Docking Interactions in Protein Kinase and Phosphatase Networks Attila Reme´ Nyi, Matthew C Good and Wendell a Lim
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Circulating Supar in Two Cohorts of Primary FSGS
CLINICAL RESEARCH www.jasn.org Circulating suPAR in Two Cohorts of Primary FSGS † ‡ Changli Wei,* Howard Trachtman, Jing Li,* Chuanhui Dong, Aaron L. Friedman,§ | | | Jennifer J. Gassman, June L. McMahan, Milena Radeva, Karsten M. Heil,¶ †† ‡‡ Agnes Trautmann,¶ Ali Anarat,** Sevinc Emre, Gian M. Ghiggeri, Fatih Ozaltin,§§ || ††† Dieter Haffner, Debbie S. Gipson,¶¶ Frederick Kaskel,*** Dagmar-Christiane Fischer, ‡‡‡ Franz Schaefer,¶ and Jochen Reiser, for the PodoNet and FSGS CT Study Consortia Departments of *Medicine and ‡Neurology, University of Miami Miller School of Medicine, Miami, Florida; †Department of Pediatrics, NYU Langone Medical Center, New York, New York; §Department of Pediatrics, University of Minnesota, Minneapolis, Minnesota; |Department of Quantitative Health Sciences, Cleveland Clinic, Cleveland, Ohio; ¶Center for Pediatric and Adolescent Medicine, University of Heidelberg, Heidelberg, Germany; **Department of Pediatric Nephrology, Cukurova University School of Medicine, Adana, Turkey; ††Department of Pediatrics, Istanbul Medical Faculty, University of Istanbul, Istanbul, Turkey; ‡‡Division of Nephrology, Dialysis, and Transplantation, Laboratory on Pathophysiology of Uremia, G. Gaslini Children’s Hospital, Genoa, Italy; §§Pediatric Nephrology Unit, Department of Pediatrics, Faculty of Medicine, Hacettepe University, Ankara, Turkey; ||Department of Pediatric Kidney, Liver, and Metabolic Diseases, Hannover Medical School, Hannover, Germany; ¶¶Department of Pediatrics, University of Michigan, Ann Arbor, Michigan; ***Children’s Hospital at Montefiore, Albert Einstein College of Medicine, Bronx, New York; †††Department of Pediatrics, Rostock University Hospital, Rostock, Germany; and ‡‡‡Department of Medicine, Rush University Medical Center, Chicago, Illinois ABSTRACT Overexpression of soluble urokinase receptor (suPAR) causes pathology in animal models similar to pri- mary FSGS, and one recent study demonstrated elevated levels of serum suPAR in patients with the disease. -

Regulation of Calmodulin-Stimulated Cyclic Nucleotide Phosphodiesterase (PDE1): Review
95-105 5/6/06 13:44 Page 95 INTERNATIONAL JOURNAL OF MOLECULAR MEDICINE 18: 95-105, 2006 95 Regulation of calmodulin-stimulated cyclic nucleotide phosphodiesterase (PDE1): Review RAJENDRA K. SHARMA, SHANKAR B. DAS, ASHAKUMARY LAKSHMIKUTTYAMMA, PONNIAH SELVAKUMAR and ANURAAG SHRIVASTAV Department of Pathology and Laboratory Medicine, College of Medicine, University of Saskatchewan, Cancer Research Division, Saskatchewan Cancer Agency, 20 Campus Drive, Saskatoon SK S7N 4H4, Canada Received January 16, 2006; Accepted March 13, 2006 Abstract. The response of living cells to change in cell 6. Differential inhibition of PDE1 isozymes and its environment depends on the action of second messenger therapeutic applications molecules. The two second messenger molecules cAMP and 7. Role of proteolysis in regulating PDE1A2 Ca2+ regulate a large number of eukaryotic cellular events. 8. Role of PDE1A1 in ischemic-reperfused heart Calmodulin-stimulated cyclic nucleotide phosphodiesterase 9. Conclusion (PDE1) is one of the key enzymes involved in the complex interaction between cAMP and Ca2+ second messenger systems. Some PDE1 isozymes have similar kinetic and 1. Introduction immunological properties but are differentially regulated by Ca2+ and calmodulin. Accumulating evidence suggests that the A variety of cellular activities are regulated through mech- activity of PDE1 is selectively regulated by cross-talk between anisms controlling the level of cyclic nucleotides. These Ca2+ and cAMP signalling pathways. These isozymes are mechanisms include synthesis, degradation, efflux and seque- also further distinguished by various pharmacological agents. stration of cyclic adenosine 3':5'-monophosphate (cAMP) and We have demonstrated a potentially novel regulation of PDE1 cyclic guanosine 3':5'- monophosphate (cGMP) within the by calpain. -

