The Deep Atlantic Ocean
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
North America Other Continents
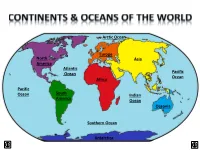
Arctic Ocean Europe North Asia America Atlantic Ocean Pacific Ocean Africa Pacific Ocean South Indian America Ocean Oceania Southern Ocean Antarctica LAND & WATER • The surface of the Earth is covered by approximately 71% water and 29% land. • It contains 7 continents and 5 oceans. Land Water EARTH’S HEMISPHERES • The planet Earth can be divided into four different sections or hemispheres. The Equator is an imaginary horizontal line (latitude) that divides the earth into the Northern and Southern hemispheres, while the Prime Meridian is the imaginary vertical line (longitude) that divides the earth into the Eastern and Western hemispheres. • North America, Earth’s 3rd largest continent, includes 23 countries. It contains Bermuda, Canada, Mexico, the United States of America, all Caribbean and Central America countries, as well as Greenland, which is the world’s largest island. North West East LOCATION South • The continent of North America is located in both the Northern and Western hemispheres. It is surrounded by the Arctic Ocean in the north, by the Atlantic Ocean in the east, and by the Pacific Ocean in the west. • It measures 24,256,000 sq. km and takes up a little more than 16% of the land on Earth. North America 16% Other Continents 84% • North America has an approximate population of almost 529 million people, which is about 8% of the World’s total population. 92% 8% North America Other Continents • The Atlantic Ocean is the second largest of Earth’s Oceans. It covers about 15% of the Earth’s total surface area and approximately 21% of its water surface area. -

Marine Ecology Progress Series 600:21
Vol. 600: 21–39, 2018 MARINE ECOLOGY PROGRESS SERIES Published July 30 https://doi.org/10.3354/meps12663 Mar Ecol Prog Ser OPENPEN ACCESSCCESS Short-term processing of ice algal- and phytoplankton- derived carbon by Arctic benthic communities revealed through isotope labelling experiments Anni Mäkelä1,*, Ursula Witte1, Philippe Archambault2 1School of Biological Sciences, University of Aberdeen, Aberdeen AB24 3UU, UK 2Département de biologie, Québec Océan, Université Laval, Québec, QC G1V 0A6, Canada ABSTRACT: Benthic ecosystems play a significant role in the carbon (C) cycle through remineral- ization of organic matter reaching the seafloor. Ice algae and phytoplankton are major C sources for Arctic benthic consumers, but climate change-mediated loss of summer sea ice is predicted to change Arctic marine primary production by increasing phytoplankton and reducing ice algal contributions. To investigate the impact of changing algal C sources on benthic C processing, 2 isotope tracing experiments on 13C-labelled ice algae and phytoplankton were conducted in the North Water Polynya (NOW; 709 m depth) and Lancaster Sound (LS; 794 m) in the Canadian Arc- tic, during which the fate of ice algal (CIA) and phytoplankton (CPP) C added to sediment cores was traced over 4 d. No difference in sediment community oxygen consumption (SCOC, indicative of total C turnover) between the background measurements and ice algal or phytoplankton cores was found at either site. Most of the processed algal C was respired, with significantly more CPP than CIA being released as dissolved inorganic C at both sites. Macroinfaunal uptake of algal C was minor, but bacterial assimilation accounted for 33−44% of total algal C processing, with no differences in bacterial uptake of CPP and CIA found at either site. -

