Robust Extraction, Separation, and Quantitation of Structural Isomer Steroids from Human Plasma by SPE-UHPLC-MS/MS
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Medical Record Requirements for Pre-Service Reviews
Medical Record Requirements for Pre-Service Reviews This document lists medical record requirements for pre-service reviews. These requirements are developed using the clinical criteria in UnitedHealthcare medical policies in conjunction with the guidance provided by UnitedHealthcare physicians and pharmacists with experience in reviewing pre-service requests for coverage. These medical record requirements were developed in an effort to decrease the need for repeated requests for additional information and to improve turnaround time for coverage decisions. Please prepare the suggested materials in advance. We reserve the right to request more information, if necessary. Medical record requirements for case review(s) may vary among various UnitedHealthcare Commercial, UnitedHealthcare Community Plan and UnitedHealthcare Medicare Advantage benefit plans. Please review the requirements for notifications and prior authorization requests at UHCprovider.com/priorauth. These medical record requirements are provided for reference purposes only and may not include all services or codes. Listing of a service or code in this document does not imply that it is a covered or non-covered health service or code. Benefit coverage for health services is determined by the member specific benefit plan document and applicable laws. This document is the property of UnitedHealthcare and unauthorized copying, use or distribution of this information is strictly prohibited. It is regularly reviewed, updated and subject to change. Click a service category from the Table of Contents to jump to the applicable section of this document. Proprietary Information of UnitedHealthcare. Copyright 2020 United HealthCare Services, Inc. Page 1 of 141 Table of Contents Click a service category below to jump to the applicable section of this document. -

Progesterone and 17 Α-Hydroxyprogesterone: Novel Stimulators of Calcium Influx in Human Sperm Peter F
Old Dominion University ODU Digital Commons Bioelectrics Publications Frank Reidy Research Center for Bioelectrics 1990 Progesterone and 17 α-Hydroxyprogesterone: Novel Stimulators of Calcium Influx in Human Sperm Peter F. Blackmore Stephen J. Beebe Old Dominion University, [email protected] Douglas R. Danforth Nancy Alexander Follow this and additional works at: https://digitalcommons.odu.edu/bioelectrics_pubs Part of the Cell and Developmental Biology Commons, Molecular Biology Commons, Reproductive and Urinary Physiology Commons, and the Structural Biology Commons Repository Citation Blackmore, Peter F.; Beebe, Stephen J.; Danforth, Douglas R.; and Alexander, Nancy, "Progesterone and 17 α-Hydroxyprogesterone: Novel Stimulators of Calcium Influx in Human Sperm" (1990). Bioelectrics Publications. 78. https://digitalcommons.odu.edu/bioelectrics_pubs/78 Original Publication Citation Blackmore, P.F., Beebe, S.J., Danforth, D.R., & Alexander, N. (1990). Progesterone and 17 α-hydroxyprogesterone: Novel stimulators of calcium influx in human sperm. Journal of Biological Chemistry, 265(3), 1376-1380. This Article is brought to you for free and open access by the Frank Reidy Research Center for Bioelectrics at ODU Digital Commons. It has been accepted for inclusion in Bioelectrics Publications by an authorized administrator of ODU Digital Commons. For more information, please contact [email protected]. THE JOURNAL OF B~OLOCXCAL CHEMISTRY Vol. 265, No. 3, Issue of January 25, pp. 1376-1380,199O 0 1990 by The American Society for Biochemistry and Molecular Biolow, Inc Printed in U. S. A. Progesterone and 17wHydroxyprogesterone NOVEL STIMULATORS OF CALCIUM INFLUX IN HUMAN SPERM* (Received for publication, August 21, 1989) Peter F. BlackmoreS& Stephen J. Beeben, Douglas R. Danforthn, and Nancy Alexanderll From the Departments of SPharmacology and IlObstetrics and Gynecology and the Jones Institute, Eastern Virginia Medical School, Norfolk, Virginia 23501 Progesterone and 17a-hydroxyprogesterone (but not reaction (AR)’ in human (e.g. -

