Multi-Scale Systems Genomics Analysis Predicts Pathways, Cell Types and Drug Targets Involved in Normative Human Cognition Varia
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Oxidative Stress-Induced Chromosome Breaks Within
Tan et al. Human Genomics (2018) 12:29 https://doi.org/10.1186/s40246-018-0160-8 PRIMARY RESEARCH Open Access Oxidative stress-induced chromosome breaks within the ABL gene: a model for chromosome rearrangement in nasopharyngeal carcinoma Sang-Nee Tan1, Sai-Peng Sim1* and Alan Soo-Beng Khoo2 Abstract Background: The mechanism underlying chromosome rearrangement in nasopharyngeal carcinoma (NPC) remains elusive. It is known that most of the aetiological factors of NPC trigger oxidative stress. Oxidative stress is a potent apoptotic inducer. During apoptosis, chromatin cleavage and DNA fragmentation occur. However, cells may undergo DNA repair and survive apoptosis. Non-homologous end joining (NHEJ) pathway has been known as the primary DNA repair system in human cells. The NHEJ process may repair DNA ends without any homology, although region of microhomology (a few nucleotides) is usually utilised by this DNA repair system. Cells that evade apoptosis via erroneous DNA repair may carry chromosomal aberration. Apoptotic nuclease was found to be associated with nuclear matrix during apoptosis. Matrix association region/scaffold attachment region (MAR/SAR) is the binding site of the chromosomal DNA loop structure to the nuclear matrix. When apoptotic nuclease is associated with nuclear matrix during apoptosis, it potentially cleaves at MAR/SAR. Cells that survive apoptosis via compromised DNA repair may carry chromosome rearrangement contributing to NPC tumourigenesis. The Abelson murine leukaemia (ABL) gene at 9q34 was targeted in this study as 9q34 is a common region of loss in NPC. This study aimed to identify the chromosome breakages and/or rearrangements in the ABL gene in cells undergoing oxidative stress-induced apoptosis. -

Design of a Bioactive Small Molecule That Targets R(AUUCU) Repeats in Spinocerebellar Ataxia 10
ARTICLE Received 24 Jun 2015 | Accepted 18 Apr 2016 | Published 1 Jun 2016 DOI: 10.1038/ncomms11647 OPEN Design of a bioactive small molecule that targets r(AUUCU) repeats in spinocerebellar ataxia 10 Wang-Yong Yang1, Rui Gao2, Mark Southern3, Partha S. Sarkar2 & Matthew D. Disney1 RNA is an important target for chemical probes of function and lead therapeutics; however, it is difficult to target with small molecules. One approach to tackle this problem is to identify compounds that target RNA structures and utilize them to multivalently target RNA. Here we show that small molecules can be identified to selectively bind RNA base pairs by probing a library of RNA-focused small molecules. A small molecule that selectively binds AU base pairs informed design of a dimeric compound (2AU-2) that targets the pathogenic RNA, expanded r(AUUCU) repeats, that causes spinocerebellar ataxia type 10 (SCA10) in patient- derived cells. Indeed, 2AU-2 (50 nM) ameliorates various aspects of SCA10 pathology including improvement of mitochondrial dysfunction, reduced activation of caspase 3, and reduction of nuclear foci. These studies provide a first-in-class chemical probe to study SCA10 RNA toxicity and potentially define broadly applicable compounds targeting RNA AU base pairs in cells. 1 Departments of Chemistry and Neuroscience, The Scripps Research Institute, Scripps Florida, Jupiter, Florida 33458, USA. 2 Mitchell Center for Neurodegenerative Disorders, Department of Neurology, Neuroscience and Cell Biology, University of Texas Medical Branch, Galveston, Texas 77555, USA. 3 Informatics Core, The Scripps Research Institute, Scripps Florida, Jupiter, Florida 33458, USA. Correspondence and requests for materials should be addressed to M.D.D. -