Impact of Large-Scale Natural Physical Disturbance on the Diversity of Deep-Sea North Atlantic Nematodes
MARINE ECOLOGY PROGRESS SERIES Vol. 214: 121–126, 2001 Published April 26 Mar Ecol Prog Ser Impact of large-scale natural physical disturbance on the diversity of deep-sea North Atlantic nematodes P. J. D. Lambshead1,*, J. Tietjen2, A. Glover1, T. Ferrero1, D. Thistle3, A. J. Gooday4 1Department of Zoology, The Natural History Museum, London SW7 5BD, United Kindom 2Department of Biology, City College of New York, New York, New York 10031 & Division of Invertebrates, American Museum of Natural History, New York, New York 10024, USA 3Department of Oceanography, Florida State University, Tallahassee, Florida 32306-3048, USA 4Southampton Oceanography Centre, George Deacon Division for Ocean Processes, Empress Dock, Southampton SO14 3ZH, United Kindom ABSTRACT: Nematode alpha diversity from 3 physically disturbed sites in the deep North Atlantic was compared with reference sites. Nematode diversity at the HEBBLE benthic storm site was statis- tically, and significantly, lower than at reference sites. Nematode diversity at the Madeira Abyssal Plain site, which was subject to a turbidite dated at 930 BP, also showed a significantly lower diver- sity than reference sites. However, limited data suggest that diversity was not low at a Venezuela Basin turbidite site. The difference in nematode diversity between the 2 turbidite sites is ascribed to a long term change in sediment conditions at the Madeira site. The Venezuela Basin turbidite site has a sedimentation rate greater than the Maderia site by 1 to 2 orders of magnitude, and this was reflected in the sediment profiles. Another possibility is that the Venezuela Basin turbidite is consid- erably older, by at least 1000 yr, than the Madeira turbidite, allowing more time for recolonisation. -

Geologists Suggest Horseshoe Abyssal Plain May Be Start of a Subduction Zone 8 May 2019, by Bob Yirka
Geologists suggest Horseshoe Abyssal Plain may be start of a subduction zone 8 May 2019, by Bob Yirka against one another. Over by the Iberian Peninsula, the opposite appears to be happening—the African and Eurasian plates are pulling apart as the former creeps east toward the Americas. Duarte noted that back in 2012, other researchers conducting seismic wave tests found what appeared to be a dense mass of unknown material beneath the epicenter of the 1969 quake. Some in the field suggested it could be the start of a subduction zone. Then, last year, another team conducted high-resolution imaging of the area and also found evidence of the mass, confirming that it truly existed. Other research has shown that the area just above the mass experiences routine tiny earthquakes. Duarte suggests the evidence to date indicates that the bottom of the plate is peeling away. This could happen, he explained, due to serpentinization in which water percolates through plate fractures and reacts with material beneath the surface, resulting A composite image of the Western hemisphere of the in the formation of soft green minerals. The soft Earth. Credit: NASA mineral layer, he suggests, is peeling away. And if that is the case, then it is likely the area is in the process of creating a subduction zone. He reports that he and his team members built models of their A team of geologists led by João Duarte gave a ideas and that they confirmed what he suspected. presentation at this past month's European The earthquakes were the result of the process of Geosciences Union meeting that included a birthing a new subduction zone. -

Coastal and Marine Ecological Classification Standard (2012)
FGDC-STD-018-2012 Coastal and Marine Ecological Classification Standard Marine and Coastal Spatial Data Subcommittee Federal Geographic Data Committee June, 2012 Federal Geographic Data Committee FGDC-STD-018-2012 Coastal and Marine Ecological Classification Standard, June 2012 ______________________________________________________________________________________ CONTENTS PAGE 1. Introduction ..................................................................................................................... 1 1.1 Objectives ................................................................................................................ 1 1.2 Need ......................................................................................................................... 2 1.3 Scope ........................................................................................................................ 2 1.4 Application ............................................................................................................... 3 1.5 Relationship to Previous FGDC Standards .............................................................. 4 1.6 Development Procedures ......................................................................................... 5 1.7 Guiding Principles ................................................................................................... 7 1.7.1 Build a Scientifically Sound Ecological Classification .................................... 7 1.7.2 Meet the Needs of a Wide Range of Users ...................................................... -

