Aenigmachannidae, a New Family of Snakehead Fishes
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

§4-71-6.5 LIST of CONDITIONALLY APPROVED ANIMALS November
§4-71-6.5 LIST OF CONDITIONALLY APPROVED ANIMALS November 28, 2006 SCIENTIFIC NAME COMMON NAME INVERTEBRATES PHYLUM Annelida CLASS Oligochaeta ORDER Plesiopora FAMILY Tubificidae Tubifex (all species in genus) worm, tubifex PHYLUM Arthropoda CLASS Crustacea ORDER Anostraca FAMILY Artemiidae Artemia (all species in genus) shrimp, brine ORDER Cladocera FAMILY Daphnidae Daphnia (all species in genus) flea, water ORDER Decapoda FAMILY Atelecyclidae Erimacrus isenbeckii crab, horsehair FAMILY Cancridae Cancer antennarius crab, California rock Cancer anthonyi crab, yellowstone Cancer borealis crab, Jonah Cancer magister crab, dungeness Cancer productus crab, rock (red) FAMILY Geryonidae Geryon affinis crab, golden FAMILY Lithodidae Paralithodes camtschatica crab, Alaskan king FAMILY Majidae Chionocetes bairdi crab, snow Chionocetes opilio crab, snow 1 CONDITIONAL ANIMAL LIST §4-71-6.5 SCIENTIFIC NAME COMMON NAME Chionocetes tanneri crab, snow FAMILY Nephropidae Homarus (all species in genus) lobster, true FAMILY Palaemonidae Macrobrachium lar shrimp, freshwater Macrobrachium rosenbergi prawn, giant long-legged FAMILY Palinuridae Jasus (all species in genus) crayfish, saltwater; lobster Panulirus argus lobster, Atlantic spiny Panulirus longipes femoristriga crayfish, saltwater Panulirus pencillatus lobster, spiny FAMILY Portunidae Callinectes sapidus crab, blue Scylla serrata crab, Samoan; serrate, swimming FAMILY Raninidae Ranina ranina crab, spanner; red frog, Hawaiian CLASS Insecta ORDER Coleoptera FAMILY Tenebrionidae Tenebrio molitor mealworm, -

Influence of Dietary Protein Levels on Growth, Feed Utilization and Carcass Composition of Snakehead, Parachanna Obscura (Günther, 1861) Fingerlings
Vol. 5(5), pp. 71-77, May, 2013 DOI: 10.5897/IJFA12.074 International Journal of Fisheries and ISSN 2006-9839 ©2013 Academic Journals Aquaculture http://www.academicjournals.org/IJFA Full Length Research Paper Influence of dietary protein levels on growth, feed utilization and carcass composition of snakehead, Parachanna obscura (Günther, 1861) fingerlings Diane N. S. Kpogue 1*, Grace A. Ayanou 2, Ibrahim I. Toko 2, Guy A. Mensah 3 and Emile D. Fiogbe 1 1Unité de Recherches sur les Zones Humides, Département de Zoologie et Génétique, Faculté des Sciences et Techniques, Université d’Abomey - Calavi, B.P. 526 Cotonou, Bénin. 2Unité de Recherche en Aquaculture et Ecotoxicologie Aquatique, Faculté d’Agronomie, Université de Parakou, B.P. 123 Parakou, Bénin. 3Institut National des Recherches Agricoles du Bénin, Centre de Recherches Agricoles d’Agonkanmey.01 BP 884, Recette Principale Cotonou 01, Bénin. Accepted 12 March, 2013 Five isoenergetic semi-purified diets were formulated to evaluate the effects of dietary crude protein levels on growth and feed utilization of snakehead, Parachanna obscura (4.08 ± 0.07 g). Experimental diets were formulated to contain graded levels of crude protein (CP; 30, 40, 45, 50 and 60 g/100 g of diet). Fish feed on the tested diet in triplicate for 45 days. Seventy fingerlings were stocked per a 225 L cement tank. Growth performances and nutrient utilization parameters of fingerlings fed different diets varied significantly (P < 0.05) and the highest growth performance and nutrient utilization were obtained with fish fed on a 50% CP diet. The relationship between the dietary CP and specific growth rate (SGR) indicated that protein requirements of P. -

