Tropomyosin 1 Genetically Constrains in Vitro Hematopoiesis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Oxidative Stress-Induced Chromosome Breaks Within
Tan et al. Human Genomics (2018) 12:29 https://doi.org/10.1186/s40246-018-0160-8 PRIMARY RESEARCH Open Access Oxidative stress-induced chromosome breaks within the ABL gene: a model for chromosome rearrangement in nasopharyngeal carcinoma Sang-Nee Tan1, Sai-Peng Sim1* and Alan Soo-Beng Khoo2 Abstract Background: The mechanism underlying chromosome rearrangement in nasopharyngeal carcinoma (NPC) remains elusive. It is known that most of the aetiological factors of NPC trigger oxidative stress. Oxidative stress is a potent apoptotic inducer. During apoptosis, chromatin cleavage and DNA fragmentation occur. However, cells may undergo DNA repair and survive apoptosis. Non-homologous end joining (NHEJ) pathway has been known as the primary DNA repair system in human cells. The NHEJ process may repair DNA ends without any homology, although region of microhomology (a few nucleotides) is usually utilised by this DNA repair system. Cells that evade apoptosis via erroneous DNA repair may carry chromosomal aberration. Apoptotic nuclease was found to be associated with nuclear matrix during apoptosis. Matrix association region/scaffold attachment region (MAR/SAR) is the binding site of the chromosomal DNA loop structure to the nuclear matrix. When apoptotic nuclease is associated with nuclear matrix during apoptosis, it potentially cleaves at MAR/SAR. Cells that survive apoptosis via compromised DNA repair may carry chromosome rearrangement contributing to NPC tumourigenesis. The Abelson murine leukaemia (ABL) gene at 9q34 was targeted in this study as 9q34 is a common region of loss in NPC. This study aimed to identify the chromosome breakages and/or rearrangements in the ABL gene in cells undergoing oxidative stress-induced apoptosis. -

Regulation of Neuronal Gene Expression and Survival by Basal NMDA Receptor Activity: a Role for Histone Deacetylase 4
The Journal of Neuroscience, November 12, 2014 • 34(46):15327–15339 • 15327 Cellular/Molecular Regulation of Neuronal Gene Expression and Survival by Basal NMDA Receptor Activity: A Role for Histone Deacetylase 4 Yelin Chen,1 Yuanyuan Wang,1 Zora Modrusan,3 Morgan Sheng,1 and Joshua S. Kaminker1,2 Departments of 1Neuroscience, 2Bioinformatics and Computational Biology, and 3Molecular Biology, Genentech Inc., South San Francisco, California 94080 Neuronal gene expression is modulated by activity via calcium-permeable receptors such as NMDA receptors (NMDARs). While gene expression changes downstream of evoked NMDAR activity have been well studied, much less is known about gene expression changes that occur under conditions of basal neuronal activity. In mouse dissociated hippocampal neuronal cultures, we found that a broad NMDAR antagonist, AP5, induced robust gene expression changes under basal activity, but subtype-specific antagonists did not. While some of the gene expression changes are also known to be downstream of stimulated NMDAR activity, others appear specific to basal NMDARactivity.ThegenesalteredbyAP5treatmentofbasalcultureswereenrichedforpathwaysrelatedtoclassIIahistonedeacetylases (HDACs), apoptosis, and synapse-related signaling. Specifically, AP5 altered the expression of all three class IIa HDACs that are highly expressed in the brain, HDAC4, HDAC5, and HDAC9, and also induced nuclear accumulation of HDAC4. HDAC4 knockdown abolished a subset of the gene expression changes induced by AP5, and led to neuronal death under -

