Technical Report Series Number 73-7 a MANUAL of FLATFISH
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Practical Handbook for Determining the Ages of Gulf of Mexico And
A Practical Handbook for Determining the Ages of Gulf of Mexico and Atlantic Coast Fishes THIRD EDITION GSMFC No. 300 NOVEMBER 2020 i Gulf States Marine Fisheries Commission Commissioners and Proxies ALABAMA Senator R.L. “Bret” Allain, II Chris Blankenship, Commissioner State Senator District 21 Alabama Department of Conservation Franklin, Louisiana and Natural Resources John Roussel Montgomery, Alabama Zachary, Louisiana Representative Chris Pringle Mobile, Alabama MISSISSIPPI Chris Nelson Joe Spraggins, Executive Director Bon Secour Fisheries, Inc. Mississippi Department of Marine Bon Secour, Alabama Resources Biloxi, Mississippi FLORIDA Read Hendon Eric Sutton, Executive Director USM/Gulf Coast Research Laboratory Florida Fish and Wildlife Ocean Springs, Mississippi Conservation Commission Tallahassee, Florida TEXAS Representative Jay Trumbull Carter Smith, Executive Director Tallahassee, Florida Texas Parks and Wildlife Department Austin, Texas LOUISIANA Doug Boyd Jack Montoucet, Secretary Boerne, Texas Louisiana Department of Wildlife and Fisheries Baton Rouge, Louisiana GSMFC Staff ASMFC Staff Mr. David M. Donaldson Mr. Bob Beal Executive Director Executive Director Mr. Steven J. VanderKooy Mr. Jeffrey Kipp IJF Program Coordinator Stock Assessment Scientist Ms. Debora McIntyre Dr. Kristen Anstead IJF Staff Assistant Fisheries Scientist ii A Practical Handbook for Determining the Ages of Gulf of Mexico and Atlantic Coast Fishes Third Edition Edited by Steve VanderKooy Jessica Carroll Scott Elzey Jessica Gilmore Jeffrey Kipp Gulf States Marine Fisheries Commission 2404 Government St Ocean Springs, MS 39564 and Atlantic States Marine Fisheries Commission 1050 N. Highland Street Suite 200 A-N Arlington, VA 22201 Publication Number 300 November 2020 A publication of the Gulf States Marine Fisheries Commission pursuant to National Oceanic and Atmospheric Administration Award Number NA15NMF4070076 and NA15NMF4720399. -

Seamap Environmental and Biological Atlas of the Gulf of Mexico, 2014
environmental and biological atlas of the gulf of mexico 2014 gulf states marine fisheries commission number 262 february 2017 seamap SEAMAP ENVIRONMENTAL AND BIOLOGICAL ATLAS OF THE GULF OF MEXICO, 2014 Edited by Jeffrey K. Rester Gulf States Marine Fisheries Commission Manuscript Design and Layout Ashley P. Lott Gulf States Marine Fisheries Commission GULF STATES MARINE FISHERIES COMMISSION FEBRUARY 2017 NUMBER 262 This project was supported in part by the National Oceanic and Atmospheric Administration, National Marine Fisheries Service, under State/Federal Project Number NA16NMFS4350111. GULF STATES MARINE FISHERIES COMMISSION COMMISSIONERS ALABAMA John Roussel N. Gunter Guy, Jr. 1221 Plains Port Hudson Road Alabama Department of Conservation Zachary, LA 70791 and Natural Resources 64 North Union Street MISSISSIPPI Montgomery, AL 36130-1901 Jamie Miller, Executive Director Mississippi Department of Marine Resources Steve McMillan 1141 Bayview Avenue P.O. Box 337 Biloxi, MS 39530 Bay Minette, AL 36507 Senator Brice Wiggins Chris Nelson 1501 Roswell Street Bon Secour Fisheries, Inc. Pascagoula, MS 39581 P.O. Box 60 Bon Secour, AL 36511 Joe Gill, Jr. Joe Gill Consulting, LLC FLORIDA 910 Desoto Street Nick Wiley, Executive Director Ocean Springs, MS 39566-0535 FL Fish and Wildlife Conservation Commission 620 South Meridian Street TEXAS Tallahassee, FL 32399-1600 Carter Smith, Executive Director Texas Parks and Wildlife Department Senator Thad Altman 4200 Smith School Road State Senator, District 24 Austin, TX 78744 6767 North Wickham Road, Suite 211 Melbourne, FL 32940 Troy B. Williamson, II P.O. Box 967 TBA Corpus Christi, TX 78403 LOUISIANA Representative Wayne Faircloth Jack Montoucet, Secretary Texas House of Representatives LA Department of Wildlife and Fisheries 2121 Market Street, Suite 205 P.O. -

Sand Flounder (Family Paralichthyidae) Diversity in North Carolina by the Ncfishes.Com Team