Kinome Profiling of Clinical Cancer Specimens
Published OnlineFirst March 23, 2010; DOI: 10.1158/0008-5472.CAN-09-3989 Review Cancer Research Kinome Profiling of Clinical Cancer Specimens Kaushal Parikh and Maikel P. Peppelenbosch Abstract Over the past years novel technologies have emerged to enable the determination of the transcriptome and proteome of clinical samples. These data sets will prove to be of significant value to our elucidation of the mechanisms that govern pathophysiology and may provide biological markers for future guidance in person- alized medicine. However, an equally important goal is to define those proteins that participate in signaling pathways during the disease manifestation itself or those pathways that are made active during successful clinical treatment of the disease: the main challenge now is the generation of large-scale data sets that will allow us to define kinome profiles with predictive properties on the outcome-of-disease and to obtain insight into tissue-specific analysis of kinase activity. This review describes the current techniques available to gen- erate kinome profiles of clinical tissue samples and discusses the future strategies necessary to achieve new insights into disease mechanisms and treatment targets. Cancer Res; 70(7); 2575–8. ©2010 AACR. Background the current state of the cell or tissue as characterized by pro- teomic and metabolic measurements (3). The past five years have seen an exponential increase in In this review we describe and evaluate the various tech- technological development to explore the genome and the niques that have recently become available to study the ki- proteome to understand the molecular basis of disease. How- nomes of clinical tissue samples. -
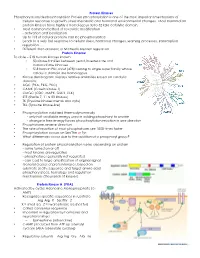
Protein Kinases Phosphorylation/Dephosphorylation Protein Phosphorylation Is One of the Most Important Mechanisms of Cellular Re
Protein Kinases Phosphorylation/dephosphorylation Protein phosphorylation is one of the most important mechanisms of cellular responses to growth, stress metabolic and hormonal environmental changes. Most mammalian protein kinases have highly a homologous 30 to 32 kDa catalytic domain. • Most common method of reversible modification - activation and localization • Up to 1/3 of cellular proteins can be phosphorylated • Leads to a very fast response to cellular stress, hormonal changes, learning processes, transcription regulation .... • Different than allosteric or Michealis Menten regulation Protein Kinome To date – 518 human kinases known • 50 kinase families between yeast, invertebrate and mammaliane kinomes • 518 human PKs, most (478) belong to single super family whose catalytic domain are homologous. • Kinase dendrogram displays relative similarities based on catalytic domains. • AGC (PKA, PKG, PKC) • CAMK (Casein kinase 1) • CMGC (CDC, MAPK, GSK3, CLK) • STE (Sterile 7, 11 & 20 kinases) • TK (Tryosine kinases memb and cyto) • TKL (Tyrosine kinase-like) • Phosphorylation stabilized thermodynamically - only half available energy used in adding phosphoryl to protein - change in free energy forces phosphorylation reaction in one direction • Phosphatases reverse direction • The rate of reaction of most phosphatases are 1000 times faster • Phosphorylation occurs on Ser/The or Tyr • What differences occur due to the addition of a phosphoryl group? • Regulation of protein phosphorylation varies depending on protein - some turned on or off -

