A Thermodynamic Theory of Biological Evolution
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Emergy and Efficiency Analysis of Historical Bubbles
Emergy Synthesis 10, Proceedings of the 10th Biennial Emergy Conference (2019) 29 Emergy and Efficiency Analysis of Historical Bubbles Mark P. A. Ciotola ABSTRACT Human societies comprise energy-consuming beings, who are vast consumers of energy, as both individual organisms and organized societies. Hence, in principle, it should be possible to utilize thermodynamic approaches to generate models of historical phenomena. The existing energy-related efficiency-discounted exponential growth (EDEG) framework for historical resource bubbles and dynasties involving a nonrenewable resource is presented. As EDEG is presented, its relationship to emergy-related concepts and approaches are explored and analyzed. Finally, future research possibilities for expanding the physical, closed system framework behind EDEG into the open system emergy framework. CONTEXT A framework for a science of human history has been developed from a physics perspective utilizing no-renewable quantities and approximating historical dynasties as closed systems. However, it is important to also consider open systems and better constrain models with better physical data. The emergy approach offers both capabilities. This paper will explore emergy concepts as related to past work. INTRODUCTION Following the success of physical scientists such as Isaac Newton with the law of universal gravitation, it had been the dream of some social scientists to develop a science of society and history. Coming at the top of a wave of scientific discoveries, and excited also by the political and social changes in the European world around him, he [father of Sociology August Comte] conceived the idea, which was shared in varying degrees by other thinkers of his time, that science might be unified and used in such a way as to harmonize the discords of human life (Marvin, 1936). -

Framing Major Prebiotic Transitions As Stages of Protocell Development: Three Challenges for Origins-Of-Life Research
Framing major prebiotic transitions as stages of protocell development: three challenges for origins-of-life research Ben Shirt-Ediss1, Sara Murillo-Sánchez2,3 and Kepa Ruiz-Mirazo*2,3 Commentary Open Access Address: Beilstein J. Org. Chem. 2017, 13, 1388–1395. 1Interdisciplinary Computing and Complex BioSystems Group, doi:10.3762/bjoc.13.135 University of Newcastle, UK, 2Dept. Logic and Philosophy of Science, University of the Basque Country, Spain and 3Biofisika Institute Received: 16 February 2017 (CSIC, UPV-EHU), Spain Accepted: 27 June 2017 Published: 13 July 2017 Email: Kepa Ruiz-Mirazo* - [email protected] This article is part of the Thematic Series "From prebiotic chemistry to molecular evolution". * Corresponding author Guest Editor: L. Cronin Keywords: functional integration; origins of life; prebiotic evolution; protocells © 2017 Shirt-Ediss et al.; licensee Beilstein-Institut. License and terms: see end of document. Abstract Conceiving the process of biogenesis as the evolutionary development of highly dynamic and integrated protocell populations provides the most appropriate framework to address the difficult problem of how prebiotic chemistry bridged the gap to full-fledged living organisms on the early Earth. In this contribution we briefly discuss the implications of taking dynamic, functionally inte- grated protocell systems (rather than complex reaction networks in bulk solution, sets of artificially evolvable replicating molecules, or even these same replicating molecules encapsulated in passive compartments) -