Regulation of Neuronal Gene Expression and Survival by Basal NMDA Receptor Activity: a Role for Histone Deacetylase 4
The Journal of Neuroscience, November 12, 2014 • 34(46):15327–15339 • 15327 Cellular/Molecular Regulation of Neuronal Gene Expression and Survival by Basal NMDA Receptor Activity: A Role for Histone Deacetylase 4 Yelin Chen,1 Yuanyuan Wang,1 Zora Modrusan,3 Morgan Sheng,1 and Joshua S. Kaminker1,2 Departments of 1Neuroscience, 2Bioinformatics and Computational Biology, and 3Molecular Biology, Genentech Inc., South San Francisco, California 94080 Neuronal gene expression is modulated by activity via calcium-permeable receptors such as NMDA receptors (NMDARs). While gene expression changes downstream of evoked NMDAR activity have been well studied, much less is known about gene expression changes that occur under conditions of basal neuronal activity. In mouse dissociated hippocampal neuronal cultures, we found that a broad NMDAR antagonist, AP5, induced robust gene expression changes under basal activity, but subtype-specific antagonists did not. While some of the gene expression changes are also known to be downstream of stimulated NMDAR activity, others appear specific to basal NMDARactivity.ThegenesalteredbyAP5treatmentofbasalcultureswereenrichedforpathwaysrelatedtoclassIIahistonedeacetylases (HDACs), apoptosis, and synapse-related signaling. Specifically, AP5 altered the expression of all three class IIa HDACs that are highly expressed in the brain, HDAC4, HDAC5, and HDAC9, and also induced nuclear accumulation of HDAC4. HDAC4 knockdown abolished a subset of the gene expression changes induced by AP5, and led to neuronal death under -

Differentially Expressed on Collagen Networks 1, 2, 10 © 2000-2009 Ingenuity Systems, Inc. All Rights Reserved. Symbol Entrez
Differentially expressed on collagen Networks 1, 2, 10 © 2000-2009 Ingenuity Systems, Inc. All rights reserved. Symbol Entrez Gene Name Affymetrix Fold Change Location Family ALDH1A3 aldehyde dehydrogenase 1 family, member A3 203180_at -5.43125168 Cytoplasm enzyme 209772_s_ Plasma CD24 CD24 molecule at -4.32890229 Membrane other HSD11B2 hydroxysteroid (11-beta) dehydrogenase 2 204130_at -4.1099197 Cytoplasm enzyme Plasma AMOTL2 angiomotin like 2 203002_at -2.82872773 Membrane other transcription DLX2 distal-less homeobox 2 207147_at -2.74996362 Nucleus regulator 221215_s_ RIPK4 receptor-interacting serine-threonine kinase 4 at -2.56556472 Nucleus kinase PLK2 polo-like kinase 2 (Drosophila) 201939_at -2.47054478 Nucleus kinase ALDH3A1 aldehyde dehydrogenase 3 family, memberA1 205623_at -2.30532989 Cytoplasm enzyme TXNRD1 thioredoxin reductase 1 201266_at -2.27936909 Cytoplasm enzyme Extracellular CYR61 cysteine-rich, angiogenic inducer, 61 201289_at -2.09052668 Space other 214212_x_ FERMT2 fermitin family homolog 2 (Drosophila) at -1.87478183 Cytoplasm other Plasma RIT1 Ras-like without CAAX 1 209882_at -1.77586775 Membrane enzyme 210297_s_ Extracellular MSMB microseminoprotein, beta- at -1.72177723 Space other Extracellular PI3 peptidase inhibitor 3, skin-derived 203691_at -1.68135697 Space other ALDH3B1 aldehyde dehydrogenase 3 family, member B1 205640_at -1.67376791 Cytoplasm enzyme 202124_s_ Plasma TRAK2 trafficking protein, kinesin binding 2 at -1.6367793 Membrane transporter BMP and activin membrane-bound inhibitor Plasma BAMBI -

Supplemental Information
Supplemental information Dissection of the genomic structure of the miR-183/96/182 gene. Previously, we showed that the miR-183/96/182 cluster is an intergenic miRNA cluster, located in a ~60-kb interval between the genes encoding nuclear respiratory factor-1 (Nrf1) and ubiquitin-conjugating enzyme E2H (Ube2h) on mouse chr6qA3.3 (1). To start to uncover the genomic structure of the miR- 183/96/182 gene, we first studied genomic features around miR-183/96/182 in the UCSC genome browser (http://genome.UCSC.edu/), and identified two CpG islands 3.4-6.5 kb 5’ of pre-miR-183, the most 5’ miRNA of the cluster (Fig. 1A; Fig. S1 and Seq. S1). A cDNA clone, AK044220, located at 3.2-4.6 kb 5’ to pre-miR-183, encompasses the second CpG island (Fig. 1A; Fig. S1). We hypothesized that this cDNA clone was derived from 5’ exon(s) of the primary transcript of the miR-183/96/182 gene, as CpG islands are often associated with promoters (2). Supporting this hypothesis, multiple expressed sequences detected by gene-trap clones, including clone D016D06 (3, 4), were co-localized with the cDNA clone AK044220 (Fig. 1A; Fig. S1). Clone D016D06, deposited by the German GeneTrap Consortium (GGTC) (http://tikus.gsf.de) (3, 4), was derived from insertion of a retroviral construct, rFlpROSAβgeo in 129S2 ES cells (Fig. 1A and C). The rFlpROSAβgeo construct carries a promoterless reporter gene, the β−geo cassette - an in-frame fusion of the β-galactosidase and neomycin resistance (Neor) gene (5), with a splicing acceptor (SA) immediately upstream, and a polyA signal downstream of the β−geo cassette (Fig. -

