Internal Limiting Membrane Peeling in Macular Surgery-Morphological And
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Meet the Choroid N Optometric Management Joe Pizzimenti, OD, FAAO O Scientific Advisory Boards [email protected] N Zeiss N Zeavision N Thrombogenics N Genentech
10/29/19 Financial Disclosures o Honoraria n Review of Optometry Meet The Choroid n Optometric Management Joe Pizzimenti, OD, FAAO o Scientific Advisory Boards [email protected] n Zeiss n Zeavision n Thrombogenics n Genentech Financial Disclosures Goals for This Course o Consulting Fees n Zeiss o Functional anatomy review n Zeavision n Choroid n Maculogix o Choroid examination and evaluation o Proprietary Interests o Case examples n None o Interactive o Stockholder: Zeavision Questions? 1 10/29/19 The Choroid The Choroid: Structure, o Located between the Function, and Evaluation sclera and the RPE n Extends from ora serrata to optic nerve o Pigmented/vascular tissue .75mm thick o Nourishes the RPE n Choroiocapillaris designed to leak o Absorbs light that passes through retina The Choriod RPE Bruch’s Membrane thickness o Loose connective tissue o Basal lamina of RPE o Melanocytes o Anterior collagenous o Choriocapillaris layer Mel. n Fenestrated endothelium o Elastic layer allows diffusion of o Posterior collagenous proteins CC layer n S__________ regulation o Basal lamina of CC BM n High blood flow endothelium n Very little O-2 extracted, o Contamination of so high venous O-2 Bruch’s can result in sclera d________, CNVM Nourishing the Retina Choroid Microstructure o 2 main sources of blood supply to retina: • Choriocapillaris o Choroidal BVs n Supplies outer retinal • Sattler’s layer layers, including PRs o CRA • Haller’s layer n 4 branches nourish inner retina • Supra - choroid n Run radially toward fovea 2 10/29/19 Imaging the Vascular Layers Imaging the Choroid of the Choroid WHAT IS ENHANCED Imaging the Choroid-EDI DEPTH OCT IMAGING? • EDI-OCT • Enhanced-depth imaging (EDI) OCT modifies the standard technique of image acquisition to better reveal the structural details of the choroid. -

Vitreous and Developmental Vitreoretinopathies Kevin R
CHAPTER 3 Vitreous and Developmental Vitreoretinopathies Kevin R. Tozer, Kenneth M. P. Yee, and J. Sebag Invisible (Fig. 3.1) by design, vitreous was long unseen the central vitreous and adjacent to the anterior cortical as important in the physiology and pathology of the eye. gel. HA molecules have a different distribution from col- Recent studies have determined that vitreous plays a sig- lagen, being most abundant in the posterior cortical gel nificant role in ocular health (1) and disease (1,2), includ- with a gradient of decreasing concentration centrally ing a number of important vitreoretinal disorders that and anteriorly (6,7). arise from abnormal embryogenesis and development. Both collagen and HA are synthesized during child- Vitreous embryology is presented in detail in Chapter 1. hood. Total collagen content in the vitreous gel remains Notable is that primary vitreous is filled with blood ves- at about 0.05 mg until the third decade (8). As collagen sels during the first trimester (Fig. 3.2). During the second does not appreciably increase during this time but the trimester, these vessels begin to disappear as the second- size of the vitreous does increase with growth, the den- ary vitreous is formed, ultimately resulting in an exqui- sity of collagen fibrils effectively decreases. This could sitely clear gel (Fig. 3.1). The following will review vitreous potentially weaken the collagen network and destabilize development and the congenital disorders that arise from the gel. However, since there is active synthesis of HA abnormalities in hyaloid vessel formation and regression during this time, the dramatic increase in HA concentra- during the primary vitreous stage and biochemical abnor- tion may stabilize the thinning collagen network (9). -