Snakeheadsnepal Pakistan − (Pisces,India Channidae) PACIFIC OCEAN a Biologicalmyanmar Synopsis Vietnam
Mongolia North Korea Afghan- China South Japan istan Korea Iran SnakeheadsNepal Pakistan − (Pisces,India Channidae) PACIFIC OCEAN A BiologicalMyanmar Synopsis Vietnam and Risk Assessment Philippines Thailand Malaysia INDIAN OCEAN Indonesia Indonesia U.S. Department of the Interior U.S. Geological Survey Circular 1251 SNAKEHEADS (Pisces, Channidae)— A Biological Synopsis and Risk Assessment By Walter R. Courtenay, Jr., and James D. Williams U.S. Geological Survey Circular 1251 U.S. DEPARTMENT OF THE INTERIOR GALE A. NORTON, Secretary U.S. GEOLOGICAL SURVEY CHARLES G. GROAT, Director Use of trade, product, or firm names in this publication is for descriptive purposes only and does not imply endorsement by the U.S. Geological Survey. Copyrighted material reprinted with permission. 2004 For additional information write to: Walter R. Courtenay, Jr. Florida Integrated Science Center U.S. Geological Survey 7920 N.W. 71st Street Gainesville, Florida 32653 For additional copies please contact: U.S. Geological Survey Branch of Information Services Box 25286 Denver, Colorado 80225-0286 Telephone: 1-888-ASK-USGS World Wide Web: http://www.usgs.gov Library of Congress Cataloging-in-Publication Data Walter R. Courtenay, Jr., and James D. Williams Snakeheads (Pisces, Channidae)—A Biological Synopsis and Risk Assessment / by Walter R. Courtenay, Jr., and James D. Williams p. cm. — (U.S. Geological Survey circular ; 1251) Includes bibliographical references. ISBN.0-607-93720 (alk. paper) 1. Snakeheads — Pisces, Channidae— Invasive Species 2. Biological Synopsis and Risk Assessment. Title. II. Series. QL653.N8D64 2004 597.8’09768’89—dc22 CONTENTS Abstract . 1 Introduction . 2 Literature Review and Background Information . 4 Taxonomy and Synonymy . -

(Pellets) on Survival Rate of Juvenile Snakehead Fish (Channa Striata) on Adaptation in Happa Experiments
Effect of Artificial Feed Feeding Engineering (pellets) on Survival Rate of Juvenile snakehead fish (Channa striata) on Adaptation in Happa Experiments I Made Kawan 1*, Semara Edi 2, Dewa Sadguna 3 Universitas Warmadewa, Denpasar-Bali, Indonesia {[email protected] 1, [email protected] 2, [email protected] 3} Abstract. Effect of Artificial Feed Feeding Engineering (pellets) on Survival Rate of Juvenile snakehead fish (Channa striata) on Adaptation in Happa Experiments were carried out at the Laboratory of Aquatic Resources Development, Faculty of Agriculture, Warmadewa University, Denpasar. The purpose of this study was to determine the engineering of artificial feeding which could provide the highest juvenile survival rate for snakehead fish. Snakehead fish (Channa striata) is a type of freshwater fish that lives in public waters, including rivers, swamps and lakes. One of the methods for seed propagation is by applying hatchery technology using artificial feeding techniques (PF500 pellets) combined with natural feed for water fleas (Dapnia sp) and silk worms (Tubifex). It is hoped that the combination of feeding is expected to be able to determine the artificial feeding (pellet) engineering that provides adequate nutritional needs so as to provide the highest survival rate in juvenile growth of snakehead fish at the adaptation stage of passive food habits. The method used in the engineering research of giving pellets is an experimental method using a completely randomized design (CRD) which consists of 6 treatments and 3 replications. The treatments in this experiment consisted of: A. Giving 100% pellets; B. Providing a combination of pellets 75% with Dapnia sp 12.5% and Tubifex 12.5%; C. -