406 | Tutto Arabi - Veterinary Biorhythm
406 | TUTTO ARABI - www.tuttoarabi.com VETERINARY BIORHYTHM by Domenico Bergero, Cynthia Préfontaine Rhythm! • infradian rhythms, which are long-term cycles, such as the To horses, as for men, the passing of time has an impact on annual migration or reproduction cycles found in certain many conscious or automatic activities. When it comes to animals or the human menstrual cycle. men, in particular athletes, we talk about “biorhythm”, i.e. the • ultradian rhythms, which are short cycles, such as the 90-minute effect that longer or shorter cycles can have on performance. REM cycle, the 4-hour nasal cycle, or the 3-hour cycle of But what about our adventure buddy? growth hormone production. They have periods of less than 24 hours. Chronobiology • tidal rhythms, commonly observed in marine life, which follow Chronobiology, from the Greek “chronos” (time) and the (roughly) 12-hour transition from high to low tide “biologia” (study of life), is a field of science that examines and back. periodic (cyclic) phenomena in living organisms and their adaptation to solar and lunar related rhythms. Hormones and physical performance Chronobiological studies include but are not limited to As everyone knows, physical performance is, among other comparative anatomy, physiology, genetics, molecular things, under the control of hormones that determine higher biology and behavior of organisms within biological rhythms or lesser fitness. These hormones can be produced continually mechanics. or, more often than not, can be mero present at certain The variations of the timing and duration of biological times (peak or acrophase) and then decrease. Let’s see which activity in living organisms occur for many essential hormones are involved and what are the best moments for biological processes. -

Toward Quantifying the Climate Heat Engine: Solar Absorption and Terrestrial Emission Temperatures and Material Entropy Production
JUNE 2017 B A N N O N A N D L E E 1721 Toward Quantifying the Climate Heat Engine: Solar Absorption and Terrestrial Emission Temperatures and Material Entropy Production PETER R. BANNON AND SUKYOUNG LEE Department of Meteorology and Atmospheric Science, The Pennsylvania State University, University Park, Pennsylvania (Manuscript received 15 August 2016, in final form 22 February 2017) ABSTRACT A heat-engine analysis of a climate system requires the determination of the solar absorption temperature and the terrestrial emission temperature. These temperatures are entropically defined as the ratio of the energy exchanged to the entropy produced. The emission temperature, shown here to be greater than or equal to the effective emission temperature, is relatively well known. In contrast, the absorption temperature re- quires radiative transfer calculations for its determination and is poorly known. The maximum material (i.e., nonradiative) entropy production of a planet’s steady-state climate system is a function of the absorption and emission temperatures. Because a climate system does no work, the material entropy production measures the system’s activity. The sensitivity of this production to changes in the emission and absorption temperatures is quantified. If Earth’s albedo does not change, material entropy production would increase by about 5% per 1-K increase in absorption temperature. If the absorption temperature does not change, entropy production would decrease by about 4% for a 1% decrease in albedo. It is shown that, as a planet’s emission temperature becomes more uniform, its entropy production tends to increase. Conversely, as a planet’s absorption temperature or albedo becomes more uniform, its entropy production tends to decrease. -

Chapter 9. NATURAL SELECTION and BIOLOGICAL EVOLUTION
Chapter 9. NATURAL SELECTION AND BIOLOGICAL EVOLUTION Which beginning of time [the Creation] according to our Chronologie, fell upon the entrance of the night preced- ing the twenty third day of Octob. in the year of the Julian Calendar, 710 [i.e. B.C. 4004]. Archbishop James Ussher (1581—1656) The Annals of the World1 (1658:1) “How stupid not to have thought of that!” Thomas Huxley (1825-1895), about Darwin’s theory of Evolution by Nat- ural Selection. I. Introduction A. Classical Discoveries of Biology From the mid 18th Century to the early part of the 20th, a large fraction of biologists’ efforts went into two massive collective discoveries, the discovery of biotic diversity, and the discovery of evolution. Over the period from 1750 to 1950 the careful descriptive analyses of Swedish biol- ogist Karl von Linné and his followers showed there to be on the order of 10 million or so different types of living organisms. The differences in biotas in different parts of the world came to be appreciated, the amazing diversity of the tropics was documented, and previ- ously unimagined major groups of organisms were discovered, including microorganisms and the biota of odd habitats like the oceanic plankton. The sometimes bizarre and always impressive adaptation of organisms to their habitats and ways of life greatly impressed the early scientific naturalists, who argued that it proved the existence of an All Seeing Design- er. Similarly, paleontologists described a huge variety of fossil plants and animals. The succession of forms, and the presence of many detailed structural similarities between liv- ing and extinct forms, as well as structural parallels between living forms, strongly suggest- ed that living and ancient forms of life were connected by branching lines of descent. -