Sand Flounder (Family Paralichthyidae) Diversity in North Carolina By the NCFishes.com Team Along North Carolina’s shore there are four families of flatfish comprising 36 species having eyes on the left side of their body facing upward when lying in or atop the substrate (NCFishes.com; Table 1; Figure 1). The families and species can be confusing to tell apart. The key characteristics provided for in Table 1 should enable one to differentiate between the four families and this document will aid you in the identification of the species in the Family Paralichthyidae in North Carolina. Table 1. The four families of left-facing flounders found along and off the coast of North Carolina. Family Common Name Key Characteristics (adapted from Munroe 2002) No. Species Preopercle exposed, its posterior margin free and visible, not hidden by skin or scales. Dorsal fin long, originating above, lateral to, or anterior to upper eye. Dorsal and anal fins not attached to caudal fin. Both pectoral Paralichthyidae Sand Flounders fins present. Both pelvic fins present, with 5 or 6 rays. 20 Margin of preopercle not free (hidden beneath skin and scales). Pectoral fins absent in adults. Lateral line absent on both sides of body. Cynoglossidae Tonguefishes Dorsal and anal fins joined to caudal fin. No branched caudal-fin rays. 9 Lateral line absent or poorly developed on blind side; lateral line absent below lower eye. Lateral line of eyed side with high arch over pectoral Bothidae Lefteye Flounders fin. Pelvic fin of eyed side on midventral line. 6 Both pelvic fins elongate, placed close to midline and extending forward to urohyal. -

The Flounder Fishery of the Gulf of Mexico, United States: a Regional Management Plan
The Flounder Fishery of the Gulf of Mexico, United States: A Regional Management Plan ..... .. ·. Gulf States Marine Fisheries Commission October 2000 Number83 GULF STATES MARINE FISHERIES COMMISSION Commissioners and Proxies Alabama Warren Triche Riley Boykin Smith Louisiana House of Representatives Alabama Department of Conservation & Natural 100 Tauzin Lane Resources Thibodaux, Louisiana 70301 64 North Union Street Montgomery, Alabama 36130-1901 Frederic L. Miller proxy: Vernon Minton P.O. Box 5098 Marine Resources Division Shreveport, Louisiana 71135-5098 P.O. Drawer 458 Gulf Shores, Alabama 36547 Mississippi Glenn H. Carpenter Walter Penry Mississippi Department of Marine Resources Alabama House of Representatives 1141 Bayview Avenue, Suite 101 12040 County Road 54 Biloxi, Mississippi 39530 Daphne, Alabama 36526 proxy: William S. “Corky” Perret Mississippi Department of Marine Resources Chris Nelson 1141 Bayview Avenue, Suite 101 Bon Secour Fisheries, Inc. Biloxi, Mississippi 39530 P.O. Box 60 Bon Secour, Alabama 36511 Billy Hewes Mississippi Senate Florida P.O. Box 2387 Allan L. Egbert Gulfport, Mississippi 39505 Florida Fish & Wildlife Conservation Commission 620 Meridian Street George Sekul Tallahassee, Florida 323299-1600 805 Beach Boulevard, #302 proxies: Ken Haddad, Director Biloxi, Mississippi 39530 Florida Marine Research Institute 100 Eighth Avenue SE Texas St. Petersburg, Florida 33701 Andrew Sansom Texas Parks & Wildlife Department Ms. Virginia Vail 4200 Smith School Road Division of Marine Resources Austin, Texas 78744 Fish & Wildlife Conservation Commission proxies: Hal Osburn and Mike Ray 620 Meridian Street Texas Parks & Wildlife Department Tallahassee, Florida 32399-1600 4200 Smith School Road Austin, Texas 78744 William W. Ward 2221 Corrine Street J.E. “Buster” Brown Tampa, Florida 33605 Texas Senate P.O. -
St. Lucie, Units 1 and 2
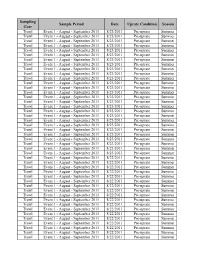
Sampling Sample Period Date Uprate Condition Season Gear Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event 1 - August - September 2011 8/23/2011 Pre-uprate Summer Trawl Event -

Isopods (Isopoda: Aegidae, Cymothoidae, Gnathiidae) Associated with Venezuelan Marine Fishes (Elasmobranchii, Actinopterygii)