Torpedo Maculopathy at the Site of the Fetal “Bulge”
SMALL CASE SERIES SECTION EDITOR: W. RICHARD GREEN, MD ward the foveola. This defect closely The flat, nonpigmented lesion mea- Torpedo Maculopathy resembles solitary CHRPE but dif- sured 2 mm horizontally and 1 mm at the Site of the fers in its nonrandom macular lo- vertically and was located 4 mm tem- Fetal “Bulge” cation and pointed torpedo shape.5-8 poral to the optic disc (Figure 2). In the few reported cases, there have Toxoplasmosis titer results were been no systemic associations. negative. Observation was advised. Torpedo maculopathy was discov- Herein, we describe 2 cases of tor- ered in 2 children as a pointed-oval pedo maculopathy and speculate as Comment. In 1992, Roseman and retinal pigment epithelial (RPE) de- to its embryogenesis. Gass3 described a 12-year-old boy fect in the temporal macula. This with a small, flat, circumscribed, oval congenital finding could be related Report of Cases. Case 1. On rou- RPE lesion in the temporal macula. to the fetal temporal macular “bulge” tine eye examination, a 3-year-old Additional reports confirmed the con- that normally occurs at 4 to 6 girl with fix-and-follow visual acu- sistent pointed oval configuration and months’ gestation at the same site. ity was discovered to have a tempo- macular location of this condition There are several congenital ral macular RPE defect with a (Table).5-8 Rigotti and associates7 re- anomalies of the RPE, including con- pointed-oval shape directed toward ported 3 cases of asymptomatic tor- genital hypertrophy of the RPE the foveola and hyperpigmented pedo maculopathy in a child and 2 (CHRPE), combined hamartoma of “frayed tail” appearance directed to- adults. -

Numerical Study of the Effect of Vitreous Support on Eye Accommodation
Acta of Bioengineering and Biomechanics Vol. 7, No. 2, 2005 Numerical study of the effect of vitreous support on eye accommodation DARJA LJUBIMOVA, ANDERS ERIKSSON KTH Mechanics, Royal Institute of Technology, Osquars backe 18, SE-100 44 Stockholm, Sweden SVETLANA BAUER Saint-Petersburg State University, Department of Theoretical and Applied Mechanics, Universitetsky pr. 28, 198504 Petergof, Russia The aim of the current work was to extend previously created finite element models of accommodation such as the one by BURD [2] by addition of vitreous. The zonule consisted of anterior and central sets and vitreous was modelled as a linear elastic incompressible body. An inverse method was used to find some important, previously not documented, aspects. The model was found to behave according to the expectations, with results consistent with classical Helmholtz theory. Key words: vitreous support, eye accomodation, finite element models 1. Introduction Accommodation of an eye is the process of adjusting its focus distance to allow near objects to be focused on the retina. According to the widely accepted classical Helmholtz theory [19] and recent research in the accommodative apparatus of an eye [17], in the unaccommodated state the lens is flattened by the passive tension of the zonular fibres which are pulled by the elastic choroid structures. During accommodation the ciliary muscle contracts, thereby slides forward and towards the axis of the eye. It pulls the choroid structures, which causes the tension in the zonular fibres to reduce. With release of the resting tension of the zonular fibers, the elastic capsule reshapes the lens; it becomes more sharply curved and thickens axially. -

Ciliary Body
Ciliary body S.Karmakar HOD Introduction • Ciliary body is the middle part of the uveal tract . It is a ring (slightly eccentric ) shaped structure which projects posteriorly from the scleral spur, with a meridional width varying from 5.5 to 6.5 mm. • It is brown in colour due to melanin pigment. Anteriorly it is confluent with the periphery of the iris (iris root) and anterior part of the ciliary body bounds a part of the anterior chamber angle. Introduction • Posteriorly ciliary body has a crenated or scalloped periphery, known as ora serrata, where it is continuous with the choroid and retina. The ora serrata exhibits forward extensions,known as dentate process, which are well defined on the nasal side and less so temporally. • Ciliary body has a width of approximately 5.9 mm on the nasal side and 6.7 mm on the temporal side. Extension of the ciliary body On the outside of the eyeball, the ciliary body extends from a point about 1.5 mm posterior to the corneal limbus to a point 6.5 to 7.5 mm posterior to this point on the temporal side and 6.5 mm posterior on the nasal side. Parts of ciliary body • Ciliary body, in cross section, is a triangular structure ( in diagram it can be compared as ∆ AOI). Outer side of the triangle (O) is attached with the sclera with suprachoroidal space in between. Anterior side of the triangle (A) forms part of the anterior & posterior chamber. In its middle, the iris is attached. The inner side of the triangle (I) is divided into two parts. -

