Downloaded As an Aligned Fasta File Including Gaps
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
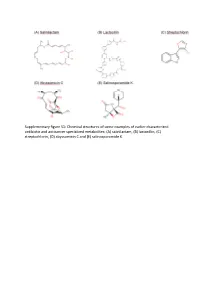
Chemical Structures of Some Examples of Earlier Characterized Antibiotic and Anticancer Specialized
Supplementary figure S1: Chemical structures of some examples of earlier characterized antibiotic and anticancer specialized metabolites: (A) salinilactam, (B) lactocillin, (C) streptochlorin, (D) abyssomicin C and (E) salinosporamide K. Figure S2. Heat map representing hierarchical classification of the SMGCs detected in all the metagenomes in the dataset. Table S1: The sampling locations of each of the sites in the dataset. Sample Sample Bio-project Site depth accession accession Samples Latitude Longitude Site description (m) number in SRA number in SRA AT0050m01B1-4C1 SRS598124 PRJNA193416 Atlantis II water column 50, 200, Water column AT0200m01C1-4D1 SRS598125 21°36'19.0" 38°12'09.0 700 and above the brine N "E (ATII 50, ATII 200, 1500 pool water layers AT0700m01C1-3D1 SRS598128 ATII 700, ATII 1500) AT1500m01B1-3C1 SRS598129 ATBRUCL SRS1029632 PRJNA193416 Atlantis II brine 21°36'19.0" 38°12'09.0 1996– Brine pool water ATBRLCL1-3 SRS1029579 (ATII UCL, ATII INF, N "E 2025 layers ATII LCL) ATBRINP SRS481323 PRJNA219363 ATIID-1a SRS1120041 PRJNA299097 ATIID-1b SRS1120130 ATIID-2 SRS1120133 2168 + Sea sediments Atlantis II - sediments 21°36'19.0" 38°12'09.0 ~3.5 core underlying ATII ATIID-3 SRS1120134 (ATII SDM) N "E length brine pool ATIID-4 SRS1120135 ATIID-5 SRS1120142 ATIID-6 SRS1120143 Discovery Deep brine DDBRINP SRS481325 PRJNA219363 21°17'11.0" 38°17'14.0 2026– Brine pool water N "E 2042 layers (DD INF, DD BR) DDBRINE DD-1 SRS1120158 PRJNA299097 DD-2 SRS1120203 DD-3 SRS1120205 Discovery Deep 2180 + Sea sediments sediments 21°17'11.0" -

The 2014 Golden Gate National Parks Bioblitz - Data Management and the Event Species List Achieving a Quality Dataset from a Large Scale Event
National Park Service U.S. Department of the Interior Natural Resource Stewardship and Science The 2014 Golden Gate National Parks BioBlitz - Data Management and the Event Species List Achieving a Quality Dataset from a Large Scale Event Natural Resource Report NPS/GOGA/NRR—2016/1147 ON THIS PAGE Photograph of BioBlitz participants conducting data entry into iNaturalist. Photograph courtesy of the National Park Service. ON THE COVER Photograph of BioBlitz participants collecting aquatic species data in the Presidio of San Francisco. Photograph courtesy of National Park Service. The 2014 Golden Gate National Parks BioBlitz - Data Management and the Event Species List Achieving a Quality Dataset from a Large Scale Event Natural Resource Report NPS/GOGA/NRR—2016/1147 Elizabeth Edson1, Michelle O’Herron1, Alison Forrestel2, Daniel George3 1Golden Gate Parks Conservancy Building 201 Fort Mason San Francisco, CA 94129 2National Park Service. Golden Gate National Recreation Area Fort Cronkhite, Bldg. 1061 Sausalito, CA 94965 3National Park Service. San Francisco Bay Area Network Inventory & Monitoring Program Manager Fort Cronkhite, Bldg. 1063 Sausalito, CA 94965 March 2016 U.S. Department of the Interior National Park Service Natural Resource Stewardship and Science Fort Collins, Colorado The National Park Service, Natural Resource Stewardship and Science office in Fort Collins, Colorado, publishes a range of reports that address natural resource topics. These reports are of interest and applicability to a broad audience in the National Park Service and others in natural resource management, including scientists, conservation and environmental constituencies, and the public. The Natural Resource Report Series is used to disseminate comprehensive information and analysis about natural resources and related topics concerning lands managed by the National Park Service. -