Isopods (Isopoda: Aegidae, Cymothoidae, Gnathiidae) associated with Venezuelan marine fishes (Elasmobranchii, Actinopterygii) Lucy Bunkley-Williams,1 Ernest H. Williams, Jr.2 & Abul K.M. Bashirullah3 1 Caribbean Aquatic Animal Health Project, Department of Biology, University of Puerto Rico, P.O. Box 9012, Mayagüez, PR 00861, USA; [email protected] 2 Department of Marine Sciences, University of Puerto Rico, P.O. Box 908, Lajas, Puerto Rico 00667, USA; ewil- [email protected] 3 Instituto Oceanografico de Venezuela, Universidad de Oriente, Cumaná, Venezuela. Author for Correspondence: LBW, address as above. Telephone: 1 (787) 832-4040 x 3900 or 265-3837 (Administrative Office), x 3936, 3937 (Research Labs), x 3929 (Office); Fax: 1-787-834-3673; [email protected] Received 01-VI-2006. Corrected 02-X-2006. Accepted 13-X-2006. Abstract: The parasitic isopod fauna of fishes in the southern Caribbean is poorly known. In examinations of 12 639 specimens of 187 species of Venezuelan fishes, the authors found 10 species in three families of isopods (Gnathiids, Gnathia spp. from Diplectrum radiale*, Heteropriacanthus cruentatus*, Orthopristis ruber* and Trachinotus carolinus*; two aegids, Rocinela signata from Dasyatis guttata*, H. cruentatus*, Haemulon auro- lineatum*, H. steindachneri* and O. ruber; and Rocinela sp. from Epinephelus flavolimbatus*; five cymothoids: Anilocra haemuli from Haemulon boschmae*, H. flavolineatum* and H. steindachneri*; Anilocra cf haemuli from Heteropriacanthus cruentatus*; Haemulon bonariense*, O. ruber*, Cymothoa excisa in H. cruentatus*; Cymothoa oestrum in Chloroscombrus chrysurus, H. cruentatus* and Priacanthus arenatus; Cymothoa sp. in O. ruber; Livoneca sp. from H. cruentatus*; and Nerocila fluviatilis from H. cruentatus* and P. arenatus*). The Rocinela sp. and A. -

Kinds and Abundance of Fish Larvae in the Eastern Tropical Pacific, Based on Collections Made on Eastropac I
KINDS AND ABUNDANCE OF FISH LARVAE IN THE EASTERN TROPICAL PACIFIC, BASED ON COLLECTIONS MADE ON EASTROPAC I ELBERTH. AHLSTROM' ABSTRACT This paper deals with kinds and counts of fish larvae obtained in 482 oblique plankton hauls taken over an extensive area of the eastern tropical Pacific on EASTROPAC I, a four-vessel cooperative survey made during February-March 1967. On the basis of abundance of larvae, the dominant fish group in oceanic waters are the myctophid lanternfishes (47 %), gonostornatid lightfishes (23 %), hatchetfishes, Sternoptychidae (6 YO),bathylagid smelts (5 %). Scombrid larvae ranked fifth, and ex- ceeded 2 % of the count. Two kinds of larvae were outstandingly abundant : larvae of the lanternfish Diogenichthys laternatus made up over 25 % of the total, while larvae of the gonostornatid genus Vinciguerriu made up almost 20 %. More fish larvae were obtained per haul, on the average, in the eastern tropical Pacific than were obtained per haul in the intensively surveyed waters of the California Current region off Cal- ifornia and Baja California. EASTROPAC I was the first and most wide- bution and abundance of sardine spawning, and ranging of a series of cooperative cruises made the factors underlying fluctuations in survival in the eastern tropical Pacific between February of the early life-history stages of sardines. The 1967 and April 1968. A vast expanse of the plankton collections not only contained eggs and eastern tropical Pacific was surveyed on EAS- larvae of sardine but those of most other pelagic TROPAC I, extending from lat 20" N to 20" S, fishes in the California Current region. -
PARALICHTHYIDAE Sand Flounders by T.A
click for previous page 1898 Bony Fishes PARALICHTHYIDAE Sand flounders by T.A. Munroe, National Marine Fisheries Service, National Museum of Natural History, Washington D.C., USA iagnostic characters: Most species with eyes on left side of head, reversals frequent in some species D(right-eyed individuals nearly as common as left-eyed in some species occurring outside the Atlantic). No spines present in fins. Mouth protractile, asymmetrical, lower jaw moderately prominent; teeth in jaws some- times canine-like; no teeth on vomer. Preopercle exposed, its posterior margin free and visible, not hid- den by skin or