Guidelines for Use of Canine Blood Components
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

27. Clinical Indications for Cryoprecipitate And
27. CLINICAL INDICATIONS FOR CRYOPRECIPITATE AND FIBRINOGEN CONCENTRATE Cryoprecipitate is indicated in the treatment of fibrinogen deficiency or dysfibrinogenaemia.1 Fibrinogen concentrate is licenced for the treatment of acute bleeding episodes in patients with congenital fibrinogen deficiency, including afibrinogenaemia and hypofibrinogenaemia,2 and is currently funded under the National Blood Agreement. Key messages y Fibrinogen is an essential component of the coagulation system, due to its role in initial platelet aggregation and formation of a stable fibrin clot.3 y The decision to transfuse cryoprecipitate or fibrinogen concentrate to an individual patient should take into account the relative risks and benefits.3 y The routine use of cryoprecipitate or fibrinogen concentrate is not advised in medical or critically ill patients.2,4 y Cryoprecipitate or fibrinogen concentrate may be indicated in critical bleeding if fibrinogen levels are not maintained using FFP. In the setting of major obstetric haemorrhage, early administration of cryoprecipitate or fibrinogen concentrate may be necessary.3 Clinical implications y The routine use of cryoprecipitate or fibrinogen concentrate in medical or critically ill patients with coagulopathy is not advised. The underlying causes of coagulopathy should be identified; where transfusion is considered necessary, the risks and benefits should be considered for each patient. Specialist opinion is advised for the management of disseminated intravascular coagulopathy (MED-PP18, CC-PP7).2,4 y Cryoprecipitate or fibrinogen concentrate may be indicated in critical bleeding if fibrinogen levels are not maintained using FFP. In patients with critical bleeding requiring massive transfusion, suggested doses of blood components is 3-4g (CBMT-PP10)3 in adults or as per the local Massive Transfusion Protocol. -

Stem Cells and Artificial Substitutes Could Ease the Dependence on Blood Donations
OUTLOOK BLOOD DOING WITHOUT DONORS Stem cells and artificial substitutes could ease the dependence on blood donations. ANDREW BAKER BY ELIE DOLGIN S12 | NATURE | VOL 549 | 28 SEPTEMBER©2017 Ma c2017millan Publishers Li mited, part of Spri nger Nature. All ri ghts reserved. ©2017 Mac millan Publishers Li mited, part of Spri nger Nature. All ri ghts reserved. BLOOD OUTLOOK ach year, at about 13,000 collection centres worldwide, phlebotomists stick needles in the veins of healthy vol- unteers and amass in excess of 110 million donations of blood. The volume collected is enough to fill 20 Olympic- sized swimming pools — but it’s nowhere near to meeting the medical demand for whole blood or its components. To fill the gap, an enterprising group of stem-cell biologists and bio- Eengineers hopes to produce a safe, reliable and bottomless supply of on-demand blood substitutes in the laboratory. According to Robert Lanza, a pioneer of stem cell therapies and head IMAGES IWM VIA GETTY CHETWYN/ SGT. of global regenerative medicine at Astellas Pharma in Marlborough, Massachusetts, current technologies are not yet ready to compete with the real stuff. “We’re not going to put blood banks out of business any time soon,” he says. But in the near future, artificial blood products could be approved for use when transfusions are not otherwise an option, such as during combat or in people with a religious objection to receiving blood transfusions. And therapies that rely on reprogrammed stem cells to produce components of blood might also help transfusion centres to relieve shortages or to avoid donor-derived contamination. -

Hemolytic Disease of the Newborn
Intensive Care Nursery House Staff Manual Hemolytic Disease of the Newborn INTRODUCTION and DEFINITION: Hemolytic Disease of the Newborn (HDN), also known as erythroblastosis fetalis, isoimmunization, or blood group incompatibility, occurs when fetal red blood cells (RBCs), which possess an antigen that the mother lacks, cross the placenta into the maternal circulation, where they stimulate antibody production. The antibodies return to the fetal circulation and result in RBC destruction. DIFFERENTIAL DIAGNOSIS of hemolytic anemia in a newborn infant: -Isoimmunization -RBC enzyme disorders (e.g., G6PD, pyruvate kinase deficiency) -Hemoglobin synthesis disorders (e.g., alpha-thalassemias) -RBC membrane abnormalities (e.g., hereditary spherocytosis, elliptocytosis) -Hemangiomas (Kasabach Merritt syndrome) -Acquired conditions, such as sepsis, infections with TORCH or Parvovirus B19 (anemia due to RBC aplasia) and hemolysis secondary to drugs. ISOIMMUNIZATION A. Rh disease (Rh = Rhesus factor) (1) Genetics: Rh positive (+) denotes presence of D antigen. The number of antigenic sites on RBCs varies with genotype. Prevalence of genotype varies with the population. Rh negative (d/d) individuals comprise 15% of Caucasians, 5.5% of African Americans, and <1% of Asians. A sensitized Rh negative mother produces anti-Rh IgG antibodies that cross the placenta. Risk factors for antibody production include 2nd (or later) pregnancies*, maternal toxemia, paternal zygosity (D/D rather than D/d), feto-maternal compatibility in ABO system and antigen load. (2) Clinical presentation of HDN varies from mild jaundice and anemia to hydrops fetalis (with ascites, pleural and pericardial effusions). Because the placenta clears bilirubin, the chief risk to the fetus is anemia. Extramedullary hematopoiesis (due to anemia) results in hepatosplenomegaly. -