Inside:The Pleistocene Cooling Built
THE PLEISTOCENE COOLING BUILT CHALLENGER MOUND, A DEEP-WATER CORAL MOUND IN THE NE ATLANTIC: INSIDE: SYNTHESIS FROM IODP EXPEDITION 307 PLUS: COUNCIL’S COMMENTS 2011 SPRING SEPM SECTION MEETINGS Special Publication #95 Cenozoic Carbonate Systems of Australasia Edited by: William A. Morgan, Annette D. George, Paul M. (Mitch) Harris, Julie A. Kupecz, and J.F. (Rick) Sarg The Cenozoic carbonate systems of Australasia are the product of a diverse assortment of depositional and post- depositional processes, reflecting the interplay of eustasy, tectonics (both plate and local scale), climate, and evolutionary trends that influenced their initiation and development. These systems, which comprise both land- attached and isolated platforms, were initiated in a wide variety of tectonic settings (including rift, passive margin, and arc-related) and under warm and cool-water conditions where, locally, siliciclastic input affected their development. The lithofacies, biofacies, growth morphology, diagenesis, and hydrocarbon reservoir potential of these systems are products of these varying influences. The studies reported in this volume range from syntheses of tectonic and depositional factors influencing carbonate deposition and controls on reservoir formation and petroleum system development, to local studies from the South China Sea, Indonesia, Kalimantan, Malaysia, the Marion Plateau, the Philippines, Western Australia, and New Caledonia that incorporate outcrop and subsurface data, including 3-D seismic imaging of carbonate platforms and facies, to understand the interplay of factors affecting the development of these systems under widely differing circumstances. This volume will be of importance to geoscientists interested in the variability of Cenozoic carbonate systems and the factors that controlled their formation, and to those wanting to understand the range of potential hydrocarbon reservoirs discovered in these carbonates and the events that led to favorable reservoir and trap development. -

Phytoplankton As Key Mediators of the Biological Carbon Pump: Their Responses to a Changing Climate
sustainability Review Phytoplankton as Key Mediators of the Biological Carbon Pump: Their Responses to a Changing Climate Samarpita Basu * ID and Katherine R. M. Mackey Earth System Science, University of California Irvine, Irvine, CA 92697, USA; [email protected] * Correspondence: [email protected] Received: 7 January 2018; Accepted: 12 March 2018; Published: 19 March 2018 Abstract: The world’s oceans are a major sink for atmospheric carbon dioxide (CO2). The biological carbon pump plays a vital role in the net transfer of CO2 from the atmosphere to the oceans and then to the sediments, subsequently maintaining atmospheric CO2 at significantly lower levels than would be the case if it did not exist. The efficiency of the biological pump is a function of phytoplankton physiology and community structure, which are in turn governed by the physical and chemical conditions of the ocean. However, only a few studies have focused on the importance of phytoplankton community structure to the biological pump. Because global change is expected to influence carbon and nutrient availability, temperature and light (via stratification), an improved understanding of how phytoplankton community size structure will respond in the future is required to gain insight into the biological pump and the ability of the ocean to act as a long-term sink for atmospheric CO2. This review article aims to explore the potential impacts of predicted changes in global temperature and the carbonate system on phytoplankton cell size, species and elemental composition, so as to shed light on the ability of the biological pump to sequester carbon in the future ocean. -

Chapter 5 Water Levels and Flow
253 CHAPTER 5 WATER LEVELS AND FLOW 1. INTRODUCTION The purpose of this chapter is to provide the hydrographer and technical reader the fundamental information required to understand and apply water levels, derived water level products and datums, and water currents to carry out field operations in support of hydrographic surveying and mapping activities. The hydrographer is concerned not only with the elevation of the sea surface, which is affected significantly by tides, but also with the elevation of lake and river surfaces, where tidal phenomena may have little effect. The term ‘tide’ is traditionally accepted and widely used by hydrographers in connection with the instrumentation used to measure the elevation of the water surface, though the term ‘water level’ would be more technically correct. The term ‘current’ similarly is accepted in many areas in connection with tidal currents; however water currents are greatly affected by much more than the tide producing forces. The term ‘flow’ is often used instead of currents. Tidal forces play such a significant role in completing most hydrographic surveys that tide producing forces and fundamental tidal variations are only described in general with appropriate technical references in this chapter. It is important for the hydrographer to understand why tide, water level and water current characteristics vary both over time and spatially so that they are taken fully into account for survey planning and operations which will lead to successful production of accurate surveys and charts. Because procedures and approaches to measuring and applying water levels, tides and currents vary depending upon the country, this chapter covers general principles using documented examples as appropriate for illustration. -