scales. Urinary papilla on ocular side (Paralichthys group) or blind side (Cyclopsetta group), not attached to first anal-fin ray. Dorsal fin long, originating above, lateral to, or anterior to upper eye. Dorsal and anal fins not attached to caudal fin. Both pectoral fins present. Both pelvic fins present, with 5 or 6 rays (6 rays in nearly all species); base of pelvic fin of ocular side on midventral line (Cyclopsetta group), or pelvic fins symmetrically or nearly symmetrically placed on either side of midventral line (base of neither pelvic fin on midventral line) (Paralichthys group). Caudal fin with 17 or 18 rays, 10 to 13 rays branched (usually 11 or 13, rarely 10 or 12).Lateral line present and obvious on both sides of body; lateral line with (Paralichthys group) or without (Cyclopsetta group) high arch over pectoral fin; lateral line present (Paralichthys group) or absent (Cyclopsetta group) below lower eye. Some species of the Cyclopsetta group (some species of Syacium, Citharichthys, and possibly Etropus in this area) show sexual dimorphism in interorbital width, length of the pectoral fin on the ocular side, length of the anterior dorsal-fin rays, and color- ation. -

Light Induced Changes in Starry Flounder (Platichthys Stellatus) Opsin Expression 2 and Its Influence on Vision Estimated from a Camouflage-Based Behavioural Assay
bioRxiv preprint doi: https://doi.org/10.1101/2020.07.30.228627; this version posted July 30, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 1 Full title: Light induced changes in starry flounder (Platichthys stellatus) opsin expression 2 and its influence on vision estimated from a camouflage-based behavioural assay. 3 4 Short title: Rapid light induced changes in opsin expression. 5 6 Tom Iwanicki1,#a*, Cliff Haman2, Amy Liu1,#b, and John S. Taylor1* 7 8 1 Department of Biology, University of Victoria, Victoria, British Columbia, Canada 9 2 Department of Fine Arts, University of Victoria, Victoria, British Columbia, Canada 10 #a Current address: School of Life Sciences, Department of Biology, University of Hawai’i at 11 Mānoa, Honolulu, Hawai’i, USA 12 #b Current address: Department of Zoology, University of British Columbia, Vancouver, British 13 Columbia, Canada 14 *Authors for correspondence ([email protected] and [email protected]) 15 16 Author contributions: TI and JST were responsible for conceptualization, investigation, and 17 original draft preparation, CH contributed to methodology and resources, AL contributed to 18 investigation, JST contributed funding acquisition, and all authors reviewed and edited the final 19 manuscript. 20 21 Keywords: opsin, gene expression, camouflage, vision, spectral tuning 22 23 Grant sponsor: Natural Sciences and Engineering Research Council of Canada (NSERC) 24 Discovery Grant number: 34607-57200 (JST) 25 1 bioRxiv preprint doi: https://doi.org/10.1101/2020.07.30.228627; this version posted July 30, 2020. -

Phylogenetic Position of the Citharidae, a Family of Flatfishes
MISCELLANEOUS PUBLICATIONS MUSEUM OFZOOLOGY, UNIVERSITY OFMICHIGAN, NO. 63 PHYLOGENETIC POSITION OF THE CITHARIDAE, A FAMILY OF FLATFISHES BY CARL L. HUBBS ANN ARBOR UNIVERSITY OF MICHIGAN PRESS NOVEMBER23, 1945 PRICE LIST OF THE MISCELLANEOUS PUBLICATIONS OF THE blIUSEUM OF ZOOLOGY, UNIVERSITY OF MICHIGAN Address inquiries to the Director of the Museum of Zoology, AM Arbor, Michigan. Bound in Paper No. 1. Directions for Collecting and Preserving Specimens of Dragonflies for Museum Purposes. By E. B. WILLIAMSON.(1916) Pp. 15, 3 figures GO.% NO. 2. An Annotated List of the Odonata of Indiana. By E. B. WILLIAMSON. (1917) Pp. 12, 1 map $0.26 No. 3. A Collecting Trip to Col (1918) Pp. 24. (Out of print) No. 4. Contributions to the Botany of Michigan. By C. E. DODGE. (1918) $0.26 NO. 5. $0.46 No. 6. 213,l plate, 223 figures $3.00 No. 7. (1922) Pp. 57, 3 plates $0.76 No. 8. The Amphibians and Reptiles of the Sierra Nevada de Santa Marta, Colombia. By ALEXANDERQ. RUTHVEN.(1922) Pp. 69, 13 plates, 2 flgures, 1 map $1.00 No. 9. Notes on American Species of Triaca a and Gynacantha. E. B. WILLIAMSON.(1923) Pp. 67, 7 plate $0.76 NO. 10. A Preliminary Survey of the Bird Life of North Dakota. By NORMAN A. WOOD. (1923) Pp. 85, G plates, 1 map $1.00 No. 11. Notes on the Genus Erythemis, with a Description of a New Species (Odonata). By E. B. WILLIAMSON. The Phylogeny and the Distribution of the Genus Erythemis (Odonata). By CLARENCEH. KENNEDY.(1923) Pp. -