Absorption of Food Along the Guts of Parachanna Obscura and Clarias Gariepinus
American Journal of Environmental Sciences Original Research Paper Absorption of Food Along the Guts of Parachanna obscura and Clarias gariepinus 1Eunice O. Idowu, 1Adewumi A. Adejoke, 1Josephine B. Edward, 1,2Dolapo F. Odeyemi, 1Folasade A. Ola-Oladimeji and 1Akintayo Joshua Oluwaseun 1Department of Zoology and Environmental Biology, Faculty of Science, Ekiti State University, Ado Ekiti, Ekiti State, Nigeria 2Department of Science Laboratory Technology, (Biotechnology Option), Faculty of Science, Ekiti State University, Ado Ekiti, Ekiti State, Nigeria Article history Abstract: The knowledge of feeding habit and absorption of food in the gut Received: 12-11-2019 of fish is important for fish farmers to boost their production and meet the Revised: 10-01-2020 ever increasing demand for man consumption. The carbohydrate, protein Accepted: 24-02-2020 and lipid contents of the food ingested and their absorption in the intestine of Parachanna obscura and Clarias gareipinus inhabiting Ogbese River in Corresponding Author: Eunice O. Idowu Ekiti State were investigated. For Parachanna obscura, total protein of the Department of Zoology and ingested food was 64.54%, total carbohydrate 20.20% while the total lipid Environmental Biology, in the stomach content was 6.12% of organic matter. The total percentages Faculty of Science, Ekiti State of the food absorbed were 76.4% protein, 35.9% carbohydrate and 25.2% University, Ado Ekiti, Ekiti lipid. For Clarias gariepinus, total protein of the ingested food was 60.87%, State, Nigeria total carbohydrate 30.42% while total lipid in the stomach content was Tell: 08039422005 3.30% of organic matter. The total percentages of the food absorbed were Email: [email protected] 68.0% protein, 45.8% carbohydrate and 24.5% lipid. -

Snakehead Free
FREE SNAKEHEAD PDF Anthony Horowitz | 388 pages | 04 Sep 2008 | Penguin Putnam Inc | 9780142412121 | English | New York, NY, United States Snakehead Fish: Invasive Predators in North America - Owlcation - Education Northern Snakeheads were first confirmed in Pennsylvania in July after an angler caught and preserved two from Snakehead acre Meadow Lake in Philadelphia County. PFBC biologists confirmed they were indeed Northern Snakeheads and Snakehead additional ones from the lake. The lake is part of a maze of interconnected embayments and tidal sloughs and the Commission believes additional Snakeheads Snakehead present elsewhere in the system, including the nearby lower Schuylkill and Delaware rivers. Northern Snakeheads first drew attention in Snakehead mid-Atlantic region in when a pair were discovered in a Maryland pond. Northern Snakeheads are a predatory fish and will compete Snakehead other fish species for forage and habitat. It Snakehead too early to say what impact the presence of Snakeheads will have on other species already in Meadow Snakehead and other waters. Partly based on the experiences of other states, Commission biologists have concluded that there is no practical method for eradicating Snakeheads from Meadow Lake and given the nature of the waterway Snakeheads may have already accessed adjoining waters. The PFBC has Snakehead that while it will continue to monitor the pond and surrounding waterways, it will take no concerted effort to eliminate the species. All Snakeheads are distinguished by their torpedo shaped body, long dorsal and anal fins without spines, and toothed jaws. Northern Snakeheads are typically Snakehead by a flattened, pointy head with long lower jaws. Northern Snakeheads can be confused with native Pennsylvania species like the Bowfin and Burbot. -

