The Molpro Quantum Chemistry Package
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Free and Open Source Software for Computational Chemistry Education
Free and Open Source Software for Computational Chemistry Education Susi Lehtola∗,y and Antti J. Karttunenz yMolecular Sciences Software Institute, Blacksburg, Virginia 24061, United States zDepartment of Chemistry and Materials Science, Aalto University, Espoo, Finland E-mail: [email protected].fi Abstract Long in the making, computational chemistry for the masses [J. Chem. Educ. 1996, 73, 104] is finally here. We point out the existence of a variety of free and open source software (FOSS) packages for computational chemistry that offer a wide range of functionality all the way from approximate semiempirical calculations with tight- binding density functional theory to sophisticated ab initio wave function methods such as coupled-cluster theory, both for molecular and for solid-state systems. By their very definition, FOSS packages allow usage for whatever purpose by anyone, meaning they can also be used in industrial applications without limitation. Also, FOSS software has no limitations to redistribution in source or binary form, allowing their easy distribution and installation by third parties. Many FOSS scientific software packages are available as part of popular Linux distributions, and other package managers such as pip and conda. Combined with the remarkable increase in the power of personal devices—which rival that of the fastest supercomputers in the world of the 1990s—a decentralized model for teaching computational chemistry is now possible, enabling students to perform reasonable modeling on their own computing devices, in the bring your own device 1 (BYOD) scheme. In addition to the programs’ use for various applications, open access to the programs’ source code also enables comprehensive teaching strategies, as actual algorithms’ implementations can be used in teaching. -

Correlation.Pdf
INTRODUCTION TO THE ELECTRONIC CORRELATION PROBLEM PAUL E.S. WORMER Institute of Theoretical Chemistry, University of Nijmegen, Toernooiveld, 6525 ED Nijmegen, The Netherlands Contents 1 Introduction 2 2 Rayleigh-SchrÄodinger perturbation theory 4 3 M¿ller-Plesset perturbation theory 7 4 Diagrammatic perturbation theory 9 5 Unlinked clusters 14 6 Convergence of MP perturbation theory 17 7 Second quantization 18 8 Coupled cluster Ansatz 21 9 Coupled cluster equations 26 9.1 Exact CC equations . 26 9.2 The CCD equations . 30 9.3 CC theory versus MP theory . 32 9.4 CCSD(T) . 33 A Hartree-Fock, Slater-Condon, Brillouin 34 B Exponential structure of the wavefunction 37 C Bibliography 40 1 1 Introduction These are notes for a six hour lecture series on the electronic correlation problem, given by the author at a Dutch national winterschool in 1999. The main purpose of this course was to give some theoretical background on the M¿ller-Plesset and coupled cluster methods. Both computational methods are available in many quantum chemical \black box" programs. The audi- ence consisted of graduate students, mostly with an undergraduate chemistry education and doing research in theoretical chemistry. A basic knowledge of quantum mechanics and quantum chemistry is pre- supposed. In particular a knowledge of Slater determinants, Slater-Condon rules and Hartree-Fock theory is a prerequisite of understanding the following notes. In Appendix A this theory is reviewed briefly. Because of time limitations hardly any proofs are given, the theory is sketchily outlined. No attempt is made to integrate out the spin, the theory is formulated in terms of spin-orbitals only. -

Basis Sets Running a Calculation • in Performing a Computational
Session 2: Basis Sets • Two of the major methods (ab initio and DFT) require some understanding of basis sets and basis functions • This session describes the essentials of basis sets: – What they are – How they are constructed – How they are used – Significance in choice 1 Running a Calculation • In performing a computational chemistry calculation, the chemist has to make several decisions of input to the code: – The molecular geometry (and spin state) – The basis set used to determine the wavefunction – The properties to be calculated – The type(s) of calculations and any accompanying assumptions 2 Running a calculation •For ab initio or DFT calculations, many programs require a basis set choice to be made – The basis set is an approx- imate representation of the atomic orbitals (AOs) – The program then calculates molecular orbitals (MOs) using the Linear Combin- ation of Atomic Orbitals (LCAO) approximation 3 Computational Chemistry Map Chemist Decides: Computer calculates: Starting Molecular AOs determine the Geometry AOs O wavefunction (ψ) Basis Set (with H H ab initio and DFT) Type of Calculation LCAO (Method and Assumptions) O Properties to HH be Calculated MOs 4 Critical Choices • Choice of the method (and basis set) used is critical • Which method? • Molecular Mechanics, Ab initio, Semiempirical, or DFT • Which approximation? • MM2, MM3, HF, AM1, PM3, or B3LYP, etc. • Which basis set (if applicable)? • Minimal basis set • Split-valence • Polarized, Diffuse, High Angular Momentum, ...... 5 Why is Basis Set Choice Critical? • The -

