true for entire communities, but also for the same species if they foraged in both strata. Citation: Houadria, M.; Menzel, F. -

Larvae of the Green Lacewing Mallada Desjardinsi (Neuroptera: Chrysopidae) Protect Themselves Against Aphid-Tending Ants by Carrying Dead Aphids on Their Backs
Appl Entomol Zool (2011) 46:407–413 DOI 10.1007/s13355-011-0053-y ORIGINAL RESEARCH PAPER Larvae of the green lacewing Mallada desjardinsi (Neuroptera: Chrysopidae) protect themselves against aphid-tending ants by carrying dead aphids on their backs Masayuki Hayashi • Masashi Nomura Received: 6 March 2011 / Accepted: 11 May 2011 / Published online: 28 May 2011 Ó The Japanese Society of Applied Entomology and Zoology 2011 Abstract Larvae of the green lacewing Mallada desj- Introduction ardinsi Navas are known to place dead aphids on their backs. To clarify the protective role of the carried dead Many ants tend myrmecophilous homopterans such as aphids against ants and the advantages of carrying them for aphids and scale insects, and utilize the secreted honeydew lacewing larvae on ant-tended aphid colonies, we carried as a sugar resource; in return, the homopterans receive out some laboratory experiments. In experiments that beneficial services from the tending ants (Way 1963; Breton exposed lacewing larvae to ants, approximately 40% of the and Addicott 1992; Nielsen et al. 2010). These mutualistic larvae without dead aphids were killed by ants, whereas no interactions between ants and homopterans reduce the larvae carrying dead aphids were killed. The presence of survival and abundance of other arthropods, including the dead aphids did not affect the attack frequency of the non-honeydew-producing herbivores and other predators ants. When we introduced the lacewing larvae onto plants (Bristow 1984; Buckley 1987; Suzuki et al. 2004; Kaplan colonized by ant-tended aphids, larvae with dead aphids and Eubanks 2005), because the tending ants become more stayed for longer on the plants and preyed on more aphids aggressive and attack arthropods that they encounter on than larvae without dead aphids. -

Zootaxa, Hymenoptera, Formicidae, Dolichoderinae
Zootaxa 776: 1–10 (2004) ISSN 1175-5326 (print edition) www.mapress.com/zootaxa/ ZOOTAXA 776 Copyright © 2004 Magnolia Press ISSN 1175-5334 (online edition) A new species of the genus Bothriomyrmex Emery, 1869 (Hymenoptera: Formicidae: Dolichoderinae) from Costa Rica DMITRY A. DUBOVIKOFF1 & JOHN T. LONGINO2 1 Department of Entomology, Faculty of Biology and Soil Sciences, Saint Petersburg State University, 7/9 Uni- versiteskaya nab., St. Petersburg, 199034, Russia, [email protected] 2 The Evergreen State College, Olympia WA 98505 USA, [email protected] Abstract Bothriomyrmex paradoxus Dubovikov and Longino sp. nov. is described from Costa Rica, based on two collections from widely separated localities. These are the first collections of the genus Bothri- omyrmex in the Americas. The genus Bothriomyrmex can be divided into Palearctic species (Both- riomyrmex s.s.) and Oriental and Australian species, based on differences in palp formula and queen wing venation. Bothriomyrmex paradoxus shares palp and wing characters with the Palearc- tic species. It is probably native to Central America and long separated from its Old World relatives. Key words: Bothriomyrmex, Formicidae, Dolichoderinae, Costa Rica Introduction The subfamily Dolichoderinae is comprised of 22 genera, the majority of which are Old World (Shattuck 1992, Bolton 2003, Brandão et al. 1999). The dolichoderine genera with significant indigenous radiations in the New World are Azteca, Dolichoderus, Dory- myrmex, Forelius, Linepithema, Liometopum, and Tapinoma. Three additional genera have been reported as rare or introduced elements. Technomyrmex, a diverse genus in the Old World, is represented in the New World by one introduced tramp species and, paradoxi- cally, one native species from Panama and Costa Rica (Wheeler 1934, Longino pers. -