Role of RUNX1 in Aberrant Retinal Angiogenesis Jonathan D
Page 1 of 25 Diabetes Identification of RUNX1 as a mediator of aberrant retinal angiogenesis Short Title: Role of RUNX1 in aberrant retinal angiogenesis Jonathan D. Lam,†1 Daniel J. Oh,†1 Lindsay L. Wong,1 Dhanesh Amarnani,1 Cindy Park- Windhol,1 Angie V. Sanchez,1 Jonathan Cardona-Velez,1,2 Declan McGuone,3 Anat O. Stemmer- Rachamimov,3 Dean Eliott,4 Diane R. Bielenberg,5 Tave van Zyl,4 Lishuang Shen,1 Xiaowu Gai,6 Patricia A. D’Amore*,1,7 Leo A. Kim*,1,4 Joseph F. Arboleda-Velasquez*1 Author affiliations: 1Schepens Eye Research Institute/Massachusetts Eye and Ear, Department of Ophthalmology, Harvard Medical School, 20 Staniford St., Boston, MA 02114 2Universidad Pontificia Bolivariana, Medellin, Colombia, #68- a, Cq. 1 #68305, Medellín, Antioquia, Colombia 3C.S. Kubik Laboratory for Neuropathology, Massachusetts General Hospital, 55 Fruit St., Boston, MA 02114 4Retina Service, Massachusetts Eye and Ear Infirmary, Department of Ophthalmology, Harvard Medical School, 243 Charles St., Boston, MA 02114 5Vascular Biology Program, Boston Children’s Hospital, Department of Surgery, Harvard Medical School, 300 Longwood Ave., Boston, MA 02115 6Center for Personalized Medicine, Children’s Hospital Los Angeles, Los Angeles, 4650 Sunset Blvd, Los Angeles, CA 90027, USA 7Department of Pathology, Harvard Medical School, 25 Shattuck St., Boston, MA 02115 Corresponding authors: Joseph F. Arboleda-Velasquez: [email protected] Ph: (617) 912-2517 Leo Kim: [email protected] Ph: (617) 912-2562 Patricia D’Amore: [email protected] Ph: (617) 912-2559 Fax: (617) 912-0128 20 Staniford St. Boston MA, 02114 † These authors contributed equally to this manuscript Word Count: 1905 Tables and Figures: 4 Diabetes Publish Ahead of Print, published online April 11, 2017 Diabetes Page 2 of 25 Abstract Proliferative diabetic retinopathy (PDR) is a common cause of blindness in the developed world’s working adult population, and affects those with type 1 and type 2 diabetes mellitus. -