Supporting Information
Electronic Supplementary Material (ESI) for RSC Advances. This journal is © The Royal Society of Chemistry 2020 Supporting Information How to Select Ionic Liquids as Extracting Agent Systematically? Special Case Study for Extractive Denitrification Process Shurong Gaoa,b,c,*, Jiaxin Jina,b, Masroor Abroc, Ruozhen Songc, Miao Hed, Xiaochun Chenc,* a State Key Laboratory of Alternate Electrical Power System with Renewable Energy Sources, North China Electric Power University, Beijing, 102206, China b Research Center of Engineering Thermophysics, North China Electric Power University, Beijing, 102206, China c Beijing Key Laboratory of Membrane Science and Technology & College of Chemical Engineering, Beijing University of Chemical Technology, Beijing 100029, PR China d Office of Laboratory Safety Administration, Beijing University of Technology, Beijing 100124, China * Corresponding author, Tel./Fax: +86-10-6443-3570, E-mail: [email protected], [email protected] 1 COSMO-RS Computation COSMOtherm allows for simple and efficient processing of large numbers of compounds, i.e., a database of molecular COSMO files; e.g. the COSMObase database. COSMObase is a database of molecular COSMO files available from COSMOlogic GmbH & Co KG. Currently COSMObase consists of over 2000 compounds including a large number of industrial solvents plus a wide variety of common organic compounds. All compounds in COSMObase are indexed by their Chemical Abstracts / Registry Number (CAS/RN), by a trivial name and additionally by their sum formula and molecular weight, allowing a simple identification of the compounds. We obtained the anions and cations of different ILs and the molecular structure of typical N-compounds directly from the COSMObase database in this manuscript. -
![Automated Construction of Quantum–Classical Hybrid Models Arxiv:2102.09355V1 [Physics.Chem-Ph] 18 Feb 2021](https://docslib.b-cdn.net/cover/7378/automated-construction-of-quantum-classical-hybrid-models-arxiv-2102-09355v1-physics-chem-ph-18-feb-2021-177378.webp)
Automated Construction of Quantum–Classical Hybrid Models Arxiv:2102.09355V1 [Physics.Chem-Ph] 18 Feb 2021
Automated construction of quantum{classical hybrid models Christoph Brunken and Markus Reiher∗ ETH Z¨urich, Laboratorium f¨urPhysikalische Chemie, Vladimir-Prelog-Weg 2, 8093 Z¨urich, Switzerland February 18, 2021 Abstract We present a protocol for the fully automated construction of quantum mechanical-(QM){ classical hybrid models by extending our previously reported approach on self-parametri- zing system-focused atomistic models (SFAM) [J. Chem. Theory Comput. 2020, 16 (3), 1646{1665]. In this QM/SFAM approach, the size and composition of the QM region is evaluated in an automated manner based on first principles so that the hybrid model describes the atomic forces in the center of the QM region accurately. This entails the au- tomated construction and evaluation of differently sized QM regions with a bearable com- putational overhead that needs to be paid for automated validation procedures. Applying SFAM for the classical part of the model eliminates any dependence on pre-existing pa- rameters due to its system-focused quantum mechanically derived parametrization. Hence, QM/SFAM is capable of delivering a high fidelity and complete automation. Furthermore, since SFAM parameters are generated for the whole system, our ansatz allows for a con- venient re-definition of the QM region during a molecular exploration. For this purpose, a local re-parametrization scheme is introduced, which efficiently generates additional clas- sical parameters on the fly when new covalent bonds are formed (or broken) and moved to the classical region. arXiv:2102.09355v1 [physics.chem-ph] 18 Feb 2021 ∗Corresponding author; e-mail: [email protected] 1 1 Introduction In contrast to most protocols of computational quantum chemistry that consider isolated molecules, chemical processes can take place in a vast variety of complex environments. -