Easychair Preprint Ant Microbial Endosymbionts and the Emergent
EasyChair Preprint № 1661 Ant microbial endosymbionts and the emergent properties of social groups Alessio Sclocco, Shirlyn Jia Yun Ong and Serafino Teseo EasyChair preprints are intended for rapid dissemination of research results and are integrated with the rest of EasyChair. October 14, 2019 Ant microbial endosymbionts and the emergent properties of social groups Alessio Sclocco1, Shirlyn Jia Yun Ong2 and Serafino Teseo2† 1Netherlands eScience Center, Amsterdam, The Netherlands (E-mail: [email protected]) 2School of Biological Sciences, Nanyang Technological University, Singapore (Tel: +65 63167088; E-mail: [email protected]) Abstract: In the last fifteen years, research on animal models has provided advances on how gut symbiotic microbes affect behavior and its underlying neurophysiology. However, most studies on the gut microbiota only take into exam individual behavior without considering social dynamics. Contrarily, animals and humans live in complex societies where they constantly adjust physiology and behavior to social interactions. To improve our understanding of how microbes and hosts interact and produce functional individual, social and collective phenotypes, we need to broaden our experimental approaches to a group-level dimension. The ideal models for this purpose are social animals living in stable symbioses with microbes, such as eusocial insects. In our research, we investigate Camponotus carpenter ants and their obligate bacterial symbiont Blochmannia from a behavioral ecology perspective. We aim to create ant colonies including differential proportions of bacteria-free individuals by suppressing Blochmannia with antibiotics. Then, using a machine learning-based video tracking system, we will study network features and group-level behavior of such experimental colonies. Keywords: Social Evolution; Microbiota-Gut-Brain Axis; Ants; Group-Level Behavior; Machine Learning; Real Time Data Analysis 1. -

List of Indian Ants (Hymenoptera: Formicidae) Himender Bharti
List of Indian Ants (Hymenoptera: Formicidae) Himender Bharti Department of Zoology, Punjabi University, Patiala, India - 147002. (email: [email protected]/[email protected]) (www.antdiversityindia.com) Abstract Ants of India are enlisted herewith. This has been carried due to major changes in terms of synonymies, addition of new taxa, recent shufflings etc. Currently, Indian ants are represented by 652 valid species/subspecies falling under 87 genera grouped into 12 subfamilies. Keywords: Ants, India, Hymenoptera, Formicidae. Introduction The following 652 valid species/subspecies of myrmecology. This species list is based upon the ants are known to occur in India. Since Bingham’s effort of many ant collectors as well as Fauna of 1903, ant taxonomy has undergone major myrmecologists who have published on the taxonomy changes in terms of synonymies, discovery of new of Indian ants and from inputs provided by taxa, shuffling of taxa etc. This has lead to chaotic myrmecologists from other parts of world. However, state of affairs in Indian scenario, many lists appeared the other running/dynamic list continues to appear on web without looking into voluminous literature on http://www.antweb.org/india.jsp, which is which has surfaced in last many years and currently periodically updated and contains information about the pace at which new publications are appearing in new/unconfirmed taxa, still to be published or verified. Subfamily Genus Species and subspecies Aenictinae Aenictus 28 Amblyoponinae Amblyopone 3 Myopopone -

Nest Site Selection During Colony Relocation in Yucatan Peninsula Populations of the Ponerine Ants Neoponera Villosa (Hymenoptera: Formicidae)
insects Article Nest Site Selection during Colony Relocation in Yucatan Peninsula Populations of the Ponerine Ants Neoponera villosa (Hymenoptera: Formicidae) Franklin H. Rocha 1, Jean-Paul Lachaud 1,2, Yann Hénaut 1, Carmen Pozo 1 and Gabriela Pérez-Lachaud 1,* 1 El Colegio de la Frontera Sur, Conservación de la Biodiversidad, Avenida Centenario km 5.5, Chetumal 77014, Quintana Roo, Mexico; [email protected] (F.H.R.); [email protected] (J.-P.L.); [email protected] (Y.H.); [email protected] (C.P.) 2 Centre de Recherches sur la Cognition Animale (CRCA), Centre de Biologie Intégrative (CBI), Université de Toulouse; CNRS, UPS, 31062 Toulouse, France * Correspondence: [email protected]; Tel.: +52-98-3835-0440 Received: 15 January 2020; Accepted: 19 March 2020; Published: 23 March 2020 Abstract: In the Yucatan Peninsula, the ponerine ant Neoponera villosa nests almost exclusively in tank bromeliads, Aechmea bracteata. In this study, we aimed to determine the factors influencing nest site selection during nest relocation which is regularly promoted by hurricanes in this area. Using ants with and without previous experience of Ae. bracteata, we tested their preference for refuges consisting of Ae. bracteata leaves over two other bromeliads, Ae. bromeliifolia and Ananas comosus. We further evaluated bromeliad-associated traits that could influence nest site selection (form and size). Workers with and without previous contact with Ae. bracteata significantly preferred this species over others, suggesting the existence of an innate attraction to this bromeliad. However, preference was not influenced by previous contact with Ae. bracteata. Workers easily discriminated between shelters of Ae. bracteata and A.