Download Full Article As
Article Genomic Signature Analysis for the Strategic Bioremediation of Polycyclic Aromatic Hydrocarbons in Mangrove Ecosystems in the Gulf of Tonkin Nam H. Dao1, Quan M. Le2, Thang X. Dao3 1The Hill School, USA 2The Loomis Chaffee School, USA 3Unaffiliated SUMMARY processes. Once oil has entered the roots and rhizosphere Current oil spill cleanup methods can be inefficient, (the soil directly influenced by plant roots) of mangrove costly, and have many more downsides. In an effort to trees, they fail to participate in regular salt management and create an efficient, cost-effective, and environmentally- respiration (3). As mangrove trees are responsible for various friendly solution to cleaning oil spills, there has been important functions in this habitat such as protection of soil a flurry of advancements in microbiological vectors. These plasmids confer oil-degrading genes that can be from erosion, improving water quality or helping provide implemented into the native microbial population. For nutrients for microbial communities, the death or failure of this strategy to work, it is critical that an appropriate these trees to function can pose heavy consequences for naturally-found plasmid be picked as a base to local organisms (3). Upon direct contamination by an oil spill, ensure long-term plasmid survival and activation of mangrove trees usually face death within several months, and its genes. To help realize this goal, this study relies other organisms living in the area that depend on a host of on emerging computational technologies to measure benefits provided by mangrove trees are subsequently either genetic similarity between the host chromosome and impacted directly by the influx of oil or indirectly through the the potential plasmid vector, utilizing an algorithm for loss of mangrove trees (3). -

First Genomic Insights Into Members of a Candidate Bacterial Phylum Responsible for Wastewater Bulking
First genomic insights into members of a candidate bacterial phylum responsible for wastewater bulking Yuji Sekiguchi1, Akiko Ohashi1, Donovan H. Parks2, Toshihiro Yamauchi3, Gene W. Tyson2,4 and Philip Hugenholtz2,5 1 Biomedical Research Institute, National Institute of Advanced Industrial Science and Technology (AIST), Tsukuba, Ibaraki, Japan 2 Australian Centre for Ecogenomics, School of Chemistry and Molecular Biosciences, The University of Queensland, St. Lucia, Queensland, Australia 3 Administrative Management Department, Kubota Kasui Corporation, Minato-ku, Tokyo, Japan 4 Advanced Water Management Centre, The University of Queensland, St. Lucia, Queensland, Australia 5 Institute for Molecular Bioscience, The University of Queensland, St. Lucia, Queensland, Australia ABSTRACT Filamentous cells belonging to the candidate bacterial phylum KSB3 were previously identified as the causative agent of fatal filament overgrowth (bulking) in a high-rate industrial anaerobic wastewater treatment bioreactor. Here, we obtained near complete genomes from two KSB3 populations in the bioreactor, including the dominant bulking filament, using diVerential coverage binning of metagenomic data. Fluorescence in situ hybridization with 16S rRNA-targeted probes specific for the two populations confirmed that both are filamentous organisms. Genome-based metabolic reconstruction and microscopic observation of the KSB3 filaments in the presence of sugar gradients indicate that both filament types are Gram-negative, strictly anaerobic fermenters capable of -