Effective Hamiltonians Derived from Equation-Of-Motion Coupled-Cluster
Effective Hamiltonians derived from equation-of-motion coupled-cluster wave-functions: Theory and application to the Hubbard and Heisenberg Hamiltonians Pavel Pokhilkoa and Anna I. Krylova a Department of Chemistry, University of Southern California, Los Angeles, California 90089-0482 Effective Hamiltonians, which are commonly used for fitting experimental observ- ables, provide a coarse-grained representation of exact many-electron states obtained in quantum chemistry calculations; however, the mapping between the two is not triv- ial. In this contribution, we apply Bloch's formalism to equation-of-motion coupled- cluster (EOM-CC) wave functions to rigorously derive effective Hamiltonians in the Bloch's and des Cloizeaux's forms. We report the key equations and illustrate the theory by examples of systems with electronic states of covalent and ionic characters. We show that the Hubbard and Heisenberg Hamiltonians are extracted directly from the so-obtained effective Hamiltonians. By making quantitative connections between many-body states and simple models, the approach also facilitates the analysis of the correlated wave functions. Artifacts affecting the quality of electronic structure calculations such as spin contamination are also discussed. I. INTRODUCTION Coarse graining is commonly used in computational chemistry and physics. It is ex- ploited in a number of classic models serving as a foundation of modern solid-state physics: tight binding[1, 2], Drude{Sommerfeld's model[3{5], Hubbard's [6] and Heisenberg's[7{9] Hamiltonians. These models explain macroscopic properties of materials through effective interactions whose strengths are treated as model parameters. The values of these pa- rameters are determined either from more sophisticated theoretical models or by fitting to experimental observables. -

An Introduction to Hartree-Fock Molecular Orbital Theory
An Introduction to Hartree-Fock Molecular Orbital Theory C. David Sherrill School of Chemistry and Biochemistry Georgia Institute of Technology June 2000 1 Introduction Hartree-Fock theory is fundamental to much of electronic structure theory. It is the basis of molecular orbital (MO) theory, which posits that each electron's motion can be described by a single-particle function (orbital) which does not depend explicitly on the instantaneous motions of the other electrons. Many of you have probably learned about (and maybe even solved prob- lems with) HucÄ kel MO theory, which takes Hartree-Fock MO theory as an implicit foundation and throws away most of the terms to make it tractable for simple calculations. The ubiquity of orbital concepts in chemistry is a testimony to the predictive power and intuitive appeal of Hartree-Fock MO theory. However, it is important to remember that these orbitals are mathematical constructs which only approximate reality. Only for the hydrogen atom (or other one-electron systems, like He+) are orbitals exact eigenfunctions of the full electronic Hamiltonian. As long as we are content to consider molecules near their equilibrium geometry, Hartree-Fock theory often provides a good starting point for more elaborate theoretical methods which are better approximations to the elec- tronic SchrÄodinger equation (e.g., many-body perturbation theory, single-reference con¯guration interaction). So...how do we calculate molecular orbitals using Hartree-Fock theory? That is the subject of these notes; we will explain Hartree-Fock theory at an introductory level. 2 What Problem Are We Solving? It is always important to remember the context of a theory. -