Role of RUNX1 in Aberrant Retinal Angiogenesis Jonathan D
Page 1 of 25 Diabetes Identification of RUNX1 as a mediator of aberrant retinal angiogenesis Short Title: Role of RUNX1 in aberrant retinal angiogenesis Jonathan D. Lam,†1 Daniel J. Oh,†1 Lindsay L. Wong,1 Dhanesh Amarnani,1 Cindy Park- Windhol,1 Angie V. Sanchez,1 Jonathan Cardona-Velez,1,2 Declan McGuone,3 Anat O. Stemmer- Rachamimov,3 Dean Eliott,4 Diane R. Bielenberg,5 Tave van Zyl,4 Lishuang Shen,1 Xiaowu Gai,6 Patricia A. D’Amore*,1,7 Leo A. Kim*,1,4 Joseph F. Arboleda-Velasquez*1 Author affiliations: 1Schepens Eye Research Institute/Massachusetts Eye and Ear, Department of Ophthalmology, Harvard Medical School, 20 Staniford St., Boston, MA 02114 2Universidad Pontificia Bolivariana, Medellin, Colombia, #68- a, Cq. 1 #68305, Medellín, Antioquia, Colombia 3C.S. Kubik Laboratory for Neuropathology, Massachusetts General Hospital, 55 Fruit St., Boston, MA 02114 4Retina Service, Massachusetts Eye and Ear Infirmary, Department of Ophthalmology, Harvard Medical School, 243 Charles St., Boston, MA 02114 5Vascular Biology Program, Boston Children’s Hospital, Department of Surgery, Harvard Medical School, 300 Longwood Ave., Boston, MA 02115 6Center for Personalized Medicine, Children’s Hospital Los Angeles, Los Angeles, 4650 Sunset Blvd, Los Angeles, CA 90027, USA 7Department of Pathology, Harvard Medical School, 25 Shattuck St., Boston, MA 02115 Corresponding authors: Joseph F. Arboleda-Velasquez: [email protected] Ph: (617) 912-2517 Leo Kim: [email protected] Ph: (617) 912-2562 Patricia D’Amore: [email protected] Ph: (617) 912-2559 Fax: (617) 912-0128 20 Staniford St. Boston MA, 02114 † These authors contributed equally to this manuscript Word Count: 1905 Tables and Figures: 4 Diabetes Publish Ahead of Print, published online April 11, 2017 Diabetes Page 2 of 25 Abstract Proliferative diabetic retinopathy (PDR) is a common cause of blindness in the developed world’s working adult population, and affects those with type 1 and type 2 diabetes mellitus. -

Identification of Potential Key Genes and Pathway Linked with Sporadic Creutzfeldt-Jakob Disease Based on Integrated Bioinformatics Analyses
medRxiv preprint doi: https://doi.org/10.1101/2020.12.21.20248688; this version posted December 24, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. All rights reserved. No reuse allowed without permission. Identification of potential key genes and pathway linked with sporadic Creutzfeldt-Jakob disease based on integrated bioinformatics analyses Basavaraj Vastrad1, Chanabasayya Vastrad*2 , Iranna Kotturshetti 1. Department of Biochemistry, Basaveshwar College of Pharmacy, Gadag, Karnataka 582103, India. 2. Biostatistics and Bioinformatics, Chanabasava Nilaya, Bharthinagar, Dharwad 580001, Karanataka, India. 3. Department of Ayurveda, Rajiv Gandhi Education Society`s Ayurvedic Medical College, Ron, Karnataka 562209, India. * Chanabasayya Vastrad [email protected] Ph: +919480073398 Chanabasava Nilaya, Bharthinagar, Dharwad 580001 , Karanataka, India NOTE: This preprint reports new research that has not been certified by peer review and should not be used to guide clinical practice. medRxiv preprint doi: https://doi.org/10.1101/2020.12.21.20248688; this version posted December 24, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. All rights reserved. No reuse allowed without permission. Abstract Sporadic Creutzfeldt-Jakob disease (sCJD) is neurodegenerative disease also called prion disease linked with poor prognosis. The aim of the current study was to illuminate the underlying molecular mechanisms of sCJD. The mRNA microarray dataset GSE124571 was downloaded from the Gene Expression Omnibus database. Differentially expressed genes (DEGs) were screened. -
Supplementary Figures and Tables
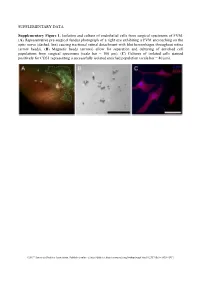
SUPPLEMENTARY DATA Supplementary Figure 1. Isolation and culture of endothelial cells from surgical specimens of FVM. (A) Representative pre-surgical fundus photograph of a right eye exhibiting a FVM encroaching on the optic nerve (dashed line) causing tractional retinal detachment with blot hemorrhages throughout retina (arrow heads). (B) Magnetic beads (arrows) allow for separation and culturing of enriched cell populations from surgical specimens (scale bar = 100 μm). (C) Cultures of isolated cells stained positively for CD31 representing a successfully isolated enriched population (scale bar = 40 μm). ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Figure 2. Efficient siRNA knockdown of RUNX1 expression and function demonstrated by qRT-PCR, Western Blot, and scratch assay. (A) RUNX1 siRNA induced a 60% reduction of RUNX1 expression measured by qRT-PCR 48 hrs post-transfection whereas expression of RUNX2 and RUNX3, the two other mammalian RUNX orthologues, showed no significant changes, indicating specificity of our siRNA. Functional inhibition of Runx1 signaling was demonstrated by a 330% increase in insulin-like growth factor binding protein-3 (IGFBP3) RNA expression level, a known target of RUNX1 inhibition. Western blot demonstrated similar reduction in protein levels. (B) siRNA- 2’s effect on RUNX1 was validated by qRT-PCR and western blot, demonstrating a similar reduction in both RNA and protein. Scratch assay demonstrates functional inhibition of RUNX1 by siRNA-2. ns: not significant, * p < 0.05, *** p < 0.001 ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. -