Assessment of Deep Demersal Fish Fauna Diversity of the Colombian Caribbean Sea
MARINE AND FISHERY SCIENCES 33 (2): 227-246 (2020) https://doi.org/10.47193/mafis.3322020301106 227 MARINE IMPACTS IN THE ANTHROPOCENE Assessment of deep demersal fish fauna diversity of the Colombian Caribbean Sea CAMILO B. GARCÍA* and JORGE M. GAMBOA Departamento de Biología, Universidad Nacional de Colombia, Carrera 45 # 26-85, Bogotá, Colombia ABSTRACT. Marine and We compiled georeferenced records of deep demersal fishes from the Colombian Fishery Sciences Caribbean Sea in order to assess the level of survey coverage and geographic completeness of MAFIS species richness inventories at a scale of 15 min by 15 min cells, in view of threats from fishing and oil and natural gas exploration. We identified a rich fauna with a minimum of 362 species registered. Areas with high observed and predicted species richness were identified. Survey coverage and geo- graphic richness completeness resulted in being deficient with no cell reaching the status of well- sampled spatial unit, being 83% of the Colombian Caribbean Exclusive Economic Zone bottoms unexplored, particularly depths beyond 1,000 m. A plea is made for renewed survey efforts with a focus on the protection of the Colombian Caribbean deep-sea biota. Key words: Colombian Caribbean, deep fishes, records, soft-bottoms, species richness. Evaluación de la diversidad de la fauna de peces demersales profundos del Mar Caribe colom- biano RESUMEN. Se recopilaron registros georreferenciados de peces demersales profundos del Mar Caribe colombiano con el fin de evaluar el nivel de cobertura de la prospección y la integridad geográfica de los inventarios de riqueza específica a una escala de celdas de 15 min por 15 min, en vista de las amenazas de la pesca y la explotación de petróleo y gas. -
Molecular and Morphological Analysis of Living and Fossil Taxa
Received: 22 December 2018 | Revised: 10 June 2019 | Accepted: 10 June 2019 DOI: 10.1111/zsc.12372 ORIGINAL ARTICLE Origins and relationships of the Pleuronectoidei: Molecular and morphological analysis of living and fossil taxa Matthew A. Campbell1 | Bruno Chanet2 | Jhen‐Nien Chen1 | Mao‐Ying Lee1 | Wei‐Jen Chen1 1Institute of Oceanography, National Taiwan University, Taipei, Taiwan Abstract 2Département Origines et Évolution, Flatfishes (Pleuronectiformes) are a species‐rich and distinct group of fishes charac- Muséum National d‘Histoire Naturelle, terized by cranial asymmetry. Flatfishes occupy a wide diversity of habitats, includ- Paris, France ing the tropical deep‐sea and freshwaters, and often are small‐bodied fishes. Most Correspondence scientific effort, however, has been focused on large‐bodied temperate marine species Wei‐Jen Chen, Institute of Oceanography, important in fisheries. Phylogenetic study of flatfishes has also long been limited in National Taiwan University, Room 301, scope and focused on the placement and monophyly of flatfishes. As a result, several No.1 Sec. 4 Roosevelt Rd., Taipei 10617, Taiwan. questions in systematic biology have persisted that molecular phylogenetic study can Email: [email protected] answer. We examine the Pleuronectoidei, the largest suborder of Pleuronectiformes with >99% of species diversity of the order, in detail with a multilocus nuclear and Funding information Ministry of Science and Technology, mitochondrial data set of 57 pleuronectoids from 13 families covering a wide range of Taiwan, Grant/Award Number: 102-2923- habitats. We combine the molecular data with a morphological matrix to construct a B-002 -001 and 107-2611-M-002-007; Fulbright Taiwan; Agence Nationale de la total evidence phylogeny that places fossil flatfishes among extant lineages.