Revealing the Full Biosphere Structure and Versatile Metabolic Functions In
Chen et al. Genome Biology (2021) 22:207 https://doi.org/10.1186/s13059-021-02408-w RESEARCH Open Access Revealing the full biosphere structure and versatile metabolic functions in the deepest ocean sediment of the Challenger Deep Ping Chen1†, Hui Zhou1,2†, Yanyan Huang3,4†, Zhe Xie5†, Mengjie Zhang1,2†, Yuli Wei5, Jia Li1,2, Yuewei Ma3, Min Luo5, Wenmian Ding3, Junwei Cao5, Tao Jiang1,2, Peng Nan3*, Jiasong Fang5* and Xuan Li1,2* * Correspondence: nanpeng@fudan. edu.cn; [email protected]; lixuan@ Abstract sippe.ac.cn †Ping Chen, Hui Zhou, Yanyan Background: The full biosphere structure and functional exploration of the microbial Huang, Zhe Xie and Mengjie Zhang communities of the Challenger Deep of the Mariana Trench, the deepest known contributed equally to this work. hadal zone on Earth, lag far behind that of other marine realms. 3Ministry of Education Key Laboratory for Biodiversity Science Results: We adopt a deep metagenomics approach to investigate the microbiome and Ecological Engineering, School in the sediment of Challenger Deep, Mariana Trench. We construct 178 of Life Sciences, Fudan University, Shanghai, China metagenome-assembled genomes (MAGs) representing 26 phyla, 16 of which are 5Shanghai Engineering Research reported from hadal sediment for the first time. Based on the MAGs, we find the Center of Hadal Science and microbial community functions are marked by enrichment and prevalence of Technology, College of Marine Sciences, Shanghai Ocean mixotrophy and facultative anaerobic metabolism. The microeukaryotic community is University, Shanghai, China found to be dominated by six fungal groups that are characterized for the first time 1CAS-Key Laboratory of Synthetic in hadal sediment to possess the assimilatory and dissimilatory nitrate/sulfate Biology, CAS Center for Excellence in Molecular Plant Sciences, Institute reduction, and hydrogen sulfide oxidation pathways. -

Draft Genome Sequence of Thalassobius Mediterraneus CECT 5383T, a Poly-Beta-Hydroxybutyrate Producer
Genomics Data 7 (2016) 237–239 Contents lists available at ScienceDirect Genomics Data journal homepage: www.elsevier.com/locate/gdata Data in Brief Draft genome sequence of Thalassobius mediterraneus CECT 5383T, a poly-beta-hydroxybutyrate producer Lidia Rodrigo-Torres, María J. Pujalte, David R. Arahal ⁎ Departamento de Microbiología y Ecología and Colección Española de Cultivos Tipo (CECT), Universidad de Valencia, Valencia, Spain article info abstract Article history: Thalassobius mediterraneus is the type species of the genus Thalassobius and a member of the Roseobacter clade, an Received 23 December 2015 abundant representative of marine bacteria. T. mediterraneus XSM19T (=CECT 5383T) was isolated from the Received in revised form 8 January 2016 Western Mediterranean coast near Valencia (Spain) in 1989. We present here the draft genome sequence and Accepted 14 January 2016 annotation of this strain (ENA/DDBJ/NCBI accession number CYSF00000000), which is comprised of Available online 15 January 2016 3,431,658 bp distributed in 19 contigs and encodes 10 rRNA genes, 51 tRNA genes and 3276 protein coding fi Keywords: genes. Relevant ndings are commented, including the complete set of genes required for poly-beta- Rhodobacteraceae hydroxybutyrate (PHB) synthesis and genes related to degradation of aromatic compounds. Roseobacter clade © 2016 The Authors. Published by Elsevier Inc. This is an open access article under the CC BY-NC-ND license PHB (http://creativecommons.org/licenses/by-nc-nd/4.0/). Aromatic compounds 1. Direct link to deposited data has not been yet validated, and even more recently T. abysii has been Specifications also proposed [6]. Organism/cell line/tissue Thalassobius mediterraneus Strain CECT 5383T Sequencer or array type Illumina MiSeq 2. -