Popular GPU-Accelerated Applications
LIFE & MATERIALS SCIENCES GPU-ACCELERATED APPLICATIONS | CATALOG | AUG 12 LIFE & MATERIALS SCIENCES APPLICATIONS CATALOG Application Description Supported Features Expected Multi-GPU Release Status Speed Up* Support Bioinformatics BarraCUDA Sequence mapping software Alignment of short sequencing 6-10x Yes Available now reads Version 0.6.2 CUDASW++ Open source software for Smith-Waterman Parallel search of Smith- 10-50x Yes Available now protein database searches on GPUs Waterman database Version 2.0.8 CUSHAW Parallelized short read aligner Parallel, accurate long read 10x Yes Available now aligner - gapped alignments to Version 1.0.40 large genomes CATALOG GPU-BLAST Local search with fast k-tuple heuristic Protein alignment according to 3-4x Single Only Available now blastp, multi cpu threads Version 2.2.26 GPU-HMMER Parallelized local and global search with Parallel local and global search 60-100x Yes Available now profile Hidden Markov models of Hidden Markov Models Version 2.3.2 mCUDA-MEME Ultrafast scalable motif discovery algorithm Scalable motif discovery 4-10x Yes Available now based on MEME algorithm based on MEME Version 3.0.12 MUMmerGPU A high-throughput DNA sequence alignment Aligns multiple query sequences 3-10x Yes Available now LIFE & MATERIALS& LIFE SCIENCES APPLICATIONS program against reference sequence in Version 2 parallel SeqNFind A GPU Accelerated Sequence Analysis Toolset HW & SW for reference 400x Yes Available now assembly, blast, SW, HMM, de novo assembly UGENE Opensource Smith-Waterman for SSE/CUDA, Fast short -

User Manual for the Uppsala Quantum Chemistry Package UQUANTCHEM V.35
User manual for the Uppsala Quantum Chemistry package UQUANTCHEM V.35 by Petros Souvatzis Uppsala 2016 Contents 1 Introduction 6 2 Compiling the code 7 3 What Can be done with UQUANTCHEM 9 3.1 Hartree Fock Calculations . 9 3.2 Configurational Interaction Calculations . 9 3.3 M¨ollerPlesset Calculations (MP2) . 9 3.4 Density Functional Theory Calculations (DFT)) . 9 3.5 Time Dependent Density Functional Theory Calculations (TDDFT)) . 10 3.6 Quantum Montecarlo Calculations . 10 3.7 Born Oppenheimer Molecular Dynamics . 10 4 Setting up a UQANTCHEM calculation 12 4.1 The input files . 12 4.1.1 The INPUTFILE-file . 12 4.1.2 The BASISFILE-file . 13 4.1.3 The BASISFILEAUX-file . 14 4.1.4 The DENSMATSTARTGUESS.dat-file . 14 4.1.5 The MOLDYNRESTART.dat-file . 14 4.1.6 The INITVELO.dat-file . 15 4.1.7 Running Uquantchem . 15 4.2 Input parameters . 15 4.2.1 CORRLEVEL ................................. 15 4.2.2 ADEF ..................................... 15 4.2.3 DOTDFT ................................... 16 4.2.4 NSCCORR ................................... 16 4.2.5 SCERR .................................... 16 4.2.6 MIXTDDFT .................................. 16 4.2.7 EPROFILE .................................. 16 4.2.8 DOABSSPECTRUM ............................... 17 4.2.9 NEPERIOD .................................. 17 4.2.10 EFIELDMAX ................................. 17 4.2.11 EDIR ..................................... 17 4.2.12 FIELDDIR .................................. 18 4.2.13 OMEGA .................................... 18 2 CONTENTS 3 4.2.14 NCHEBGAUSS ................................. 18 4.2.15 RIAPPROX .................................. 18 4.2.16 LIMPRECALC (Only openmp-version) . 19 4.2.17 DIAGDG ................................... 19 4.2.18 NLEBEDEV .................................. 19 4.2.19 MOLDYN ................................... 19 4.2.20 DAMPING ................................... 19 4.2.21 XLBOMD .................................. -

On the Calculation of Molecular Properties of Heavy Element Systems with Ab Initio Approaches: from Gas-Phase to Complex Systems André Severo Pereira Gomes
On the calculation of molecular properties of heavy element systems with ab initio approaches: from gas-phase to complex systems André Severo Pereira Gomes To cite this version: André Severo Pereira Gomes. On the calculation of molecular properties of heavy element systems with ab initio approaches: from gas-phase to complex systems. Theoretical and/or physical chemistry. Universite de Lille, 2016. tel-01960393 HAL Id: tel-01960393 https://hal.archives-ouvertes.fr/tel-01960393 Submitted on 19 Dec 2018 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. M´emoirepr´esent´epour obtenir le dipl^omed' Habilitation `adiriger des recherches { Sciences Physiques de l'Universit´ede Lille (Sciences et Technologies) ANDRE´ SEVERO PEREIRA GOMES Universit´ede Lille - CNRS Laboratoire PhLAM UMR 8523 On the calculation of molecular properties of heavy element systems with ab initio approaches: from gas-phase to complex systems M´emoirepr´esent´epour obtenir le dipl^omed' Habilitation `adiriger des recherches { Sciences Physiques de l'Universit´ede Lille (Sciences et -