Dehydroepiandrosterone: a Potential Therapeutic Agent in the Treatment
Bentley et al. Burns & Trauma (2019) 7:26 https://doi.org/10.1186/s41038-019-0158-z REVIEW Open Access Dehydroepiandrosterone: a potential therapeutic agent in the treatment and rehabilitation of the traumatically injured patient Conor Bentley1,2,3* , Jon Hazeldine1,3, Carolyn Greig2,4, Janet Lord1,3,4 and Mark Foster1,5 Abstract Severe injuries are the major cause of death in those aged under 40, mainly due to road traffic collisions. Endocrine, metabolic and immune pathways respond to limit the tissue damage sustained and initiate wound healing, repair and regeneration mechanisms. However, depending on age and sex, the response to injury and patient prognosis differ significantly. Glucocorticoids are catabolic and immunosuppressive and are produced as part of the stress response to injury leading to an intra-adrenal shift in steroid biosynthesis at the expense of the anabolic and immune enhancing steroid hormone dehydroepiandrosterone (DHEA) and its sulphated metabolite dehydroepiandrosterone sulphate (DHEAS). The balance of these steroids after injury appears to influence outcomes in injured humans, with high cortisol: DHEAS ratio associated with increased morbidity and mortality. Animal models of trauma, sepsis, wound healing, neuroprotection and burns have all shown a reduction in pro- inflammatory cytokines, improved survival and increased resistance to pathological challenges with DHEA supplementation. Human supplementation studies, which have focused on post-menopausal females, older adults, or adrenal insufficiency have shown that restoring the cortisol: DHEAS ratio improves wound healing, mood, bone remodelling and psychological well-being. Currently, there are no DHEA or DHEAS supplementation studies in trauma patients, but we review here the evidence for this potential therapeutic agent in the treatment and rehabilitation of the severely injured patient. -

2019 Prohibited List
THE WORLD ANTI-DOPING CODE INTERNATIONAL STANDARD PROHIBITED LIST JANUARY 2019 The official text of the Prohibited List shall be maintained by WADA and shall be published in English and French. In the event of any conflict between the English and French versions, the English version shall prevail. This List shall come into effect on 1 January 2019 SUBSTANCES & METHODS PROHIBITED AT ALL TIMES (IN- AND OUT-OF-COMPETITION) IN ACCORDANCE WITH ARTICLE 4.2.2 OF THE WORLD ANTI-DOPING CODE, ALL PROHIBITED SUBSTANCES SHALL BE CONSIDERED AS “SPECIFIED SUBSTANCES” EXCEPT SUBSTANCES IN CLASSES S1, S2, S4.4, S4.5, S6.A, AND PROHIBITED METHODS M1, M2 AND M3. PROHIBITED SUBSTANCES NON-APPROVED SUBSTANCES Mestanolone; S0 Mesterolone; Any pharmacological substance which is not Metandienone (17β-hydroxy-17α-methylandrosta-1,4-dien- addressed by any of the subsequent sections of the 3-one); List and with no current approval by any governmental Metenolone; regulatory health authority for human therapeutic use Methandriol; (e.g. drugs under pre-clinical or clinical development Methasterone (17β-hydroxy-2α,17α-dimethyl-5α- or discontinued, designer drugs, substances approved androstan-3-one); only for veterinary use) is prohibited at all times. Methyldienolone (17β-hydroxy-17α-methylestra-4,9-dien- 3-one); ANABOLIC AGENTS Methyl-1-testosterone (17β-hydroxy-17α-methyl-5α- S1 androst-1-en-3-one); Anabolic agents are prohibited. Methylnortestosterone (17β-hydroxy-17α-methylestr-4-en- 3-one); 1. ANABOLIC ANDROGENIC STEROIDS (AAS) Methyltestosterone; a. Exogenous* -