Geography Notes.Pdf
THE GLOBE What is a globe? a small model of the Earth Parts of a globe: equator - the line on the globe halfway between the North Pole and the South Pole poles - the northern-most and southern-most points on the Earth 1. North Pole 2. South Pole hemispheres - half of the earth, divided by the equator (North & South) and the prime meridian (East and West) 1. Northern Hemisphere 2. Southern Hemisphere 3. Eastern Hemisphere 4. Western Hemisphere continents - the largest land areas on Earth 1. North America 2. South America 3. Europe 4. Asia 5. Africa 6. Australia 7. Antarctica oceans - the largest water areas on Earth 1. Atlantic Ocean 2. Pacific Ocean 3. Indian Ocean 4. Arctic Ocean 5. Antarctic Ocean WORLD MAP ** NOTE: Our textbooks call the “Southern Ocean” the “Antarctic Ocean” ** North America The three major countries of North America are: 1. Canada 2. United States 3. Mexico Where Do We Live? We live in the Western & Northern Hemispheres. We live on the continent of North America. The other 2 large countries on this continent are Canada and Mexico. The name of our country is the United States. There are 50 states in it, but when it first became a country, there were only 13 states. The name of our state is New York. Its capital city is Albany. GEOGRAPHY STUDY GUIDE You will need to know: VOCABULARY: equator globe hemisphere continent ocean compass WORLD MAP - be able to label 7 continents and 5 oceans 3 Large Countries of North America 1. United States 2. Canada 3. -

Sediments, Fauna and the Dispersal O Radionuclides at the N.E. Atlantic Dumpsite for Low-Level Radioactive Wast
NETHERLANDS INSTITUTE FOR SEA RESEARC Sediments, fauna and the dispersal o radionuclides at the N.E. Atlantic dumpsite for low-level radioactive wast INIS-mf--11394 Report of the Dutch DORA program **? M.M. Rutgers van der Loeff & M.S.S. Lavaleye Front cover: Physiographic Diagram of the North Atlantic by B. C. Heezen and M. Tharp, Copyrighted © 1984 by Marie Thpro, published by permission of Marie Tharp. The posi- tion of the dumpsite is indicated. Tl.e deep water in the eastern basin is ventilated from the south. Back cover: Rat-tail fish Coryphaenoides armatus caught at the dumpsite. Sediments, fauna, and the dispersal of radionuclides at the N.E. Atlantic dumpsite for low-level radioactive waste Report of the Dutch DORA program M.M. Rutgers van der Loeff & M.S.S. Lavaleye Netherlands Institute for Sea Research, P.O. Box 59,NL 1790 AB DEN BURG, TEXEL 1986 SEDIMENTS, FAUNA, AND THE DISPERSAL OF RADIONUCLIDES AT THE NE ATLANTIC DUMPSITE FOR LOW-LEVEL RADIOACTIVE WASTE Final report of the Dutch DORA program MM. RUTGERS VAN DER LOEFF AND M.S.S. LAVALEYE NIOZ, P.O. Box 59, 1790 AB Den Burg, Texel, The Netherlands. CONTENTS 1. Introduction 1.1. Research at the NEA dumpsite: CRESP and the DORA project 1.2. Acknowledgments 1.3. Source term: the radionuclides of interest 2. Material and methods 2.1. Cruises to the dumpsite 2.2. Sampling scheme 3. Geochemistry 3.1. Introduction 3.2. Methods 3.3. Recent sedimentological history 3.4. Elemental and mineralogical composition of the sediment 3.4.1. -