Kipho: Malaria Parasite Kinome and Phosphatome Portal Rajan Pandey†, Pawan Kumar† and Dinesh Gupta*
Database, 2017, 1–5 doi: 10.1093/database/bax063 Original article Original article KiPho: malaria parasite kinome and phosphatome portal Rajan Pandey†, Pawan Kumar† and Dinesh Gupta* Translational Bioinformatics Group, International Centre for Genetic Engineering and Biotechnology, Aruna Asaf Ali Marg, New Delhi 110067, India *Corresponding author: Tel: +91 11 26741007; Fax: +91 11 26742316; Email: [email protected] †The authors contributed equally to this work. Citation details: Pandey,R., Kumar,P., Gupta,D. KiPho: malaria parasite kinome and phosphatome portal. Database (2017) Vol. 2017: article ID bax063; doi: 10.1093/database/bax063 Received 30 April 2017; Revised 19 July 2017; Accepted 31 July 2017 Abstract The Plasmodium kinases and phosphatases play an essential role in the regulation of sub- strate reversible-phosphorylation and overall cellular homeostasis. Reversible phosphoryl- ation is one of the key post-translational modifications (PTMs) essential for parasite sur- vival. Thus, a complete and comprehensive information of malarial kinases and phosphatases as a single web resource will not only aid in systematic and better under- standing of the PTMs, but also facilitate efforts to look for novel drug targets for malaria. In the current work, we have developed KiPho, a comprehensive and one step web-based in- formation resource for Plasmodium kinases and phosphatases. To develop KiPho, we have made use of search methods to retrieve, consolidate and integrate predicted as well as annotated information from several publically available web repositories. Additionally, we have incorporated relevant and manually curated data, which will be updated from time to time with the availability of new information. The KiPho (Malaria Parasite Kinome—Phosphatome) resource is freely available at http://bioinfo.icgeb.res.in/kipho. -

Identification of Protein O-Glcnacylation Sites Using Electron Transfer Dissociation Mass Spectrometry on Native Peptides
Identification of protein O-GlcNAcylation sites using electron transfer dissociation mass spectrometry on native peptides Robert J. Chalkleya, Agnes Thalhammerb, Ralf Schoepfera,b, and A. L. Burlingamea,1 aDepartment of Pharmaceutical Chemistry, University of California, 600 16th Street, Genentech Hall, Suite N472, San Francisco, CA 94158; and bLaboratory for Molecular Pharmacology, and Department of Neuroscience, Physiology, and Pharmacology, University College London, Gower Street, London WC1E 6BT, United Kingdom Edited by James A. Wells, University of California, San Francisco, CA, and approved April 15, 2009 (received for review January 12, 2009) Protein O-GlcNAcylation occurs in all animals and plants and is and synaptic plasticity have been found O-GlcNAcylated (8). The implicated in modulation of a wide range of cytosolic and nuclear importance of O-GlcNAcylation is highlighted by the brain-specific protein functions, including gene silencing, nutrient and stress sens- OGT knockout mouse model that displays developmental defects ing, phosphorylation signaling, and diseases such as diabetes and that result in neonatal death (9). Most interestingly, in analogy to Alzheimer’s. The limiting factor impeding rapid progress in decipher- phosphorylation, synaptic activity leads to a dynamic modulation of ing the biological functions of protein O-GlcNAcylation has been the O-GlcNAc levels (10) and is also involved in long-term potentiation inability to easily identify exact residues of modification. We describe and memory (11). a robust, high-sensitivity strategy able to assign O-GlcNAcylation sites Phosphorylation is the most heavily studied regulatory PTM, and of native modified peptides using electron transfer dissociation mass proteomic approaches for global enrichment, detection, and char- spectrometry. -

The Use of Phosphoproteomic Data to Identify Altered Kinases and Signaling Pathways in Cancer
The use of phosphoproteomic data to identify altered kinases and signaling pathways in cancer By Sara Renee Savage Thesis Submitted to the Faculty of the Graduate School of Vanderbilt University in partial fulfillment of the requirements for the degree of MASTER OF SCIENCE in Biomedical Informatics August 10, 2018 Nashville, Tennessee Approved: Bing Zhang, Ph.D. Carlos Lopez, Ph.D. Qi Liu, Ph.D. ACKNOWLEDGEMENTS The work presented in this thesis would not have been possible without the funding provided by the NLM training grant (T15-LM007450) and the support of the Biomedical Informatics department at Vanderbilt. I am particularly indebted to Rischelle Jenkins, who helped me solve all administrative issues. Furthermore, this work is the result of a collaboration between all members of the Zhang lab and the larger CPTAC consortium. I would like to thank the other CPTAC centers for processing the data, and Chen Huang and Suhas Vasaikar in the Zhang lab for analyzing the colon cancer copy number and proteomic data, respectively. All members of the Zhang lab have been extremely helpful in answering any questions I had and offering suggestions on my work. Finally, I would like to acknowledge my mentor, Bing Zhang. I am extremely grateful for his guidance and for giving me the opportunity to work on these projects. ii TABLE OF CONTENTS Page ACKNOWLEDGEMENTS ................................................................................................ ii LIST OF TABLES............................................................................................................ -