Association Between ABO and Duffy Blood Types and Circulating Chemokines and Cytokines
Genes & Immunity (2021) 22:161–171 https://doi.org/10.1038/s41435-021-00137-5 ARTICLE Association between ABO and Duffy blood types and circulating chemokines and cytokines 1 2 3 4 5 6 Sarah C. Van Alsten ● John G. Aversa ● Loredana Santo ● M. Constanza Camargo ● Troy Kemp ● Jia Liu ● 4 7 8 Wen-Yi Huang ● Joshua Sampson ● Charles S. Rabkin Received: 11 February 2021 / Revised: 30 April 2021 / Accepted: 17 May 2021 / Published online: 8 June 2021 This is a U.S. government work and not under copyright protection in the U.S.; foreign copyright protection may apply 2021, corrected publication 2021 Abstract Blood group antigens are inherited traits that may play a role in immune and inflammatory processes. We investigated associations between blood groups and circulating inflammation-related molecules in 3537 non-Hispanic white participants selected from the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial. Whole-genome scans were used to infer blood types for 12 common antigen systems based on well-characterized single-nucleotide polymorphisms. Serum levels of 96 biomarkers were measured on multiplex fluorescent bead-based panels. We estimated marker associations with blood type using weighted linear or logistic regression models adjusted for age, sex, smoking status, and principal components of p 1234567890();,: 1234567890();,: population substructure. Bonferroni correction was used to control for multiple comparisons, with two-sided values < 0.05 considered statistically significant. Among the 1152 associations tested, 10 were statistically significant. Duffy blood type was associated with levels of CXCL6/GCP2, CXCL5/ENA78, CCL11/EOTAXIN, CXCL1/GRO, CCL2/MCP1, CCL13/ MCP4, and CCL17/TARC, whereas ABO blood type was associated with levels of sVEGFR2, sVEGFR3, and sGP130. -

Blood Product Replacement: Obstetric Hemorrhage
CMQCC OBSTETRIC HEMORRHAGE TOOLKIT Version 2.0 3/24/15 BLOOD PRODUCT REPLACEMENT: OBSTETRIC HEMORRHAGE Richard Lee, MD, Los Angeles County and University of Southern California Medical Center Laurence Shields, MD, Marian Regional Medical Center/Dignity Health Holli Mason, MD, Cedars-Sinai Medical Center Mark Rollins, MD, PhD, University of California, San Francisco Jed Gorlin, MD, Innovative Blood Resources/Memorial Blood Center, St. Paul, Minnesota Maurice Druzin, MD, Lucile Packard Children’s Hospital Stanford University Jennifer McNulty, MD, Long Beach Memorial Medical Center EXECUTIVE SUMMARY • Outcomes are improved with early and aggressive intervention. • Both emergency blood release and massive transfusion protocols should be in place. • In the setting of significant obstetric hemorrhage, resuscitation transfusion should be based on vital signs and blood loss and should not be delayed by waiting for laboratory results. • Calcium replacement will often be necessary with massive transfusion due to the citrate used for anticoagulation in blood products. • During massive transfusion resuscitation, the patient’s arterial blood gas, electrolytes, and core temperature should be monitored to guide clinical management and all transfused fluids should be warmed; direct warming of the patient should be initiated as needed to maintain euthermia and to avoid added coagulopathy. BACKGROUND AND LITERATURE REVIEW After the first several units of packed red blood cells (PRBCs) and in the face of continuing or worsening hemorrhage, aggressive transfusion therapy becomes critical. This report covers the experience with massive transfusion protocols. Lessons from military trauma units as well as civilian experience with motor vehicle accidents and massive obstetric hemorrhage have identified new principles such as earlier use of plasma (FFP/thawed plasma/plasma frozen within 24 hours/liquid plasma) and resuscitation transfusion while laboratory results are pending. -