Uniting Micro- with Macroevolution Into an Extended Synthesis: Reintegrating Life’S Natural History Into Evolution Studies
Uniting Micro- with Macroevolution into an Extended Synthesis: Reintegrating Life’s Natural History into Evolution Studies Nathalie Gontier Abstract The Modern Synthesis explains the evolution of life at a mesolevel by identifying phenotype–environmental interactions as the locus of evolution and by identifying natural selection as the means by which evolution occurs. Both micro- and macroevolutionary schools of thought are post-synthetic attempts to evolution- ize phenomena above and below organisms that have traditionally been conceived as non-living. Microevolutionary thought associates with the study of how genetic selection explains higher-order phenomena such as speciation and extinction, while macroevolutionary research fields understand species and higher taxa as biological individuals and they attribute evolutionary causation to biotic and abiotic factors that transcend genetic selection. The microreductionist and macroholistic research schools are characterized as two distinct epistemic cultures where the former favor mechanical explanations, while the latter favor historical explanations of the evolu- tionary process by identifying recurring patterns and trends in the evolution of life. I demonstrate that both cultures endorse radically different notions on time and explain how both perspectives can be unified by endorsing epistemic pluralism. Keywords Microevolution · Macroevolution · Origin of life · Evolutionary biology · Sociocultural evolution · Natural history · Organicism · Biorealities · Units, levels and mechanisms of evolution · Major transitions · Hierarchy theory But how … shall we describe a process which nobody has seen performed, and of which no written history gives any account? This is only to be investigated, first, in examining the nature of those solid bodies, the history of which we want to know; and 2dly, in exam- ining the natural operations of the globe, in order to see if there now actually exist such operations, as, from the nature of the solid bodies, appear to have been necessary to their formation. -

Appendix a Rate of Entropy Production and Kinetic Implications
Appendix A Rate of Entropy Production and Kinetic Implications According to the second law of thermodynamics, the entropy of an isolated system can never decrease on a macroscopic scale; it must remain constant when equilib- rium is achieved or increase due to spontaneous processes within the system. The ramifications of entropy production constitute the subject of irreversible thermody- namics. A number of interesting kinetic relations, which are beyond the domain of classical thermodynamics, can be derived by considering the rate of entropy pro- duction in an isolated system. The rate and mechanism of evolution of a system is often of major or of even greater interest in many problems than its equilibrium state, especially in the study of natural processes. The purpose of this Appendix is to develop some of the kinetic relations relating to chemical reaction, diffusion and heat conduction from consideration of the entropy production due to spontaneous processes in an isolated system. A.1 Rate of Entropy Production: Conjugate Flux and Force in Irreversible Processes In order to formally treat an irreversible process within the framework of thermo- dynamics, it is important to identify the force that is conjugate to the observed flux. To this end, let us start with the question: what is the force that drives heat flux by conduction (or heat diffusion) along a temperature gradient? The intuitive answer is: temperature gradient. But the answer is not entirely correct. To find out the exact force that drives diffusive heat flux, let us consider an isolated composite system that is divided into two subsystems, I and II, by a rigid diathermal wall, the subsystems being maintained at different but uniform temperatures of TI and TII, respectively. -