Identification of Potential Key Genes and Pathway Linked with Sporadic Creutzfeldt-Jakob Disease Based on Integrated Bioinformatics Analyses
medRxiv preprint doi: https://doi.org/10.1101/2020.12.21.20248688; this version posted December 24, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. All rights reserved. No reuse allowed without permission. Identification of potential key genes and pathway linked with sporadic Creutzfeldt-Jakob disease based on integrated bioinformatics analyses Basavaraj Vastrad1, Chanabasayya Vastrad*2 , Iranna Kotturshetti 1. Department of Biochemistry, Basaveshwar College of Pharmacy, Gadag, Karnataka 582103, India. 2. Biostatistics and Bioinformatics, Chanabasava Nilaya, Bharthinagar, Dharwad 580001, Karanataka, India. 3. Department of Ayurveda, Rajiv Gandhi Education Society`s Ayurvedic Medical College, Ron, Karnataka 562209, India. * Chanabasayya Vastrad [email protected] Ph: +919480073398 Chanabasava Nilaya, Bharthinagar, Dharwad 580001 , Karanataka, India NOTE: This preprint reports new research that has not been certified by peer review and should not be used to guide clinical practice. medRxiv preprint doi: https://doi.org/10.1101/2020.12.21.20248688; this version posted December 24, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. All rights reserved. No reuse allowed without permission. Abstract Sporadic Creutzfeldt-Jakob disease (sCJD) is neurodegenerative disease also called prion disease linked with poor prognosis. The aim of the current study was to illuminate the underlying molecular mechanisms of sCJD. The mRNA microarray dataset GSE124571 was downloaded from the Gene Expression Omnibus database. Differentially expressed genes (DEGs) were screened. -
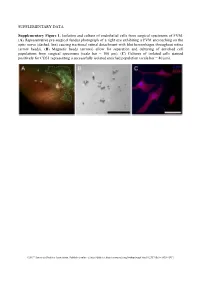
Supplementary Figures and Tables
SUPPLEMENTARY DATA Supplementary Figure 1. Isolation and culture of endothelial cells from surgical specimens of FVM. (A) Representative pre-surgical fundus photograph of a right eye exhibiting a FVM encroaching on the optic nerve (dashed line) causing tractional retinal detachment with blot hemorrhages throughout retina (arrow heads). (B) Magnetic beads (arrows) allow for separation and culturing of enriched cell populations from surgical specimens (scale bar = 100 μm). (C) Cultures of isolated cells stained positively for CD31 representing a successfully isolated enriched population (scale bar = 40 μm). ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Figure 2. Efficient siRNA knockdown of RUNX1 expression and function demonstrated by qRT-PCR, Western Blot, and scratch assay. (A) RUNX1 siRNA induced a 60% reduction of RUNX1 expression measured by qRT-PCR 48 hrs post-transfection whereas expression of RUNX2 and RUNX3, the two other mammalian RUNX orthologues, showed no significant changes, indicating specificity of our siRNA. Functional inhibition of Runx1 signaling was demonstrated by a 330% increase in insulin-like growth factor binding protein-3 (IGFBP3) RNA expression level, a known target of RUNX1 inhibition. Western blot demonstrated similar reduction in protein levels. (B) siRNA- 2’s effect on RUNX1 was validated by qRT-PCR and western blot, demonstrating a similar reduction in both RNA and protein. Scratch assay demonstrates functional inhibition of RUNX1 by siRNA-2. ns: not significant, * p < 0.05, *** p < 0.001 ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. -

Meta-Analysis of Gene Signatures and Key Pathways Indicates
www.nature.com/scientificreports OPEN Meta‑analysis of gene signatures and key pathways indicates suppression of JNK pathway as a regulator of chemo‑resistance in AML Parastoo Modarres1, Farzaneh Mohamadi Farsani1,3, Amir Abas Nekouie2 & Sadeq Vallian1* The pathways and robust deregulated gene signatures involved in AML chemo‑resistance are not fully understood. Multiple subgroups of AMLs which are under treatment of various regimens seem to have similar regulatory gene(s) or pathway(s) related to their chemo‑resistance phenotype. In this study using gene set enrichment approach, deregulated genes and pathways associated with relapse after chemotherapy were investigated in AML samples. Five AML libraries compiled from GEO and ArrayExpress repositories were used to identify signifcantly diferentially expressed genes between chemo‑resistance and chemo‑sensitive groups. Functional and pathway enrichment analysis of diferentially expressed genes was performed to assess molecular mechanisms related to AML chemotherapeutic resistance. A total of 34 genes selected to be diferentially expressed in the chemo‑ resistance compared to the chemo‑sensitive group. Among the genes selected, c-Jun, AKT3, ARAP3, GABBR1, PELI2 and SORT1 are involved in neurotrophin, estrogen, cAMP and Toll‑like receptor signaling pathways. All these pathways are located upstream and regulate JNK signaling pathway which functions as a key regulator of cellular apoptosis. Our expression data are in favor of suppression of JNK pathway, which could induce pro‑apoptotic gene expression as well as down regulation of survival factors, introducing this pathway as a key regulator of drug‑resistance development in AML. Acute myeloid leukemia (AML) is one of the most aggressive, life-threatening hematological malignancies char- acterized by uncontrolled proliferation of abnormal diferentiated and nonfunctional myeloid precursor cells1. -