Platelet-Rich Plasmapheresis: a Meta-Analysis of Clinical Outcomes and Costs
THE jOURNAL OF EXTRA-CORPOREAL TECHNOLOGY Original Article Platelet-Rich Plasmapheresis: A Meta-Analysis of Clinical Outcomes and Costs Chris Brown Mahoney , PhD Industrial Relations Center, Carlson School of Management, University of Minnesota, Minneapolis, MN Keywords: platelet-rich plasmapheresis, sequestration, cardiopulmonary bypass, outcomes, economics, meta-analysis Presented at the American Society of Extra-Corporeal Technology 35th International Conference, April 3-6, 1997, Phoenix, Arizona ABSTRACT Platelet-rich plasmapheresis (PRP) just prior to cardiopulmonary bypass (CPB) surgery is used to improve post CPB hemostasis and to minimize the risks associated with exposure to allogeneic blood and its components. Meta-analysis examines evidence ofPRP's impact on clinical outcomes by integrating the results across published research studies. Data on clinical outcomes was collected from 20 pub lished studies. These outcomes, DRG payment rates, and current national average costs were used to examine the impact of PRP on costs. This study provides evidence that the use of PRP results in improved clinical outcomes when compared to the identical control groups not receiving PRP. These improved clinical out comes result in subsequent lower costs per patient in the PRP groups. All clinical outcomes analyzed were improved: blood product usage, length of stay, intensive care stay, time to extu bation, incidence of cardiovascular accident, and incidence of reoperation. The most striking differences occur in use of all blood products, particularly packed red blood cells. This study provides an example of how initial expenditure on technology used during CPB results in overall cost savings. Estimated cost savings range from $2,505.00 to $4,209.00. -
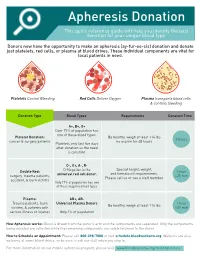
Apheresis Donation This Quick Reference Guide Will Help You Identify the Best Donation for Your Unique Blood Type
Apheresis Donation This quick reference guide will help you identify the best donation for your unique blood type. Donors now have the opportunity to make an apheresis (ay-fur-ee-sis) donation and donate just platelets, red cells, or plasma at blood drives. These individual components are vital for local patients in need. Platelets Control Bleeding Red Cells Deliver Oxygen Plasma transports blood cells & controls bleeding Donation Type Blood Types Requirements Donation Time A+, B+, O+ Over 75% of population has one of these blood types. Platelet Donation: Be healthy, weigh at least 114 lbs 2 hours cancer & surgery patients no aspirin for 48 hours Platelets only last five days after donation so the need is constant. O-, O+, A-, B- Special height, weight, Double Red: O-Negative is the 1 hour and hematocrit requirements. surgery, trauma patients, universal red cell donor. +25 min Please call us or see a staff member accident, & burn victims Only 17% of population has one of these negative blood types Plasma: AB+, AB- Trauma patients, burn Universal Plasma Donors 1 hour Be healthy, weigh at least 114 lbs victims, & patients with +30 min serious illness or injuries Only 4% of population How Apheresis works: Blood is drawn from the donor’s arm and the components are separated. Only the components being donated are collected while the remaining components are safely returned to the donor How to Schedule an Appointment: Please call 800-398-7888 or visit schedule.bloodworksnw.org. Walk-ins are also welcome at some blood drives, so be sure to ask our staff when you stop in. -

Blood Product Modifications: Leukofiltration, Irradiation and Washing
Blood Product Modifications: Leukofiltration, Irradiation and Washing 1. Leukocyte Reduction Definitions and Standards: o Process also known as leukoreduction, or leukofiltration o Applicable AABB Standards, 25th ed. Leukocyte-reduced RBCs At least 85% of original RBCs < 5 x 106 WBCs in 95% of units tested . Leukocyte-reduced Platelet Concentrates: At least 5.5 x 1010 platelets in 75% of units tested < 8.3 x 105 WBCs in 95% of units tested pH≥6.2 in at least 90% of units tested . Leukocyte-reduced Apheresis Platelets: At least 3.0 x 1011 platelets in 90% of units tested < 5.0 x 106 WBCs 95% of units tested pH≥6.2 in at least 90% of units tested Methods o Filter: “Fourth-generation” filters remove 99.99% WBCs o Apheresis methods: most apheresis machines have built-in leukoreduction mechanisms o Less efficient methods of reducing WBC content . Washing, deglycerolizing after thawing a frozen unit, centrifugation . These methods do not meet requirement of < 5.0 x 106 WBCs per unit of RBCs/apheresis platelets. Types of leukofiltration/leukoreduction o “Pre-storage” . Done within 24 hours of collection . May use inline filters at time of collection (apheresis) or post collection o “Pre-transfusion” leukoreduction/bedside leukoreduction . Done prior to transfusion . “Bedside” leukoreduction uses gravity-based filters at time of transfusion. Least desirable given variability in practice and absence of proficiency . Alternatively performed by transfusion service prior to issuing Benefits of leukoreduction o Prevention of alloimmunization to donor HLA antigens . Anti-HLA can mediate graft rejection and immune mediated destruction of platelets o Leukoreduced products are indicated for transplant recipients or patients who are likely platelet transfusion dependent o Prevention of febrile non-hemolytic transfusion reactions (FNHTR) . -