Nomina Histologica Veterinaria, First Edition
NOMINA HISTOLOGICA VETERINARIA Submitted by the International Committee on Veterinary Histological Nomenclature (ICVHN) to the World Association of Veterinary Anatomists Published on the website of the World Association of Veterinary Anatomists www.wava-amav.org 2017 CONTENTS Introduction i Principles of term construction in N.H.V. iii Cytologia – Cytology 1 Textus epithelialis – Epithelial tissue 10 Textus connectivus – Connective tissue 13 Sanguis et Lympha – Blood and Lymph 17 Textus muscularis – Muscle tissue 19 Textus nervosus – Nerve tissue 20 Splanchnologia – Viscera 23 Systema digestorium – Digestive system 24 Systema respiratorium – Respiratory system 32 Systema urinarium – Urinary system 35 Organa genitalia masculina – Male genital system 38 Organa genitalia feminina – Female genital system 42 Systema endocrinum – Endocrine system 45 Systema cardiovasculare et lymphaticum [Angiologia] – Cardiovascular and lymphatic system 47 Systema nervosum – Nervous system 52 Receptores sensorii et Organa sensuum – Sensory receptors and Sense organs 58 Integumentum – Integument 64 INTRODUCTION The preparations leading to the publication of the present first edition of the Nomina Histologica Veterinaria has a long history spanning more than 50 years. Under the auspices of the World Association of Veterinary Anatomists (W.A.V.A.), the International Committee on Veterinary Anatomical Nomenclature (I.C.V.A.N.) appointed in Giessen, 1965, a Subcommittee on Histology and Embryology which started a working relation with the Subcommittee on Histology of the former International Anatomical Nomenclature Committee. In Mexico City, 1971, this Subcommittee presented a document entitled Nomina Histologica Veterinaria: A Working Draft as a basis for the continued work of the newly-appointed Subcommittee on Histological Nomenclature. This resulted in the editing of the Nomina Histologica Veterinaria: A Working Draft II (Toulouse, 1974), followed by preparations for publication of a Nomina Histologica Veterinaria. -
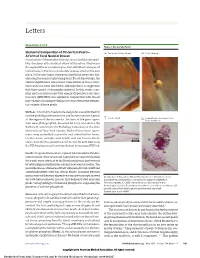
Elemental Composition of Ora Serrata Pearls—A Form of Focal Nodular
Letters RESEARCH LETTER Figure 1. Ora Serrata Pearls Elemental Composition of Ora Serrata Pearls— A Two pearls (50 and 10 µm) B Detailed image A Form of Focal Nodular Drusen Examination of the peripheral fundus may reveal discrete pearl- like structures at the furthest extent of the retina. They occur throughout the ora serrata region, but only where a tongue of retina tissue in the form of a dentate process overlies the pars plana. In the later stages, the pigment epithelial covering is lost, exposing the pearl as a glistening bead. To our knowledge, the clinical significance and precise composition of these struc- tures have not been described, although there is suggestion that they consist of drusenlike material. In this study, scan- ning electron microscopy with energy dispersive x-ray spec- troscopy (SEM-EDS) was applied in conjunction with classic histochemical staining techniques to characterize the elemen- tal content of these pearls. Methods | A formalin-fixed enucleated globe was submitted for routine pathological examination and found to contain 3 pearls C Calcified pearl D Pearl without attachment to the at the region of the ora serrata. Sections of the gross speci- Bruch membrane men were photographed, dissected (R.E.E.), and sent to the Barbara W. Streeten Ocular Pathology Laboratory at the State University of New York Upstate Medical University. Speci- mens were embedded in paraffin and submitted for hema- toxylin-eosin, periodic acid–Schiff, and van Gieson elastic 200 µm stains. A section was placed on a carbon stub for SEM-EDS using the FEI Aspex personal scanning electron microscope (FEI Inc). -