Thermodynamics of Manufacturing Processes—The Workpiece and the Machinery
inventions Article Thermodynamics of Manufacturing Processes—The Workpiece and the Machinery Jude A. Osara Mechanical Engineering Department, University of Texas at Austin, EnHeGi Research and Engineering, Austin, TX 78712, USA; [email protected] Received: 27 March 2019; Accepted: 10 May 2019; Published: 15 May 2019 Abstract: Considered the world’s largest industry, manufacturing transforms billions of raw materials into useful products. Like all real processes and systems, manufacturing processes and equipment are subject to the first and second laws of thermodynamics and can be modeled via thermodynamic formulations. This article presents a simple thermodynamic model of a manufacturing sub-process or task, assuming multiple tasks make up the entire process. For example, to manufacture a machined component such as an aluminum gear, tasks include cutting the original shaft into gear blanks of desired dimensions, machining the gear teeth, surfacing, etc. The formulations presented here, assessing the workpiece and the machinery via entropy generation, apply to hand-crafting. However, consistent isolation and measurement of human energy changes due to food intake and work output alone pose a significant challenge; hence, this discussion focuses on standardized product-forming processes typically via machine fabrication. Keywords: thermodynamics; manufacturing; product formation; entropy; Helmholtz energy; irreversibility 1. Introduction Industrial processes—manufacturing or servicing—involve one or more forms of electrical, mechanical, chemical (including nuclear), and thermal energy conversion processes. For a manufactured component, an interpretation of the first law of thermodynamics indicates that the internal energy content of the component is the energy that formed the product [1]. Cursorily, this sums all the work that goes into the manufacturing process from electrical to mechanical, chemical, and thermal power consumption by the manufacturing equipment. -

Reproduction of a Protocell by Replication of Minority Molecule in Catalytic Reaction Network
APS/123-QED Reproduction of a Protocell by Replication of Minority Molecule in Catalytic Reaction Network Atsushi Kamimura1 and Kunihiko Kaneko2, 3 1Department of Applied Physics, Graduate School of Engineering, The University of Tokyo, 7-3-1, Hongo, Bunkyo-ku, Tokyo 113-8656, Japan. 2Department of Basic Science, Graduate School of Arts and Sciences, The University of Tokyo, 3-8-1, Komaba, Meguro-ku, Tokyo 153-8902, Japan 3Complex Systems Biology Project, ERATO, JST, Tokyo, Japan. (Dated: October 31, 2018) Abstract For understanding the origin of life, it is essential to explain the development of a compartmen- talized structure, which undergoes growth and division, from a set of chemical reactions. In this study, a hypercycle with two chemicals that mutually catalyze each other is considered in order to show that the reproduction of a protocell with a growth-division process naturally occurs when the replication speed of one chemical is considerably slower than that of the other chemical. It is observed that the protocell divides after a minority molecule is replicated at a slow synthesis rate, and thus, a synchrony between the reproduction of a cell and molecule replication is achieved. The robustness of such protocells against the invasion of parasitic molecules is also demonstrated. arXiv:1005.1142v1 [q-bio.CB] 7 May 2010 PACS numbers: 87.17.-d, 05.40.-a, 82.39.-k 1 Filling a gap between just a set of catalytic reactions and a reproducing cell is a difficult problem; however, it is essential to fully understand this relationship to unveil the origin of life, to establish a theory for a living state, and to experimentally synthesize protocells[1]. -