EXPLORING DEEP SEA HYDROTHERMAL VENTS on EARTH and OCEAN WORLDS. P. Sobron1,2, L. M. Barge3, the Invader Team. 1Impossible Sensing, St
52nd Lunar and Planetary Science Conference 2021 (LPI Contrib. No. 2548) 2505.pdf EXPLORING DEEP SEA HYDROTHERMAL VENTS ON EARTH AND OCEAN WORLDS. P. Sobron1,2, L. M. Barge3, the InVADER Team. 1Impossible Sensing, St. Louis, MO ([email protected]) 2SETI Institute, Mtn. View, CA, , 3Jet Propulsion Laboratory, Pasadena, CA The Mission: InVADER (In-situ Vent Analysis precipitates showing exposed minerals and organic Divebot for Exobiology Research, Figure 4.1, content. The UNOLS ROV will use our coring tool and https://invader-mission.org/) is NASA’s most advanced its own manipulator and cameras to take ground truth subsea sensing payload, a tightly integrated imaging and samples as part of the InVADER deplotment. laser Raman spectroscopy/laser-induced breakdown In contrast to existing methods, InVADER allows spectroscopy/laser induced native fluorescence in-situ, autonomous, non-destructive measurements of instrument capable of in-situ, rapid, long-term these vent characteristics. InVADER will fill these gaps, underwater analyses of vent fluid and precipitates. and advance readiness in vent exploration on Earth and Such analyses are critical for finding and studying Ocean Worlds by simplifying operational strategies for life and life’s precursors at vent systems on Ocean identifying and characterizing seafloor environments. Worlds. To demonstrate the scientific potential and We will use statistical analysis tools for the fusion of functionality of the instrument, in July 2021 our team multi-sensor datasets, and develop real-time science will deploy InVADER on the Ocean Observatories data evaluation and payload control routines to Initiative’s (OOI) Regional Cabled Array (RCA), a establish, and then validate, adaptive science operations power/data distribution network off the Oregon coast, at strategies that maximize science return in a mission-like the underwater hydrothermal systems of Axial scenario. -

Provenance and Pathways of Late Quaternary Turbidites in the Deep-Water Agadir Basin, Northwest African Margin
Int J Earth Sci (Geol Rundsch) (2009) 98:721–733 DOI 10.1007/s00531-008-0313-4 ORIGINAL PAPER Provenance and pathways of late Quaternary turbidites in the deep-water Agadir Basin, northwest African margin Michael Frenz Æ Russell B. Wynn Æ Aggeliki Georgiopoulou Æ Vera B. Bender Æ Gayle Hough Æ Douglas G. Masson Æ Peter J. Talling Æ Bryan T. Cronin Received: 29 January 2007 / Accepted: 11 March 2008 / Published online: 28 March 2008 Ó Springer-Verlag 2008 Abstract A series of individual turbidites, correlated volume volcaniclastic turbidites are attributed to a Canary over distances[100 km, are present in the recent fill of the Islands landslide source, while several small mud-domi- Agadir Basin, offshore northwest Africa. The aim here is to nated turbidites are interpreted to be locally sourced from unravel multiple turbidite source areas and flow pathways, hemipelagic-draped seamounts (e.g. Turbidite AB10). and show how turbidite provenance studies contribute to Finally, Turbidite AB1 (*1 ka) is only present in the interpretation of flow processes. Agadir Basin turbidites are western Agadir Basin, and is linked to recent ‘‘re-activa- sourced from four main areas, with the majority originating tion’’ of the Sahara Slide headwall. The muddy suspension from the siliciclastic Morocco Shelf; their sand-mud dis- clouds of three large-volume flows, all linked to large-scale tribution is strongly controlled by flow sediment volume, landslides, have covered huge areas of seafloor and flowed with relatively low-volume flows dying out within the along or even slightly upslope for long distances. It is Agadir Basin and large-volume flows bypassing significant proposed that northeastwards-flowing bottom currents have sediment volumes to basins further downslope.