The Horizontal Raphe of the Human Retina and Its Watershed Zones
vision Review The Horizontal Raphe of the Human Retina and its Watershed Zones Christian Albrecht May * and Paul Rutkowski Department of Anatomy, Medical Faculty Carl Gustav Carus, TU Dresden, 74, 01307 Dresden, Germany; [email protected] * Correspondence: [email protected] Received: 24 September 2019; Accepted: 6 November 2019; Published: 8 November 2019 Abstract: The horizontal raphe (HR) as a demarcation line dividing the retina and choroid into separate vascular hemispheres is well established, but its development has never been discussed in the context of new findings of the last decades. Although factors for axon guidance are established (e.g., slit-robo pathway, ephrin-protein-receptor pathway) they do not explain HR formation. Early morphological organization, too, fails to establish a HR. The development of the HR is most likely induced by the long posterior ciliary arteries which form a horizontal line prior to retinal organization. The maintenance might then be supported by several biochemical factors. The circulation separate superior and inferior vascular hemispheres communicates across the HR only through their anastomosing capillary beds resulting in watershed zones on either side of the HR. Visual field changes along the HR could clearly be demonstrated in vascular occlusive diseases affecting the optic nerve head, the retina or the choroid. The watershed zone of the HR is ideally protective for central visual acuity in vascular occlusive diseases but can lead to distinct pathological features. Keywords: anatomy; choroid; development; human; retina; vasculature 1. Introduction The horizontal raphe (HR) was first described in the early 1800s as a horizontal demarcation line that extends from the macula to the temporal Ora dividing the temporal retinal nerve fiber layer into a superior and inferior half [1]. -

Enzymatic Vitreolysis Using Reengineered Vibrio Mimicus–Derived Collagenase
Molecular Vision 2021; 27:125-141 <http://www.molvis.org/molvis/v27/125> © 2021 Molecular Vision Received 19 February 2020 | Accepted 30 March 2021 | Published 1 April 2021 Enzymatic vitreolysis using reengineered Vibrio mimicus–derived collagenase Mithun Santra,1 Maryada Sharma,1,4 Deeksha Katoch,2 Sahil Jain,2 Uma Nahar Saikia,3 Mangat R. Dogra,2 Manni Luthra-Guptasarma1 1Department of Immunopathology, Postgraduate Institute of Medical Education and Research, Chandigarh, India; 2Department of Ophthalmology, Postgraduate Institute of Medical Education and Research, Chandigarh, India; 3Department of Histopathology, Postgraduate Institute of Medical Education and Research, Chandigarh, India; 4Department of Otolaryngology and Head & Neck surgery, Postgraduate Institute of Medical Education and Research, Chandigarh, India Purpose: Collagen is a key player contributing to vitreoelasticity and vitreoretinal adhesions. Molecular reorganization causes spontaneous weakening of these adhesions with age, resulting in the separation of the posterior hyaloid membrane (PHM) from the retina in what is called complete posterior vitreous detachment (PVD). Incomplete separation of the posterior hyaloid or tight adherence or both can lead to retinal detachment, vitreomacular traction syndrome, or epiretinal membrane formation, which requires surgical intervention. Pharmacological vitrectomy has the potential of avoiding surgical vitrectomy; it is also useful as an adjunct during retinal surgery to induce PVD. Previously studied enzymatic reagents, such as collagenase derived from Clostridium histolyticum, are nonspecific and potentially toxic. We studied a novel collagenase from Vibrio mimicus (VMC) which remains active (VMA), even after deletion of 51 C-terminal amino acids. To limit the activity of VMA to the vitreous cavity, a fusion construct (inhibitor of hyaluronic acid-VMA [iHA-VMA]) was made in which a 12-mer peptide (iHA, which binds to HA) was fused to the N-terminus of VMA. -