Meta-Analysis of Gene Signatures and Key Pathways Indicates
www.nature.com/scientificreports OPEN Meta‑analysis of gene signatures and key pathways indicates suppression of JNK pathway as a regulator of chemo‑resistance in AML Parastoo Modarres1, Farzaneh Mohamadi Farsani1,3, Amir Abas Nekouie2 & Sadeq Vallian1* The pathways and robust deregulated gene signatures involved in AML chemo‑resistance are not fully understood. Multiple subgroups of AMLs which are under treatment of various regimens seem to have similar regulatory gene(s) or pathway(s) related to their chemo‑resistance phenotype. In this study using gene set enrichment approach, deregulated genes and pathways associated with relapse after chemotherapy were investigated in AML samples. Five AML libraries compiled from GEO and ArrayExpress repositories were used to identify signifcantly diferentially expressed genes between chemo‑resistance and chemo‑sensitive groups. Functional and pathway enrichment analysis of diferentially expressed genes was performed to assess molecular mechanisms related to AML chemotherapeutic resistance. A total of 34 genes selected to be diferentially expressed in the chemo‑ resistance compared to the chemo‑sensitive group. Among the genes selected, c-Jun, AKT3, ARAP3, GABBR1, PELI2 and SORT1 are involved in neurotrophin, estrogen, cAMP and Toll‑like receptor signaling pathways. All these pathways are located upstream and regulate JNK signaling pathway which functions as a key regulator of cellular apoptosis. Our expression data are in favor of suppression of JNK pathway, which could induce pro‑apoptotic gene expression as well as down regulation of survival factors, introducing this pathway as a key regulator of drug‑resistance development in AML. Acute myeloid leukemia (AML) is one of the most aggressive, life-threatening hematological malignancies char- acterized by uncontrolled proliferation of abnormal diferentiated and nonfunctional myeloid precursor cells1. -

WO 2017/015637 Al 26 January 2017 (26.01.2017) P O P C T
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2017/015637 Al 26 January 2017 (26.01.2017) P O P C T (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every A61K 48/00 (2006.01) C12N 15/63 (2006.01) kind of national protection available): AE, AG, AL, AM, C07K 19/00 (2006.01) C12N 15/65 (2006.01) AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, C12N 5/22 (2006.0 1) C12N 15/867 (2006.0 1) BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (21) International Application Number: HN, HR, HU, ID, IL, IN, IR, IS, JP, KE, KG, KN, KP, KR, PCT/US2016/043756 KZ, LA, LC, LK, LR, LS, LU, LY, MA, MD, ME, MG, (22) International Filing Date: MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, 22 July 2016 (22.07.2016) PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SA, SC, SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, (25) Filing Language: English TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (26) Publication Language: English (84) Designated States (unless otherwise indicated, for every (30) Priority Data: kind of regional protection available): ARIPO (BW, GH, 62/195,680 22 July 2015 (22.07.2015) US GM, KE, LR, LS, MW, MZ, NA, RW, SD, SL, ST, SZ, 62/293,3 13 9 February 2016 (09.02.2016) US TZ, UG, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, RU, TJ, TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, (71) Applicant: DUKE UNIVERSITY [US/US]; 2812 Erwin DK, EE, ES, FI, FR, GB, GR, HR, HU, IE, IS, IT, LT, LU, Road, Suite 306, Durham, NC 27705 (US). -