Phosphatome Rnai Screen Identifies Eya1 As a Positive Regulator of Hedgehog Signal Transduction
Phosphatome RNAi Screen Identifies Eya1 as a Positive Regulator of Hedgehog Signal Transduction The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation Eisner, Adriana. 2013. Phosphatome RNAi Screen Identifies Eya1 as a Positive Regulator of Hedgehog Signal Transduction. Doctoral dissertation, Harvard University. Citable link http://nrs.harvard.edu/urn-3:HUL.InstRepos:11181213 Terms of Use This article was downloaded from Harvard University’s DASH repository, and is made available under the terms and conditions applicable to Other Posted Material, as set forth at http:// nrs.harvard.edu/urn-3:HUL.InstRepos:dash.current.terms-of- use#LAA Phosphatome RNAi Screen Identifies Eya1 as a Positive Regulator of Hedgehog Signal Transduction A dissertation presented by Adriana Eisner to The Division of Medical Sciences in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the subject of Neurobiology Harvard University Cambridge, Massachusetts June 2013 © 2013 Adriana Eisner All rights reserved. Dissertation Advisor: Dr. Rosalind Segal Adriana Eisner Phosphatome RNAi Screen Identifies Eya1 as a Positive Regulator of Hedgehog Signal Transduction Abstract The Hedgehog (Hh) signaling pathway is vital for vertebrate embryogenesis and aberrant activation of the pathway can cause tumorigenesis in humans. In this study, we used a phosphatome RNAi screen for regulators of Hh signaling to identify a member of the Eyes Absent protein family, Eya1, as a positive regulator of Hh signal transduction. Eya1 is both a phosphatase and transcriptional regulator. Eya family members have been implicated in tumor biology, and Eya1 is highly expressed in a particular subtype of medulloblastoma (MB). -

GSK3 and Its Interactions with the PI3K/AKT/Mtor Signalling Network
Heriot-Watt University Research Gateway GSK3 and its interactions with the PI3K/AKT/mTOR signalling network Citation for published version: Hermida, MA, Kumar, JD & Leslie, NR 2017, 'GSK3 and its interactions with the PI3K/AKT/mTOR signalling network', Advances in Biological Regulation, vol. 65, pp. 5-15. https://doi.org/10.1016/j.jbior.2017.06.003 Digital Object Identifier (DOI): 10.1016/j.jbior.2017.06.003 Link: Link to publication record in Heriot-Watt Research Portal Document Version: Peer reviewed version Published In: Advances in Biological Regulation Publisher Rights Statement: © 2017 Elsevier B.V. General rights Copyright for the publications made accessible via Heriot-Watt Research Portal is retained by the author(s) and / or other copyright owners and it is a condition of accessing these publications that users recognise and abide by the legal requirements associated with these rights. Take down policy Heriot-Watt University has made every reasonable effort to ensure that the content in Heriot-Watt Research Portal complies with UK legislation. If you believe that the public display of this file breaches copyright please contact [email protected] providing details, and we will remove access to the work immediately and investigate your claim. Download date: 27. Sep. 2021 Accepted Manuscript GSK3 and its interactions with the PI3K/AKT/mTOR signalling network Miguel A. Hermida, J. Dinesh Kumar, Nick R. Leslie PII: S2212-4926(17)30124-0 DOI: 10.1016/j.jbior.2017.06.003 Reference: JBIOR 180 To appear in: Advances in Biological Regulation Received Date: 13 June 2017 Accepted Date: 23 June 2017 Please cite this article as: Hermida MA, Dinesh Kumar J, Leslie NR, GSK3 and its interactions with the PI3K/AKT/mTOR signalling network, Advances in Biological Regulation (2017), doi: 10.1016/ j.jbior.2017.06.003. -

Kinase-Inactive ZAP-70 Thymocyte Development By
An Improved Retroviral Gene Transfer Technique Demonstrates Inhibition of CD4− CD8− Thymocyte Development by Kinase-Inactive ZAP-70 This information is current as of September 23, 2021. Takehiko Sugawara, Vincenzo Di Bartolo, Tadaaki Miyazaki, Hiromitsu Nakauchi, Oreste Acuto and Yousuke Takahama J Immunol 1998; 161:2888-2894; ; http://www.jimmunol.org/content/161/6/2888 Downloaded from References This article cites 49 articles, 28 of which you can access for free at: http://www.jimmunol.org/content/161/6/2888.full#ref-list-1 http://www.jimmunol.org/ Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication by guest on September 23, 2021 *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 1998 by The American Association of Immunologists All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. An Improved Retroviral Gene Transfer Technique Demonstrates Inhibition of CD42CD82 Thymocyte Development by Kinase-Inactive ZAP-701 Takehiko Sugawara,* Vincenzo Di Bartolo,§ Tadaaki Miyazaki,‡ Hiromitsu Nakauchi,* Oreste Acuto,§ and Yousuke Takahama2*† ZAP-70 is a Syk family tyrosine kinase that plays an essential role in initiating TCR signals. -