The Pennsylvania State University the Graduate School College of Health and Human Development
The Pennsylvania State University The Graduate School College of Health and Human Development A FUNCTIONAL ASSESSMENT OF SALIVARY DEHYDROEPIANDROSTERONE SULFATE (DHEA-S) IN RESPONSE TO ACUTE STRESS A Dissertation in Biobehavioral Health by Courtney Allison Whetzel ©2008 Courtney Allison Whetzel Submitted in Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy August 2008 ii The dissertation of Courtney Allison Whetzel was reviewed and approved* by the following: Laura Cousino Klein Associate Professor of Biobehavioral Health Dissertation Advisor Chair of Committee Sonia A. Cavigelli Assistant Professor of Biobehavioral Health Elizabeth J. Susman Jean Phillips Shibley Professor of Biobehavioral Health David M. Almeida Professor Human Development and Family Studies Collins O. Airhihenbuwa Professor of Biobehavioral Health, Head, Department of Biobehavioral Health *Signatures are on file in the Graduate School. iii ABSTRACT Dehydroepiandrosterone sulfate (DHEA-S) is one of the most widely circulating hormones in the body, and adult men have higher DHEA-S levels compared to adult women. Due to its unique lifetime rhythm (i.e., levels rise at 6-8 yrs, peak at 20-30 yrs, decline until death) and purported protective role in the health effects of aging (e.g., cardiovascular disease, dementia), DHEA-S has been studied as an anti-aging hormone. The antiglucocorticoid properties of DHEA-S suggest that it may protect against the long-term health consequences of chronic, stress-induced cortisol release. Little is known about the role of DHEA-S in the biological stress response in humans, but a handful of studies suggest that DHEA-S levels increase following intense physical stressors. -
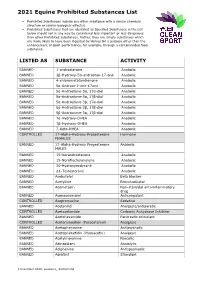
2021 Equine Prohibited Substances List
2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). Prohibited Substances that are identified as Specified Substances in the List below should not in any way be considered less important or less dangerous than other Prohibited Substances. Rather, they are simply substances which are more likely to have been ingested by Horses for a purpose other than the enhancement of sport performance, for example, through a contaminated food substance. LISTED AS SUBSTANCE ACTIVITY BANNED 1-androsterone Anabolic BANNED 3β-Hydroxy-5α-androstan-17-one Anabolic BANNED 4-chlorometatandienone Anabolic BANNED 5α-Androst-2-ene-17one Anabolic BANNED 5α-Androstane-3α, 17α-diol Anabolic BANNED 5α-Androstane-3α, 17β-diol Anabolic BANNED 5α-Androstane-3β, 17α-diol Anabolic BANNED 5α-Androstane-3β, 17β-diol Anabolic BANNED 5β-Androstane-3α, 17β-diol Anabolic BANNED 7α-Hydroxy-DHEA Anabolic BANNED 7β-Hydroxy-DHEA Anabolic BANNED 7-Keto-DHEA Anabolic CONTROLLED 17-Alpha-Hydroxy Progesterone Hormone FEMALES BANNED 17-Alpha-Hydroxy Progesterone Anabolic MALES BANNED 19-Norandrosterone Anabolic BANNED 19-Noretiocholanolone Anabolic BANNED 20-Hydroxyecdysone Anabolic BANNED Δ1-Testosterone Anabolic BANNED Acebutolol Beta blocker BANNED Acefylline Bronchodilator BANNED Acemetacin Non-steroidal anti-inflammatory drug BANNED Acenocoumarol Anticoagulant CONTROLLED Acepromazine Sedative BANNED Acetanilid Analgesic/antipyretic CONTROLLED Acetazolamide Carbonic Anhydrase Inhibitor BANNED Acetohexamide Pancreatic stimulant CONTROLLED Acetominophen (Paracetamol) Analgesic BANNED Acetophenazine Antipsychotic BANNED Acetophenetidin (Phenacetin) Analgesic BANNED Acetylmorphine Narcotic BANNED Adinazolam Anxiolytic BANNED Adiphenine Antispasmodic BANNED Adrafinil Stimulant 1 December 2020, Lausanne, Switzerland 2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). -

Dehydroepiandrosterone Sulfate and Dehydroepiandrosterone Sulfate/Cortisol Ratio in Cirrhotic Patients with Septic Shock: Anothe
Tsai et al. Critical Care (2017) 21:214 DOI 10.1186/s13054-017-1768-0 RESEARCH Open Access Dehydroepiandrosterone sulfate and dehydroepiandrosterone sulfate/cortisol ratio in cirrhotic patients with septic shock: another sign of hepatoadrenal syndrome? Ming-Hung Tsai1†, Hui-Chun Huang2,3,4†, Yun-Shing Peng5,6, Yung-Chang Chen6,7, Ya-Chung Tian6,7, Chih-Wei Yang6,7, Jau-Min Lien1, Ji-Tseng Fang6,7, Cheng-Shyong Wu6,8, Sen-Yung Hsieh1 and Fa-Yauh Lee2,3* Abstract Background: Cirrhotic patients are susceptible to sepsis and critical illness-related corticosteroid insufficiency (CIRCI). Dehydroepiandrosterone sulfate (DHEAS) is a corticotropin-dependent adrenal androgen, which has immunostimulating and antiglucocorticoid effects. Considering the synchronized synthesis of cortisol and DHEAS and their opposing effects to each other, investigators have proposed measuring these two hormones as a ratio. Severe sepsis has been associated with low DHEAS, especially relative to high cortisol. Despite growing interest in the role of adrenal androgen replacement in critical illness, there have been no data about DHEAS and the DHEAS/cortisol ratio in patients with liver cirrhosis. We studied whether low concentrations of DHEAS and decreased DHEAS/cortisol ratio are associated with poor outcome in patients with liver cirrhosis and septic shock. Methods: We recruited 46 cirrhotic patients with septic shock, and 46 noncirrhotic counterparts matched by age and sex. We evaluated adrenal function using the short corticotropin stimulation test and analyzed the relation between DHEAS and cortisol. Results: While the nonsurvivors in the cirrhotic group had significantly lower baseline DHEAS, lower baseline DHEAS/cortisol ratio, and reduced increments of both DHEAS and cortisol upon corticotropin stimulation, the survivors had lower baseline cortisol. -