Habitat and Taxon As Driving Forces of Carbohydrate
Habitat and taxon as driving forces of carbohydrate catabolism in marine heterotrophic bacteria: example of the model algae-associated bacterium Zobellia galactanivorans Dsij T Tristan Barbeyron, François Thomas, Valérie Barbe, Hanno Teeling, Chantal Schenowitz, Carole Dossat, Alexander Goesmann, Catherine Leblanc, Frank Oliver Glöckner, Mirjam Czjzek, et al. To cite this version: Tristan Barbeyron, François Thomas, Valérie Barbe, Hanno Teeling, Chantal Schenowitz, et al.. Habi- tat and taxon as driving forces of carbohydrate catabolism in marine heterotrophic bacteria: example of the model algae-associated bacterium Zobellia galactanivorans Dsij T. Environmental Microbiol- ogy, Society for Applied Microbiology and Wiley-Blackwell, 2016, Ecology and Physiology of Marine Microbes, 18 (12), pp.4610-4627. 10.1111/1462-2920.13584. hal-02137896 HAL Id: hal-02137896 https://hal.archives-ouvertes.fr/hal-02137896 Submitted on 23 May 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. 1 Environmental Microbiology ‐ Research Article 2 3 Habitat and taxon -

Bismis-2016 Abstract Book
BISMiS-2016 Abstract Book Third Meeting of Bergey's International Society for Microbial Systematics on Microbial Systematics and Metagenomics September 12-15, 2016 | Pune, INDIA PUNE UNIT Abstracts - Opening Address - Keynotes Abstract Book | BISMiS-2016 | Pune, India Opening Address TAXONOMY OF PROKARYOTES - NEW CHALLENGES IN A GLOBAL WORLD Peter Kämpfer* Justus-Liebig-University Giessen, HESSEN, Germany Email: [email protected] Systematics can be considered as a comprehensive science, because in science it is an essential aspect in comparing any two or more elements, whether they are genes or genomes, proteins or proteomes, biochemical pathways or metabolomes (just to list a few examples), or whole organisms. The development of high throughput sequencing techniques has led to an enormous amount of data (genomic and other “omic” data) and has also revealed an extensive diversity behind these data. These data are more and more used also in systematics and there is a strong trend to classify and name the taxonomic units in prokaryotic systematics preferably on the basis of sequence data. Unfortunately, the knowledge of the meaning behind the sequence data does not keep up with the tremendous increase of generated sequences. The extent of the accessory genome in any given cell, and perhaps the infinite extent of the pan-genome (as an aggregate of all the accessory genomes) is fascinating but it is an open question if and how these data should be used in systematics. Traditionally the polyphasic approach in bacterial systematics considers methods including both phenotype and genotype. And it is the phenotype that is (also) playing an essential role in driving the evolution. -

Horizontal Operon Transfer, Plasmids, and the Evolution of Photosynthesis in Rhodobacteraceae
The ISME Journal (2018) 12:1994–2010 https://doi.org/10.1038/s41396-018-0150-9 ARTICLE Horizontal operon transfer, plasmids, and the evolution of photosynthesis in Rhodobacteraceae 1 2 3 4 1 Henner Brinkmann ● Markus Göker ● Michal Koblížek ● Irene Wagner-Döbler ● Jörn Petersen Received: 30 January 2018 / Revised: 23 April 2018 / Accepted: 26 April 2018 / Published online: 24 May 2018 © The Author(s) 2018. This article is published with open access Abstract The capacity for anoxygenic photosynthesis is scattered throughout the phylogeny of the Proteobacteria. Their photosynthesis genes are typically located in a so-called photosynthesis gene cluster (PGC). It is unclear (i) whether phototrophy is an ancestral trait that was frequently lost or (ii) whether it was acquired later by horizontal gene transfer. We investigated the evolution of phototrophy in 105 genome-sequenced Rhodobacteraceae and provide the first unequivocal evidence for the horizontal transfer of the PGC. The 33 concatenated core genes of the PGC formed a robust phylogenetic tree and the comparison with single-gene trees demonstrated the dominance of joint evolution. The PGC tree is, however, largely incongruent with the species tree and at least seven transfers of the PGC are required to reconcile both phylogenies. 1234567890();,: 1234567890();,: The origin of a derived branch containing the PGC of the model organism Rhodobacter capsulatus correlates with a diagnostic gene replacement of pufC by pufX. The PGC is located on plasmids in six of the analyzed genomes and its DnaA- like replication module was discovered at a conserved central position of the PGC. A scenario of plasmid-borne horizontal transfer of the PGC and its reintegration into the chromosome could explain the current distribution of phototrophy in Rhodobacteraceae. -