2020 Issn: 2456-8643 a Review of the General A
International Journal of Agriculture, Environment and Bioresearch Vol. 5, No. 01; 2020 ISSN: 2456-8643 A REVIEW OF THE GENERAL ASPECTS OF REPRODUCTION OF A THREATENED FRESH WATER FISHSPECIES IN BENIN: PARACHANNA OBSCURA (GÜNTER, 1861) Mahunan Tobias Césaire Azon1*, Edmond Sossoukpe1, Juste Vital Vodounnou1, Antoine Chikou2, Guy Apollinaire Mensah3 and Emile Didier Fiogbe1 1Research Laboratory on Wetlands, Department of Zoology, Faculty of Sciences and Techniques, University of Abomey-Calavi, BP: 526 Cotonou (Benin) 2Laboratory of Hydrobiology and Aquaculture, University of Abomey-Calavi, Faculty of Agricultural Sciences 01 BP 526 Cotonou (Benin) 3National Institute of Agricultural Research of Benin, Center for Agricultural Research of Agonkanmey01 BP 884, Recette Principale Cotonou 01, Benin https://doi.org/10.35410/IJAEB.2020.5476 ABSTRACT Parachanna obscura is a fresh water fish belonging to Channidae family that is recognized as snakehead fish. Its first sexual maturity height is 17.5 cm TL in females and 23.4 cm TL in males. Sexual dimorphism based on observation of the genital papilla is difficult to recognize. The fish species is carnivorous with rapid growth rate. For further evaluation of its aquaculture potential in Benin Republic, the current study aims to make a review on the reproductive aspect of P. obscura. Its resistance, rapid growth and high economic and nutritional values represent important potential for aquaculture because it is highly appreciated by the West African population in general. The natural stock of P. obscurais currently threatened to disappearance in Benin due to fishing pressures that lead to overexploitation. A successful rearing of P. obscura in controlled medium could ensure not only the preservation and improvement of natural stocks but also the production of food fish for sustainable aquaculture. -

Eu Non-Native Organism Risk Assessment Scheme
EU NON-NATIVE SPECIES RISK ANALYSIS – RISK ASSESSMENT Channa spp. EU NON-NATIVE ORGANISM RISK ASSESSMENT SCHEME Name of organism: Channa spp. Author: Deputy Direction of Nature (Spanish Ministry of Agriculture and Fisheries, Food and Environment) Risk Assessment Area: Europe Draft version: December 2016 Peer reviewed by: David Almeida. GRECO, Institute of Aquatic Ecology, University of Girona, 17003 Girona, Spain ([email protected]) Date of finalisation: 23/01/2017 Peer reviewed by: Quim Pou Rovira. Coordinador tècnic del LIFE Potamo Fauna. Plaça dels estudis, 2. 17820- Banyoles ([email protected]) Final version: 31/01/2017 1 EU NON-NATIVE SPECIES RISK ANALYSIS – RISK ASSESSMENT Channa spp. EU CHAPPEAU QUESTION RESPONSE 1. In how many EU member states has this species been recorded? List An adult specimen of Channa micropeltes was captured on 22 November 2012 at Le them. Caldane (Colle di Val d’Elsa, Siena, Tuscany, Italy) (43°23′26.67′′N, 11°08′04.23′′E).This record of Channa micropeltes, the first in Europe (Piazzini et al. 2014), and it constitutes another case of introduction of an alien species. Globally, exotic fish are a major threat to native ichthyofauna due to their negative impact on local species (Crivelli 1995, Elvira 2001, Smith and Darwall 2006, Gozlan et al. 2010, Hermoso and Clavero 2011). Channa argus in Slovakia (Courtenay and Williams, 2004, Elvira, 2001) Channa argus in Czech Republic (Courtenay and Williams 2004, Elvira, 2001) 2. In how many EU member states has this species currently None established populations? List them. 3. In how many EU member states has this species shown signs of None invasiveness? List them. -