DASON COVID-19 Weekly Treatment Literature Update 7/31/2020 NIH
DASON COVID-19 Weekly Treatment Literature Update 7/31/2020 Prepared by: Angelina Davis, PharmD, MS, April Dyer, PharmD, MBA, MSCR, Elizabeth Dodds Ashley, PharmD, MHS, Melissa Johnson, PharmD, MHS, S. Shaefer Spires, MD, Travis Jones, PharmD The following summarizes key literature pertaining to treatment of COVID-19 during the past week. *Note: some of the data discussed below may be in pre-print form that has not yet been peer-reviewed. We have noted some discrepancies in some of this data, and final printed versions may ultimately differ from what is shown here. We will update as soon as possible; caution is advised when interpreting this literature. NIH Guideline Updates On Friday, July 24th and Thursday June 30th, the NIH updated treatment guidelines. The full guideline document can be found at this link: https://files.covid19treatmentguidelines.nih.gov/guidelines/covid19treatmentguidelines.pdf These are also reflected in our summary table of the NIH treatment guidelines that can be found here: https://dason.medicine.duke.edu/sites/dason.medicine.duke.edu/files/nih_covid- 19_treatment_guideline_updates_7-24-20.pdf The most recent change on July 30th included expanding the list of corticosteroids that can be used. Hydrocortisone, methylprednisolone, and prednisone are all considered alternatives when dexamethasone is in short supply. The July 24th modification was to clarify oxygen requirements for remdesivir patients and further refine considerations for situations in which remdesivir is in short supply. Patients are not separated into those who are on supplemental oxygen alone and those on high-flow oxygen, mechanical ventilation or ECMO. The updated guideline now reads: Recommendation for Prioritizing Limited Supplies of Remdesivir • Because remdesivir supplies are limited, the Panel recommends that remdesivir be prioritized for use in hospitalized patients with COVID-19 who require supplemental oxygen but who are not on high-flow oxygen, noninvasive ventilation, mechanical ventilation, or ECMO (BI). -

WO 2010/139985 Al
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date 9 December 2010 (09.12.2010) WO 2010/139985 Al (51) International Patent Classification: (74) Agent: PERRY, Robert, Edward; Gill Jennings & Every A61K 31/137 (2006.01) A61P 9/00 (2006.01) LLP, Broadgate House, 7 Eldon Street, London, Greater A61K 31/436 (2006.01) A61P 11/00 (2006.01) London EC2M 7LH (GB). A61K 45/06 (2006.01) A61P 11/06 (2006.01) (81) Designated States (unless otherwise indicated, for every (21) International Application Number: kind of national protection available): AE, AG, AL, AM, PCT/GB20 10/050905 AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, (22) International Filing Date: DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, 28 May 2010 (28.05.2010) HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, (25) Filing Language: English KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, (26) Publication Language: English NO, NZ, OM, PE, PG, PH, PL, PT, RO, RS, RU, SC, SD, (30) Priority Data: SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, 0909352.7 1 June 2009 (01 .06.2009) GB TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. 0918727.9 26 October 2009 (26.10.2009) GB (84) Designated States (unless otherwise indicated, for every 0918728.7 26 October 2009 (26.10.2009) GB kind of regional protection available): ARIPO (BW, GH, (71) Applicant (for all designated States except US): BIOCO- GM, KE, LR, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, PEA LIMITED [GB/GB]; 100 Fetter Lane, London ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, EC4A IBN (GB). -