Bacillus Pumilus Group Comparative Genomics: Toward Pangenome Features, Diversity, and Marine Environmental Adaptation
fmicb-12-571212 May 7, 2021 Time: 11:31 # 1 ORIGINAL RESEARCH published: 07 May 2021 doi: 10.3389/fmicb.2021.571212 Bacillus pumilus Group Comparative Genomics: Toward Pangenome Features, Diversity, and Marine Environmental Adaptation Xiaoteng Fu1,2,3, Linfeng Gong1,2,3, Yang Liu5, Qiliang Lai1,2,3, Guangyu Li1,2,3 and Zongze Shao1,2,3,4* 1 Key Laboratory of Marine Genetic Resources, Third Institute of Oceanography, Ministry of Natural Resources, Xiamen, China, 2 State Key Laboratory Breeding Base of Marine Genetic Resources, Xiamen, China, 3 Key Laboratory of Marine Genetic Resources of Fujian Province, Xiamen, China, 4 Southern Marine Science and Engineering Guangdong Laboratory, Zhuhai, China, 5 State Key Laboratory of Applied Microbiology Southern China, Guangdong Provincial Key Laboratory of Microbial Culture Collection and Application, Guangdong Open Laboratory of Applied Microbiology, Guangdong Microbial Culture Collection Center (GDMCC), Guangdong Institute of Microbiology, Guangdong Academy of Sciences, Guangzhou, China Edited by: John R. Battista, Background: Members of the Bacillus pumilus group (abbreviated as the Bp group) are Louisiana State University, United States quite diverse and ubiquitous in marine environments, but little is known about correlation Reviewed by: with their terrestrial counterparts. In this study, 16 marine strains that we had isolated Martín Espariz, before were sequenced and comparative genome analyses were performed with a Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), total of 52 Bp group strains. The analyses included 20 marine isolates (which included Instituto de Biología Molecular y the 16 new strains) and 32 terrestrial isolates, and their evolutionary relationships, Celular de Rosario (IBR), Argentina differentiation, and environmental adaptation. -

Systema Naturae 2000 (Phylum, 6 Nov 2017)
The Taxonomicon Systema Naturae 2000 Classification of Domain Bacteria (prokaryotes) down to Phylum Compiled by Drs. S.J. Brands Universal Taxonomic Services 6 Nov 2017 Systema Naturae 2000 - Domain Bacteria - Domain Bacteria Woese et al. 1990 1 Genus †Eoleptonema Schopf 1983, incertae sedis 2 Genus †Primaevifilum Schopf 1983, incertae sedis 3 Genus †Archaeotrichion Schopf 1968, incertae sedis 4 Genus †Siphonophycus Schopf 1968, incertae sedis 5 Genus Bactoderma Tepper and Korshunova 1973 (Approved Lists 1980), incertae sedis 6 Genus Stibiobacter Lyalikova 1974 (Approved Lists 1980), incertae sedis 7.1.1.1.1.1 Superphylum "Proteobacteria" Craig et al. 2010 1.1 Phylum "Alphaproteobacteria" 1.2.1 Phylum "Acidithiobacillia" 1.2.2.1 Phylum "Gammaproteobacteria" 1.2.2.2.1 Candidate phylum Muproteobacteria (RIF23) Anantharaman et al. 2016 1.2.2.2.2 Phylum "Betaproteobacteria" 2 Phylum "Zetaproteobacteria" 7.1.1.1.1.2 Phylum "Deltaproteobacteria_1" 7.1.1.1.2.1.1.1 Phylum "Deltaproteobacteria" [polyphyletic] 7.1.1.1.2.1.1.2.1 Phylum "Deltaproteobacteria_2" 7.1.1.1.2.1.1.2.2 Phylum "Deltaproteobacteria_3" 7.1.1.1.2.1.2 Candidate phylum Dadabacteria (CSP1-2) Hug et al. 2015 7.1.1.1.2.2.1 Candidate phylum "MBNT15" 7.1.1.1.2.2.2 Candidate phylum "Uncultured Bacterial Phylum 10 (UBP10)" Parks et al. 2017 7.1.1.2.1 Phylum "Nitrospirae_1" 7.1.1.2.2 Phylum Chrysiogenetes Garrity and Holt 2001 7.1.2.1.1 Phylum "Nitrospirae" Garrity and Holt 2001 [polyphyletic] 7.1.2.1.2.1.1 Candidate phylum Rokubacteria (CSP1-6) Hug et al. -