A Fossil Climbing Perch from the Oligocene of Tibet Helps Solve The
Science Bulletin 64 (2019) 455–463 Contents lists available at ScienceDirect Science Bulletin journal homepage: www.elsevier.com/locate/scib Article Into Africa via docked India: a fossil climbing perch from the Oligocene of Tibet helps solve the anabantid biogeographical puzzle ⇑ ⇑ Feixiang Wu a,b, , Dekui He c, , Gengyu Fang d, Tao Deng a,b,d a Key Laboratory of Vertebrate Evolution and Human Origins, Institute of Vertebrate Paleontology and Paleoanthropology, Chinese Academy of Sciences, Beijing 100044, China b Center for Excellence in Life and Paleoenvironment, Chinese Academy of Sciences, Beijing 100101, China c Key Laboratory of Aquatic Biodiversity and Conservation, Institute of Hydrobiology, Chinese Academy of Sciences, Wuhan 430072, China d College of Earth Sciences, University of Chinese Academy of Sciences, Beijing 100049, China article info abstract Article history: The northward drift of the Indian Plate and its collision with Eurasia have profoundly impacted the evo- Received 7 March 2019 lutionary history of the terrestrial organisms, especially the ones along the Indian Ocean rim. Climbing Received in revised form 22 March 2019 perches (Anabantidae) are primary freshwater fishes showing a disjunct south Asian-African distribution, Accepted 22 March 2019 but with an elusive paleobiogeographic history due to the lack of fossil evidence. Here, based on an Available online 28 March 2019 updated time-calibrated anabantiform phylogeny integrating a number of relevant fossils, the divergence between Asian and African climbing perches is estimated to have occurred in the middle Eocene (ca. Keywords: 40 Ma, Ma: million years ago), a time when India had already joined with Eurasia. The key fossil lineage Climbing perches is yEoanabas, the oldest anabantid known so far, from the upper Oligocene of the Tibetan Plateau. -