Fibroblast-Derived Induced Pluripotent Stem Cells Show No Common Retroviral Vector Insertions
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Harvard University - DASH Fibroblast-Derived Induced Pluripotent Stem Cells Show No Common Retroviral Vector Insertions The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. Citation Varas, Florencio, Matthias Stadtfeld, Luisa de Andres-Aguayo, Nimet Maherali, Alessandro di Tullio, Lorena Pantano, Cedric Notredame, et al. 2009. Fibroblast-Derived Induced Pluripotent Stem Cells Show No Common Retroviral Vector Insertions. Stem Cells 27(2): 300-306. Published Version doi:10.1634/stemcells.2008-0696 Accessed February 18, 2015 8:46:59 PM EST Citable Link http://nrs.harvard.edu/urn-3:HUL.InstRepos:4459933 Terms of Use This article was downloaded from Harvard University's DASH repository, and is made available under the terms and conditions applicable to Open Access Policy Articles, as set forth at http://nrs.harvard.edu/urn- 3:HUL.InstRepos:dash.current.terms-of-use#OAP (Article begins on next page) EMBRYONIC STEM CELLS/INDUCED PLURIPOTENT STEM CELLS Fibroblast-Derived Induced Pluripotent Stem Cells Show No Common Retroviral Vector Insertions FLORENCIO VARAS,a MATTHIAS STADTFELD,b LUISA DE ANDRES-AGUAYO,a NIMET MAHERALI,b ALESSANDRO DI TULLIO,a LORENA PANTANO,c CEDRIC NOTREDAME,c KONRAD HOCHEDLINGER,b THOMAS GRAFa,d aDifferentiation and Cancer and cBioinformatics Program, Center for Genomic Regulation and Pompeu Fabra University, Barcelona, Spain; bCancer Center and Center for Regenerative Medicine, Massachusetts General Hospital, Harvard Stem Cell Institute, Boston, Massachusetts, USA; dInstitucio´ Catalana de Recerca i Estudis Avanc¸ats Key Words. -

Identification of Genetic Modifiers of Age- At-Onset for Familial Parkinson’S Disease
Identification of genetic modifiers of age- at-onset for familial Parkinson’s disease The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation Hill-Burns, E. M., O. A. Ross, W. T. Wissemann, A. I. Soto-Ortolaza, S. Zareparsi, J. Siuda, T. Lynch, et al. 2016. “Identification of genetic modifiers of age-at-onset for familial Parkinson’s disease.” Human Molecular Genetics 25 (17): 3849-3862. doi:10.1093/hmg/ddw206. http://dx.doi.org/10.1093/hmg/ddw206. Published Version doi:10.1093/hmg/ddw206 Citable link http://nrs.harvard.edu/urn-3:HUL.InstRepos:30371016 Terms of Use This article was downloaded from Harvard University’s DASH repository, and is made available under the terms and conditions applicable to Other Posted Material, as set forth at http:// nrs.harvard.edu/urn-3:HUL.InstRepos:dash.current.terms-of- use#LAA Human Molecular Genetics, 2016, Vol. 25, No. 17 3849–3862 doi: 10.1093/hmg/ddw206 Advance Access Publication Date: 11 July 2016 Association Studies Article ASSOCIATION STUDIES ARTICLE Identification of genetic modifiers of age-at-onset for familial Parkinson’s disease Erin M. Hill-Burns1, Owen A. Ross2, William T. Wissemann1, Alexandra I. Soto-Ortolaza2, Sepideh Zareparsi3, Joanna Siuda4, Timothy Lynch5, Zbigniew K. Wszolek6, Peter A. Silburn7, George D. Mellick7, Beate Ritz8, Clemens R. Scherzer9, Cyrus P. Zabetian10, Stewart A. Factor11, Patrick J. Breheny12 and Haydeh Payami1,13,* 1Department of Neurology, University of Alabama at Birmingham, AL, USA, -