Latitudinal Cline of Chronotype
www.nature.com/scientificreports OPEN Latitudinal cline of chronotype Mario André Leocadio-Miguel 1, Fernando Mazzili Louzada2, Leandro Lourenção Duarte3, Roberta Peixoto Areas4, Marilene Alam5, Marcelo Ventura Freire4, John Fontenele-Araujo1, Luiz Menna-Barreto4 & Mario Pedrazzoli4 The rotation of the Earth around its own axis and around the sun determines the characteristics of Received: 15 February 2017 the light/dark cycle, the most stable and ancient 24 h temporal cue for all organisms. Due to the tilt in the earth’s axis in relation to the plane of the earth’s orbit around the sun, sunlight reaches the Earth Accepted: 2 June 2017 differentially depending on the latitude. The timing of circadian rhythms varies among individuals of Published: xx xx xxxx a given population and biological and environmental factors underlie this variability. In the present study, we tested the hypothesis that latitude is associated to the regulation of circadian rhythm in humans. We have studied chronotype profiles across latitudinal cline from around 0° to 32° South in Brazil in a sample of 12,884 volunteers living in the same time zone. The analysis of the results revealed that humans are sensitive to the different sunlight signals tied to differences in latitude, resulting in a morning to evening latitudinal cline of chronotypes towards higher latitudes. The concept of chronotype, which is the expression of diurnal preferences or circadian phenotype, including rise and bedtime preferences, has received a strong consideration from studies in human chronobiology1. It is generally accepted that the chronotype distribution in populations is the same, no matter where the population is geographically localized. -
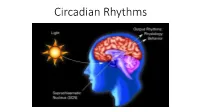
Circadian Rhythms Controlling the Timing of Behaviour by Anticipating the Environment
Circadian Rhythms Controlling the timing of behaviour by anticipating the environment • Circadian = circa + dium • Exists in most if not all unicellular and multicellular organisms The Circadian Circuit Environmental Output Inputs Rhythms Light Hormonal Cycles Temperature Rest/Wake Social Activity Feeding Central Pacemaker Health consequences of circadian misalignment Social Jet Lag Shift Work Increased risk of: - Obesity - Diabetes - Cancer - Mental Illness Light at Night Roenneberg et al. (2012) Current Biology Historical Perspective Jean-Jacques d’Ortous de Mairan (1678 – 1771) Hist de l’Acad Royal Sci (Paris), 1729 “…Il est seulement un peu moins marqué lorsqu’on la tient toujours enfermée dans un lieu obscur…” “The sensitive plant hence perceives the sun without seeing it” Rhythms in leaf-opening persist even in the absence of sunlight Historical Perspective Rat Comp Psychol Monographs, 1922 Nathaniel Kleitman (1895 – 1999) Figure 18.4 Sleep and Wakefulness, 1963 Historical Perspective ’Founders of Chronobiology’ 1960 • Conceptual framework of circadian rhythms • Long before any genes or neural circuits were identified Colin Pittendrigh Jürgen Aschoff (1918 – 1996) (1913 – 1998) Cold Spring Harbor Symposium on Quantitative Biology, Vol. XXV Biological Clocks The Circadian Circuit Environmental Output Inputs Rhythms Light Hormonal Cycles Temperature Rest/Wake Social Activity Feeding Central Pacemaker What would a circadian pacemaker look like? The Circadian Circuit Environmental Output Inputs Rhythms Light Hormonal Cycles Temperature -

Pittendrigh (1918-1996): in Fond Memory
ARTICLE-IH-A-BOX Pittendrigh (1918-1996): In Fond Memory The scientific study of biological rhythms (chronobiology) has really had a many faceted and colourful past in Europe and drew the attention of brilliant naturalists in the 18th, 19th and 20th centuries and is turning out to be one of the most interdiscipli nary branches of modem biology in the 21st century. The conceptual foundations of chronobiology in the 20th century were laid by Jurgen Aschoff, Erwin Blinning and Colin Pittendrigh. Their deep insight and contributions in the second half of that century account for the high degree of scientific elegance and experimental rigour of the subject. In fact, it was the symposium on Biological Clocks convened by Pittendrigh in the Cold Spring Harbor in New York in 1960, with Bunning as the chairperson and the seminal papers of Aschoff and Pittendrigh that ushered in an era of intensified experimental activity. Bunning's address was a definitive version of the history of chronobiology. By 1997, researchers began talking ofa 'party time' for chronobiology since it marked the twenty-fifth anniversary of the discovery of the supra chiasmatic nucleus (SeN) in the brain of mammals, which is the pacemaker of circadian organization, and the identification and cloning of the first circadian clock gene in a mammal. The journal Science, in a December 1998 issue, ranked some of the findings in the field of biological clocks as the first runner-up breakthrough-of-the-year. By 2003 one of our fraternity could write: "The successes achieved by chronobiologists over the past decade have been the envy of the scientific community.