Genetic Variation of Striped Snakehead (Channa Striatus Bloch, 1793) Populations Using Random Amplified Polymorphic DNA (RAPD) Markers
Vol. 6(5), pp. 363-372, May 2014 DOI: 10.5897/IJBC2013.0649 Article Number: C7F0F7D44255 International Journal of Biodiversity ISSN 2141-243X Copyright © 2014 and Conservation Author(s) retain the copyright of this article http://www.academicjournals.org/IJBC Full Length Research Paper Genetic variation of striped snakehead (Channa striatus Bloch, 1793) populations using random amplified polymorphic DNA (RAPD) markers Ajaz Ali Bhat1, M. A. Haniffa1*, M. James Milton1, Bilal Ahmad Paray1, P. R. Divya2 and A. Gopalakrishnan2 1Centre for Aquaculture Research and Extension, St. Xavier’s College, Palayamkottai Tamilnadu-627 002, India. 2National Bureau of Fish Genetic Resources (NBFGR), Cochin Unit, CMFRI Campus, Kochi- 682 018, India. Receive 02 November, 2013; Accepted 15 April, 2014 Random amplified polymorphic DNA-polymerase chain reaction (RAPD-PCR) was applied to analyze the genetic variation among Channa striatus collected from four geographically distant locations in India. After initial PCR screening, nine random oligodecamers viz. OPA10, OPA11, OPA15, OPAC01, OPAC03, OPAC05, OPAC07, OPAC09 and OPAC19 which generated the RAPD profile for the four C. striatus populations were selected. Amplification using these nine primers resulted in fragments ranging in length between 309-3029 bp assigned to 87 loci. Estimates of Nei’s (1978) unbiased genetic distance (D) values ranged from 0.3242-0.6320. Unweighted pair group method with arithmetic mean (UPGMA) dendrogram constructed on the basis of genetic distance revealed very close genetic relationship among C. striatus populations of river Tamirabarani (Tamil Nadu) and river Periyar (Kerala). Both populations from river Tamirabarani and Periyar were found to be genetically closer to fish populations from Kolleru Lake (Andhra Pradesh) whereas C. -
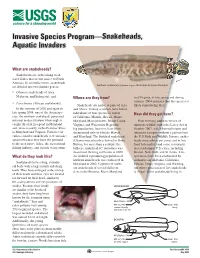
Invasive Species Program—Snakeheads, Aquatic Invaders
Invasive Species Program—Snakeheads, Aquatic Invaders What are snakeheads? Snakeheads are airbreathing fresh- water fishes that are not native to North America. In scientific terms, snakeheads are divided into two distinct genera: Northern snakehead (Channa argus) Illustration by Susan Trammell • Channa (snakeheads of Asia, Malaysia, and Indonesia); and Where are they from? and Virginia, in late spring and during summer 2004 indicates that this species is • Parachanna (African snakeheads). Snakeheads are native to parts of Asia likely reproducing there. In the summer of 2002 and again in and Africa. Fishery scientists have found late spring 2004, one of the Asian spe- individuals of four species in waters How did they get here? cies, the northern snakehead, generated of California, Florida, Hawaii, Maine, national media attention when anglers Maryland, Massachusetts, Rhode Island, Prior to being added to the list of caught this fish in a pond in Maryland Virginia, and Wisconsin. Reproduc- injurious wildlife under the Lacey Act in and, more recently, in the Potomac River ing populations, however, have been October 2002, which banned import and in Maryland and Virginia. Fisheries sci- documented only in Florida, Hawaii, interstate transport without a permit from entists consider snakeheads to be invasive and Maryland. The blotched snakehead the U.S. Fish and Wildlife Service, snake- species because they have the potential (Channa maculata) has thrived in Oahu, heads were sold in pet stores and in live to threaten native fishes, the recreational Hawaii, for more than a century; the food fish markets and some restaurants fishing industry, and aquatic ecosystems. bullseye snakehead (C. -

Ichthyofaunal Diversity of Tasek Bera Ramsar Site, Pahang, Peninsular Malaysia
Journal of Wildlife and Parks, 30: 27-43 (2015) ICHTHYOFAUNAL DIVERSITY OF TASEK BERA RAMSAR SITE, PAHANG, PENINSULAR MALAYSIA M. Fahmi-Ahmad, Syed A. Rizal & Amirrudin B.A* Pusat Pengajian Sains Marin dan Sekitaran, Universiti Malaysia Terengganu, 21030 Kuala Terengganu, Terengganu, Malaysia *Corresponding author’s email: [email protected] ABSTRACT Tasek Bera Ramsar Site is an alluvial swamp with small area of open water and is an im- portant and unique freshwater ecosystem in Peninsular Malaysia. Research on fish fauna of Tasek Bera began more than 60 years ago. A recent survey has collected a total of 52 species belonging to 20 families of freshwater fishes bringing the total number of fish species known to date to 127 species from 30 families. Thirteen species are newly recorded for the ecosystem, comprising of mainly small, cryptic and bottom dwelling species that can be found among riparian vegetation or root system of riparian plants. The remaining species are common and can be found widely distributed throughout Peninsular Malaysia. The ich- thyofaunas of Tasek Bera are dominated by Cyprinidae (51 species), Siluridae (12 species), followed by Bagridae (nine species) and Osphronemidae (nine species). The available data showed that fish diversity of Tasek Bera ecosystem is highly diverse. Future research needs to consider the peripheral habitat in order to obtain a complete picture of the ichthyofauna diversity of Tasek Bera Ramsar Site for sound conservation and management. Keywords: inland freshwater fishes, lacustrine habitat, blackwater, Ramsar Site, checklist Received (06-November-14); Accepted (01-July-15); Available online (21-October-15) Citation: Fahmi-Ahmad, M., Rizal, S.A.