Starting SCF Calculations by Superposition of Atomic Densities
Starting SCF Calculations by Superposition of Atomic Densities J. H. VAN LENTHE,1 R. ZWAANS,1 H. J. J. VAN DAM,2 M. F. GUEST2 1Theoretical Chemistry Group (Associated with the Department of Organic Chemistry and Catalysis), Debye Institute, Utrecht University, Padualaan 8, 3584 CH Utrecht, The Netherlands 2CCLRC Daresbury Laboratory, Daresbury WA4 4AD, United Kingdom Received 5 July 2005; Accepted 20 December 2005 DOI 10.1002/jcc.20393 Published online in Wiley InterScience (www.interscience.wiley.com). Abstract: We describe the procedure to start an SCF calculation of the general type from a sum of atomic electron densities, as implemented in GAMESS-UK. Although the procedure is well known for closed-shell calculations and was already suggested when the Direct SCF procedure was proposed, the general procedure is less obvious. For instance, there is no need to converge the corresponding closed-shell Hartree–Fock calculation when dealing with an open-shell species. We describe the various choices and illustrate them with test calculations, showing that the procedure is easier, and on average better, than starting from a converged minimal basis calculation and much better than using a bare nucleus Hamiltonian. © 2006 Wiley Periodicals, Inc. J Comput Chem 27: 926–932, 2006 Key words: SCF calculations; atomic densities Introduction hrstuhl fur Theoretische Chemie, University of Kahrlsruhe, Tur- bomole; http://www.chem-bio.uni-karlsruhe.de/TheoChem/turbo- Any quantum chemical calculation requires properly defined one- mole/),12 GAMESS(US) (Gordon Research Group, GAMESS, electron orbitals. These orbitals are in general determined through http://www.msg.ameslab.gov/GAMESS/GAMESS.html, 2005),13 an iterative Hartree–Fock (HF) or Density Functional (DFT) pro- Spartan (Wavefunction Inc., SPARTAN: http://www.wavefun. -
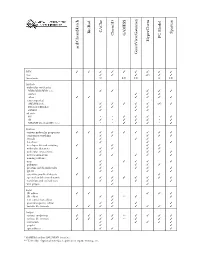
D:\Doc\Workshops\2005 Molecular Modeling\Notebook Pages\Software Comparison\Summary.Wpd
CAChe BioRad Spartan GAMESS Chem3D PC Model HyperChem acd/ChemSketch GaussView/Gaussian WIN TTTT T T T T T mac T T T (T) T T linux/unix U LU LU L LU Methods molecular mechanics MM2/MM3/MM+/etc. T T T T T Amber T T T other TT T T T T semi-empirical AM1/PM3/etc. T T T T T (T) T Extended Hückel T T T T ZINDO T T T ab initio HF * * T T T * T dft T * T T T * T MP2/MP4/G1/G2/CBS-?/etc. * * T T T * T Features various molecular properties T T T T T T T T T conformer searching T T T T T crystals T T T data base T T T developer kit and scripting T T T T molecular dynamics T T T T molecular interactions T T T T movies/animations T T T T T naming software T nmr T T T T T polymers T T T T proteins and biomolecules T T T T T QSAR T T T T scientific graphical objects T T spectral and thermodynamic T T T T T T T T transition and excited state T T T T T web plugin T T Input 2D editor T T T T T 3D editor T T ** T T text conversion editor T protein/sequence editor T T T T various file formats T T T T T T T T Output various renderings T T T T ** T T T T various file formats T T T T ** T T T animation T T T T T graphs T T spreadsheet T T T * GAMESS and/or GAUSSIAN interface ** Text only.