The Nervous System: General and Special Senses
18 The Nervous System: General and Special Senses PowerPoint® Lecture Presentations prepared by Steven Bassett Southeast Community College Lincoln, Nebraska © 2012 Pearson Education, Inc. Introduction • Sensory information arrives at the CNS • Information is “picked up” by sensory receptors • Sensory receptors are the interface between the nervous system and the internal and external environment • General senses • Refers to temperature, pain, touch, pressure, vibration, and proprioception • Special senses • Refers to smell, taste, balance, hearing, and vision © 2012 Pearson Education, Inc. Receptors • Receptors and Receptive Fields • Free nerve endings are the simplest receptors • These respond to a variety of stimuli • Receptors of the retina (for example) are very specific and only respond to light • Receptive fields • Large receptive fields have receptors spread far apart, which makes it difficult to localize a stimulus • Small receptive fields have receptors close together, which makes it easy to localize a stimulus. © 2012 Pearson Education, Inc. Figure 18.1 Receptors and Receptive Fields Receptive Receptive field 1 field 2 Receptive fields © 2012 Pearson Education, Inc. Receptors • Interpretation of Sensory Information • Information is relayed from the receptor to a specific neuron in the CNS • The connection between a receptor and a neuron is called a labeled line • Each labeled line transmits its own specific sensation © 2012 Pearson Education, Inc. Interpretation of Sensory Information • Classification of Receptors • Tonic receptors -

Computer-Animated Model of Accommodation and Theory of Reciprocal Zonular Action
Clinical Ophthalmology Dovepress open access to scientific and medical research Open Access Full Text Article ORIGINAL RESEARCH Computer-animated model of accommodation and theory of reciprocal zonular action Daniel B Goldberg1,2 Abstract: This report presents a computer-animated model of the structures of accommodation 1Ophthalmology Department, Drexel based on new understanding of the anatomy of the zonular apparatus integrated with current College of Medicine, Philadelphia, PA, understanding of the mechanism of accommodation. Analysis of this model suggests a new, 2 Eye Physicians, Little Silver, NJ, USA consolidated theory of the mechanism of accommodation including a new theory of reciprocal zonular action. A three-dimensional animated model of the eye in accommodation and disac- commodation was produced in collaboration with an experienced medical animator. Current understanding of the anatomy of the zonule and the attachments of the vitreous zonule to the anterior hyaloid membrane is incomplete. Recent studies have demonstrated three components of the vitreous zonule: (1) anterior vitreous zonule (previously “hyalocapsular” zonule), which attaches the ciliary plexus in the valleys of the ciliary processes to the anterior hyaloid mem- brane in the region medial to the ciliary body and Weiger’s ligament; (2) intermediate vitreous zonule, which attaches the ciliary plexus to the anterior hyaloid peripherally; and (3) posterior vitreous zonule, which creates a sponge-like ring at the attachment zone that anchors the pars plana zonules. The pars plana zonules attach posteriorly to the elastic choroid above the ora serrata. Analysis of the computer-animated model demonstrates the synchronized movements of the accommodative structures in accommodation and disaccommodation. -

Anatomy of the Globe 09 Hermann D. Schubert Basic and Clinical
Anatomy of the Globe 09 Hermann D. Schubert Basic and Clinical Science Course, AAO 2008-2009, Section 2, Chapter 2, pp 43-92. The globe is the home of the retina (part of the embryonic forebrain, i.e.neural ectoderm and neural crest) which it protects, nourishes, moves or holds in proper position. The retinal ganglion cells (second neurons of the visual pathway) have axons which form the optic nerve (a brain tract) and which connect to the lateral geniculate body of the brain (third neurons of the visual pathway with axons to cerebral cortex). The transparent media of the eye are: tear film, cornea, aqueous, lens, vitreous, internal limiting membrane and inner retina. Intraocular pressure is the pressure of the aqueous and vitreous compartment. The aqueous compartment is comprised of anterior(200ul) and posterior chamber(60ul). Aqueous and vitreous compartments communicate across the anterior cortical gel of the vitreous which seen from up front looks like a donut and is called the “annular diffusional gap.” The globe consists of two superimposed spheres, the corneal radius measuring 8mm and the scleral radius 12mm. The superimposition creates an external scleral sulcus, the outflow channels anterior to the scleral spur fill the internal scleral sulcus. Three layers or ocular coats are distinguished: the corneal scleral coat, the uvea and neural retina consisting of retina and pigmentedepithelium. The coats and components of the inner eye are held in place by intraocular pressure, scleral rigidity and mechanical attachments between the layers. The corneoscleral coat consists of cornea, sclera, lamina cribrosa and optic nerve sheath.