A Grainyhead-Like 2/Ovo-Like 2 Pathway Regulates Renal Epithelial Barrier Function and Lumen Expansion
BASIC RESEARCH www.jasn.org A Grainyhead-Like 2/Ovo-Like 2 Pathway Regulates Renal Epithelial Barrier Function and Lumen Expansion † ‡ | Annekatrin Aue,* Christian Hinze,* Katharina Walentin,* Janett Ruffert,* Yesim Yurtdas,*§ | Max Werth,* Wei Chen,* Anja Rabien,§ Ergin Kilic,¶ Jörg-Dieter Schulzke,** †‡ Michael Schumann,** and Kai M. Schmidt-Ott* *Max Delbrueck Center for Molecular Medicine, Berlin, Germany; †Experimental and Clinical Research Center, and Departments of ‡Nephrology, §Urology, ¶Pathology, and **Gastroenterology, Charité Medical University, Berlin, Germany; and |Berlin Institute of Urologic Research, Berlin, Germany ABSTRACT Grainyhead transcription factors control epithelial barriers, tissue morphogenesis, and differentiation, but their role in the kidney is poorly understood. Here, we report that nephric duct, ureteric bud, and collecting duct epithelia express high levels of grainyhead-like homolog 2 (Grhl2) and that nephric duct lumen expansion is defective in Grhl2-deficient mice. In collecting duct epithelial cells, Grhl2 inactivation impaired epithelial barrier formation and inhibited lumen expansion. Molecular analyses showed that GRHL2 acts as a transcrip- tional activator and strongly associates with histone H3 lysine 4 trimethylation. Integrating genome-wide GRHL2 binding as well as H3 lysine 4 trimethylation chromatin immunoprecipitation sequencing and gene expression data allowed us to derive a high-confidence GRHL2 target set. GRHL2 transactivated a group of genes including Ovol2, encoding the ovo-like 2 zinc finger transcription factor, as well as E-cadherin, claudin 4 (Cldn4), and the small GTPase Rab25. Ovol2 induction alone was sufficient to bypass the requirement of Grhl2 for E-cadherin, Cldn4,andRab25 expression. Re-expression of either Ovol2 or a combination of Cldn4 and Rab25 was sufficient to rescue lumen expansion and barrier formation in Grhl2-deficient collecting duct cells. -

UC San Diego Electronic Theses and Dissertations
UC San Diego UC San Diego Electronic Theses and Dissertations Title Cardiac Stretch-Induced Transcriptomic Changes are Axis-Dependent Permalink https://escholarship.org/uc/item/7m04f0b0 Author Buchholz, Kyle Stephen Publication Date 2016 Peer reviewed|Thesis/dissertation eScholarship.org Powered by the California Digital Library University of California UNIVERSITY OF CALIFORNIA, SAN DIEGO Cardiac Stretch-Induced Transcriptomic Changes are Axis-Dependent A dissertation submitted in partial satisfaction of the requirements for the degree Doctor of Philosophy in Bioengineering by Kyle Stephen Buchholz Committee in Charge: Professor Jeffrey Omens, Chair Professor Andrew McCulloch, Co-Chair Professor Ju Chen Professor Karen Christman Professor Robert Ross Professor Alexander Zambon 2016 Copyright Kyle Stephen Buchholz, 2016 All rights reserved Signature Page The Dissertation of Kyle Stephen Buchholz is approved and it is acceptable in quality and form for publication on microfilm and electronically: Co-Chair Chair University of California, San Diego 2016 iii Dedication To my beautiful wife, Rhia. iv Table of Contents Signature Page ................................................................................................................... iii Dedication .......................................................................................................................... iv Table of Contents ................................................................................................................ v List of Figures ................................................................................................................... -

Content Based Search in Gene Expression Databases and a Meta-Analysis of Host Responses to Infection
Content Based Search in Gene Expression Databases and a Meta-analysis of Host Responses to Infection A Thesis Submitted to the Faculty of Drexel University by Francis X. Bell in partial fulfillment of the requirements for the degree of Doctor of Philosophy November 2015 c Copyright 2015 Francis X. Bell. All Rights Reserved. ii Acknowledgments I would like to acknowledge and thank my advisor, Dr. Ahmet Sacan. Without his advice, support, and patience I would not have been able to accomplish all that I have. I would also like to thank my committee members and the Biomed Faculty that have guided me. I would like to give a special thanks for the members of the bioinformatics lab, in particular the members of the Sacan lab: Rehman Qureshi, Daisy Heng Yang, April Chunyu Zhao, and Yiqian Zhou. Thank you for creating a pleasant and friendly environment in the lab. I give the members of my family my sincerest gratitude for all that they have done for me. I cannot begin to repay my parents for their sacrifices. I am eternally grateful for everything they have done. The support of my sisters and their encouragement gave me the strength to persevere to the end. iii Table of Contents LIST OF TABLES.......................................................................... vii LIST OF FIGURES ........................................................................ xiv ABSTRACT ................................................................................ xvii 1. A BRIEF INTRODUCTION TO GENE EXPRESSION............................. 1 1.1 Central Dogma of Molecular Biology........................................... 1 1.1.1 Basic Transfers .......................................................... 1 1.1.2 Uncommon Transfers ................................................... 3 1.2 Gene Expression ................................................................. 4 1.2.1 Estimating Gene Expression ............................................ 4 1.2.2 DNA Microarrays ......................................................