Absoluteidq® Stero17 Kit
Superior Confi dence in Steroid Hormone Analysis AbsoluteIDQ® Stero17 Kit KIT Ready-to-Use Comprehensive Coverage Best Fit to Physiological Range Superior Data Quality Steroid Hormone Profiling by Mass Spectrometry (LC-MS) For research use only. Not for use in diagnostic procedures. Key Benefi ts The Most Accurate Steroid Hormone Analysis Steroid hormones are a group of metabolites that are derived from cholesterol. They are produced primarily by adrenal glands, testes, and ovaries. Based on their receptor, steroid hormones are classifi ed into fi ve groups: glucocorticoids, mineralocorticoids, progestogens, androgens, and estrogens. Steroid hormones exert multiple biological functions, including regulation of glucose levels, cell growth, and proliferation. The trend in steroid analysis is moving from measuring single or small sets of steroid hormones with immunoassays towards broader, mass spectrometry-based quantifi cation of steroid hormones (Jeanneret et al., J Chromatogr A 2016). The AbsoluteIDQ® Stero17 Kit is designed to help researchers to unravel the complexities of steroid hormone effects. It provides highly repro- ducible data on 17 functionally important steroid hormones in a single assay. Ready-to-Use Simple, Guided, Automated Workfl ow • All in one box: no method or reagent • Workfl ow manager guided procedure development required for enhanced process safety KIT • Implemented within one day • Automated quantifi cation and technical validation • For multiple (U)HPLC-MS platforms High Throughput Superior Data Quality • Only 7 minutes -

Application for Inclusion of Hydrocortisone Tablets in the WHO Model List of Essential Medicines for Children (July 2008)
Second Meeting of the Subcommittee of the Expert Committee on the Selection and Use of Essential Medicines Geneva, 29 September to 3 October 2008 Application for inclusion of Hydrocortisone tablets in the WHO Model List of Essential Medicines for Children (July 2008) Submitted by: Royal Children’s Hospital (RCH), Melbourne, Australia National Institute of Child Health (NICH), Karachi, Pakistan Caring & Living As Neighbours (CLAN), Australia To: Expert Committee on the Selection and Use of Essential Medicines Children’s Essential Medicines List World Health Organization Geneva Authors: Prof Garry Warne (RCH) Dr Valerie Sung (RCH) Prof Syed Jamal Raza (NICH) Dr Yasir Naqi Khan (NICH) Dr Kate Armstrong (CLAN) Contents Introduction 1. Summary statement of the proposal for inclusion 2. Name of the focal point in WHO supporting the application 3. Name of the organisations consulted and supporting the application 4. International nonproprietary name of the medicine 5. Whether listing is requested as an individual medicine or as an example of a therapeutic group 6. Information supporting the public health relevance 6.1 Definition of Congenital Adrenal Hyperplasia (CAH) 6.2 Clinical Manifestation and Variability 6.3 Epidemiology 7. Treatment details 7.1 Indications 7.2 Dose 7.3 Duration 7.4 Monitoring 8. Summary of comparative effectiveness in a variety of clinical settings 9. Summary of comparative evidence on safety 10. Summary of available data in comparative cost within the pharmacological class or therapeutic group 11. Summary of regulatory status of medicine 12. Availability of pharmacopoeial standard 13. Proposed text for the WHO Model Formulary 14. 14.1 Appendix 1 – Information on Congenital Adrenal Hyperplasia 14.2 Appendix 2 – Consensus Statement on 21OHD Management 14.3 Appendix 3 – Letters from organisations supporting this application 14.4 Appendix 4 – Glucocorticoid Treatment Clinical Guideline 15. -

Hydroxyprogesterone Caproate Injection)
MAKENA® Advisory Committee Briefing Document NDA 021945 / S-023 Page 1 ADVISORY COMMITTEE BRIEFING MATERIALS: AVAILABLE FOR PUBLIC RELEASE Bone, Reproductive, and Urologic Drugs Advisory Committee Meeting October 29, 2019 MAKENA® (hydroxyprogesterone caproate injection) NDA 021945 / S-023 AMAG Pharmaceuticals, Inc. 1100 Winter Street Waltham, MA 02451 MAKENA® Advisory Committee Briefing Document NDA 021945 / S-023 Page 2 TABLE OF CONTENTS TABLE OF CONTENTS .................................................................................................................2 LIST OF TABLES ...........................................................................................................................5 LIST OF FIGURES .........................................................................................................................7 LIST OF ABBREVIATIONS AND ACRONYMS ........................................................................8 1. EXECUTIVE SUMMARY ..........................................................................................9 1.1. Overview .......................................................................................................................9 1.2. Preterm Birth Prevalence and Prevention .....................................................................9 1.3. Makena .......................................................................................................................10 1.3.1. Approval .....................................................................................................................11