Autosomal Dominant Cerebellar Ataxia Type I: a Review of the Phenotypic and Genotypic Characteristics
Whaley et al. Orphanet Journal of Rare Diseases 2011, 6:33 http://www.ojrd.com/content/6/1/33 REVIEW Open Access Autosomal dominant cerebellar ataxia type I: A review of the phenotypic and genotypic characteristics Nathaniel Robb Whaley1,2, Shinsuke Fujioka2 and Zbigniew K Wszolek2* Abstract Type I autosomal dominant cerebellar ataxia (ADCA) is a type of spinocerebellar ataxia (SCA) characterized by ataxia with other neurological signs, including oculomotor disturbances, cognitive deficits, pyramidal and extrapyramidal dysfunction, bulbar, spinal and peripheral nervous system involvement. The global prevalence of this disease is not known. The most common type I ADCA is SCA3 followed by SCA2, SCA1, and SCA8, in descending order. Founder effects no doubt contribute to the variable prevalence between populations. Onset is usually in adulthood but cases of presentation in childhood have been reported. Clinical features vary depending on the SCA subtype but by definition include ataxia associated with other neurological manifestations. The clinical spectrum ranges from pure cerebellar signs to constellations including spinal cord and peripheral nerve disease, cognitive impairment, cerebellar or supranuclear ophthalmologic signs, psychiatric problems, and seizures. Cerebellar ataxia can affect virtually any body part causing movement abnormalities. Gait, truncal, and limb ataxia are often the most obvious cerebellar findings though nystagmus, saccadic abnormalities, and dysarthria are usually associated. To date, 21 subtypes have been identified: SCA1-SCA4, SCA8, SCA10, SCA12-SCA14, SCA15/16, SCA17-SCA23, SCA25, SCA27, SCA28 and dentatorubral pallidoluysian atrophy (DRPLA). Type I ADCA can be further divided based on the proposed pathogenetic mechanism into 3 subclasses: subclass 1 includes type I ADCA caused by CAG repeat expansions such as SCA1-SCA3, SCA17, and DRPLA, subclass 2 includes trinucleotide repeat expansions that fall outside of the protein-coding regions of the disease gene including SCA8, SCA10 and SCA12. -

Downloaded from Dbgap (Phs000607.V1)
bioRxiv preprint doi: https://doi.org/10.1101/751933; this version posted September 6, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 1 Genetics and Pathway Analysis of Normative Cognitive Variation 2 in the Philadelphia Neurodevelopmental Cohort 3 4 Authors: 5 Shraddha Pai1, Shirley Hui1, Philipp Weber2, Owen Whitley1,3, Peipei Li4,5, Viviane Labrie4,5, Jan 6 Baumbach2,6, Gary D Bader1,3,7,8 7 8 Affiliations: 9 1. The Donnelly Centre, University of Toronto, Toronto, Canada 10 2. Department of Mathematics and Computer Science, University of Southern Denmark, Odense, 11 Denmark 12 3. Department of Molecular Genetics, University of Toronto, Toronto, Canada 13 4. Center for Neurodegenerative Science, Van Andel Research Institute, Grand Rapids, MI, USA 14 5. Division of Psychiatry and Behavioral Medicine, College of Human Medicine, Michigan State 15 University, Grand Rapids, MI, USA 16 6. TUM School of Life Sciences Weihenstephan, Technical University of Munich, Munich, Germany. 17 7. Department of Computer Science, University of Toronto, Toronto, Canada 18 8. The Lunenfeld-Tanenbaum Research Institute, Mount Sinai Hospital, Toronto, Canada 19 20 21 22 * [email protected] 23 24 Page 1 of 23 bioRxiv preprint doi: https://doi.org/10.1101/751933; this version posted September 6, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. -

A Master Autoantigen-Ome Links Alternative Splicing, Female Predilection, and COVID-19 to Autoimmune Diseases
bioRxiv preprint doi: https://doi.org/10.1101/2021.07.30.454526; this version posted August 4, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. A Master Autoantigen-ome Links Alternative Splicing, Female Predilection, and COVID-19 to Autoimmune Diseases Julia Y. Wang1*, Michael W. Roehrl1, Victor B. Roehrl1, and Michael H. Roehrl2* 1 Curandis, New York, USA 2 Department of Pathology, Memorial Sloan Kettering Cancer Center, New York, USA * Correspondence: [email protected] or [email protected] 1 bioRxiv preprint doi: https://doi.org/10.1101/2021.07.30.454526; this version posted August 4, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. Abstract Chronic and debilitating autoimmune sequelae pose a grave concern for the post-COVID-19 pandemic era. Based on our discovery that the glycosaminoglycan dermatan sulfate (DS) displays peculiar affinity to apoptotic cells and autoantigens (autoAgs) and that DS-autoAg complexes cooperatively stimulate autoreactive B1 cell responses, we compiled a database of 751 candidate autoAgs from six human cell types. At least 657 of these have been found to be affected by SARS-CoV-2 infection based on currently available multi-omic COVID data, and at least 400 are confirmed targets of autoantibodies in a wide array of autoimmune diseases and cancer.