Libros Sobre Enfermedades Autoinmunes: Tratamientos, Tipos Y Diagnósticos- Profesor Dr
- LIBROS SOBRE ENFERMEDADES AUTOINMUNES: TRATAMIENTOS, TIPOS Y DIAGNÓSTICOS- PROFESOR DR. ENRIQUE BARMAIMON- 9 TOMOS- AÑO 2020.1- TOMO VI- - LIBROS SOBRE ENFERMEDADES AUTOINMUNES: TRATAMIENTOS, TIPOS Y DIAGNÓSTICOS . AUTOR: PROFESOR DR. ENRIQUE BARMAIMON.- - Doctor en Medicina.- - Cátedras de: - Anestesiología - Cuidados Intensivos - Neuroanatomía - Neurofisiología - Psicofisiología - Neuropsicología. - 9 TOMOS - - TOMO VI - -AÑO 2020- 1ª Edición Virtual: (.2020. 1)- - MONTEVIDEO, URUGUAY. 1 - LIBROS SOBRE ENFERMEDADES AUTOINMUNES: TRATAMIENTOS, TIPOS Y DIAGNÓSTICOS- PROFESOR DR. ENRIQUE BARMAIMON- 9 TOMOS- AÑO 2020.1- TOMO VI- - Queda terminantemente prohibido reproducir este libro en forma escrita y virtual, total o parcialmente, por cualquier medio, sin la autorización previa del autor. -Derechos reservados. 1ª Edición. Año 2020. Impresión [email protected]. - email: [email protected].; y [email protected]; -Montevideo, 15 de enero de 2020. - BIBLIOTECA VIRTUAL DE SALUD del S. M.U. del URUGUAY; y BIBLIOTECA DEL COLEGIO MÉDICO DEL URUGUAY. 0 0 0 0 0 0 0 0. 2 - LIBROS SOBRE ENFERMEDADES AUTOINMUNES: TRATAMIENTOS, TIPOS Y DIAGNÓSTICOS- PROFESOR DR. ENRIQUE BARMAIMON- 9 TOMOS- AÑO 2020.1- TOMO VI- - TOMO V I - 3 - LIBROS SOBRE ENFERMEDADES AUTOINMUNES: TRATAMIENTOS, TIPOS Y DIAGNÓSTICOS- PROFESOR DR. ENRIQUE BARMAIMON- 9 TOMOS- AÑO 2020.1- TOMO VI- - ÍNDICE.- - TOMO I . - - ÍNDICE. - PRÓLOGO.- - INTRODUCCIÓN. - CAPÍTULO I: -1)- GENERALIDADES. -1.1)- DEFINICIÓN. -1.2)- CAUSAS Y FACTORES DE RIESGO. -1.2.1)- FACTORES EMOCIONALES. -1.2.2)- FACTORES AMBIENTALES. -1.2.3)- FACTORES GENÉTICOS. -1.3)- Enterarse aquí, como las 10 Tipos de semillas pueden mejorar la salud. - 1.4)- TIPOS DE TRATAMIENTO DE ENFERMEDADES AUTOINMUNES. -1.4.1)- Remedios Naturales. -1.4.1.1)- Mejorar la Dieta.