A-Kinase Anchoring Protein–Calcineurin Signaling in Long-Term Depression of Gabaergic Synapses
2650 • The Journal of Neuroscience, February 6, 2013 • 33(6):2650–2660 Cellular/Molecular A-Kinase Anchoring Protein–Calcineurin Signaling in Long-Term Depression of GABAergic Synapses Matthieu Dacher, Shawn Gouty, Steven Dash, Brian M. Cox, and Fereshteh S. Nugent Department of Pharmacology, Uniformed Services University of the Health Sciences, Bethesda, Maryland 20814 The postsynaptic scaffolding A-kinase anchoring protein 79/150 (AKAP79/150) signaling complex regulates excitatory synaptic trans- mission and strength through tethering protein kinase A (PKA), PKC, and calcineurin (CaN) to the postsynaptic densities of neurons (Sanderson and Dell’Acqua, 2011), but its role in inhibitory synaptic transmission and plasticity is unknown. Using immunofluorescence and whole-cell patch-clamp recording in rat midbrain slices, we show that activation of postsynaptic D2-like family of dopamine (DA) receptor in the ventral tegmental area (VTA) induces long-term depression (LTD) of GABAergic synapses on DA neurons through an inositol triphosphate receptor-mediated local rise in postsynaptic Ca 2ϩ and CaN activation accompanied by PKA inhibition, which requires AKAP150 as a bridging signaling molecule. Our data also illuminate a requirement for a clathrin-mediated internalization of GABAA receptors in expression of LTDGABA. Moreover, disruption of AKAP–PKA anchoring does not affect glutamatergic synapses onto DA neurons, suggesting that the PKA–AKAP–CaN complex is uniquely situated at GABAA receptor synapses in VTA DA neurons to regulate plasticity associated with GABAA receptors. Drug-induced modulation of GABAergic plasticity in the VTA through such novel signaling mechanisms has the potential to persistently alter the output of individual DA neurons and of the VTA, which may contribute to the reinforcing or addictive properties of drugs of abuse. -

Soluble Epoxide Hydrolase Inhibition Protected Against Angiotensin II
www.nature.com/scientificreports OPEN Soluble Epoxide Hydrolase Inhibition Protected against Angiotensin II-induced Adventitial Received: 6 April 2017 Accepted: 29 June 2017 Remodeling Published online: 31 July 2017 Chi Zhou, Jin Huang, Qing Li, Jiali Nie, Xizhen Xu & Dao Wen Wang Epoxyeicosatrienoic acids (EETs), the metabolites of cytochrome P450 epoxygenases derived from arachidonic acid, exert important biological activities in maintaining cardiovascular homeostasis. Soluble epoxide hydrolase (sEH) hydrolyzes EETs to less biologically active dihydroxyeicosatrienoic acids. However, the efects of sEH inhibition on adventitial remodeling remain inconclusive. In this study, the adventitial remodeling model was established by continuous Ang II infusion for 2 weeks in C57BL/6 J mice, before which sEH inhibitor 1-trifuoromethoxyphenyl-3-(1-propionylpiperidin-4-yl) urea (TPPU) was administered by gavage. Adventitial remodeling was evaluated by histological analysis, western blot, immunofuorescent staining, calcium imaging, CCK-8 and transwell assay. Results showed that Ang II infusion signifcantly induced vessel wall thickening, collagen deposition, and overexpression of α-SMA and PCNA in aortic adventitia, respectively. Interestingly, these injuries were attenuated by TPPU administration. Additionally, TPPU pretreatment overtly prevented Ang II-induced primary adventitial fbroblasts activation, characterized by diferentiation, proliferation, migration, and collagen synthesis via Ca2+-calcineurin/NFATc3 signaling pathway in vitro. In summary, our results suggest that inhibition of sEH could be considered as a novel therapeutic strategy to treat adventitial remodeling related disorders. Aorta is composed of three tunicae: intima, media and adventitia. Te roles of intima and media on vascular functions have been extensively studied, while the contribution of adventitia to vascular functions was recently recognized.