Blood Type and Transplantation & A2 Donor to B Recipient
Page 1 of 2 Blood Type and Transplantation Information for Kidney Transplant Patients Does blood type matter in transplantation? Everyone waiting for a transplant has their blood typed. You will have one of four blood types: O, A, B or AB. Your blood type is determined by the antigens that are present on your blood cells. These antigens are A or B. These antigens will be found both in your blood and on your organs. What antigen does each blood type have? Blood type O Blood type A Blood type B Blood type AB have no have A antigens. have B antigens. have both A O antigens. A B AB and B antigens. How does my body react to antigens? Your body will react to antigens that are different than your own by attacking with antibodies. Antibodies are proteins created by your immune system to attack anything that does not belong. Antibodies are the soldiers in your body’s army protecting you from foreign invasions such as viruses. Unfortunately, the antibodies cannot tell the difference between harmful viruses and beneficial transplanted organs. What blood type will my donor be? Transplants can occur between all blood types. However, when the donor’s blood type is different than yours and there are different antigens being transplanted on your new organ, your antibodies will be triggered and attack the transplanted organ. This is called rejection. Because of this, transplants usually happen between a donor and a recipient of the same blood type. This is called an identical transplant. Can I get an organ from a donor that has a different blood type than mine? Yes! If you do not have antibodies in your body against the antigens that come from the donor, your immune system should not attack the transplanted organ. -

Red Blood Cells, Leukocyte Reduced Covenant and AHS Sites
This document applies to all Red Blood Cells, Leukocyte Reduced Covenant and AHS sites. Class: Human blood component, derived from OTHER NAMES: Packed red blood cells, Packed whole blood cells INTRAVENOUS OTHER ROUTES DIRECT IV Intermittent Continuous Infusion Infusion SC IM OTHER Acceptable No Yes No No No No Routes* * Professionals performing these restricted activities have received authorization from their regulatory college and have the knowledge and skill to perform the skill competently. DESCRIPTION OF PRODUCT: Red Blood Cell (RBC) concentrates are prepared from approximately 480mL whole blood usually collected in 70mL of anticoagulant. The unit is plasma reduced by centrifugation, platelet reduced by either centrifugation or filtration and leukocyte reduced by filtration. RBCs are typically resuspended in approximately 110mL of nutrient. Collected from volunteer donors by the Canadian Blood Services (CBS). Donor is screened and blood is tested for: i. Hepatitis B Surface Antigen (HBsAg) ii. Syphilis iii. Antibodies to Hepatitis B core antigen (HBcore), Hepatitis C Virus (HCV), Human T-cell Lymphotropic Virus (HTLV-1 and 2), Human Immune Deficiency Virus (HIV-1 and 2) iv. Nucleic Acid Testing (NAT) for presence of HCV RNA, HIV-1 RNA and West Nile Virus (WNV) RNA . CBS also tests blood for ABO/Rh and clinically significant antibodies. Some donors are tested for CMV status. RBCs do NOT contain any coagulation factors or platelets. AVAILABILITY: . Contact your local transfusion service/laboratory for ABO and Rh types stocked at your site. ABO compatible (not always identical) may be required if the transfusion service cannot provide sufficient quantities of the patient’s blood group (See ABO compatibility chart at: http://www.albertahealthservices.ca/assets/wf/lab/wf-lab-clin-tm-abo-compatability.pdf). -

Transfusion Guidelines Updated February, 2005
Transfusion Guidelines Updated February, 2005 I. Whole Blood The use of whole blood is not recommended and is not available from the Blood Center. Blood components should be selected according to the patient’s needs. II. Red Blood Cells (RBCs) Transfusion must be completed within 4 hours of issue from the Blood Bank. If transfusion is not begun immediately the RBCs must be stored at 1-6° C or returned to the Blood Bank. (Storage is only allowed in preapproved areas, such as the operating room and several of the critical care areas). The effectiveness of each transfusion should be documented in the medical record. Single unit transfusions of RBCs are often effective. In adult patients, one unit of RBCs will increase the hemoglobin level by approximately 1 g/dl (hematocrit by 3%). A. Acceptable Usage 1. Acute Blood loss exceeding 30-40% of blood volume (pediatric patients - 10-15 ml/Kg) and/or not responding to appropriate volume resuscitation, and/or with ongoing blood loss. 2. Patient is normovolemic and there is evidence to support a need for increased oxygen carrying capacity by the following : a. Hypotension not corrected by adequate volume replacement alone. b. PVO2<25 torr, when SaO2 completely saturated; extraction ratio>50%, VO2<50% of baseline. c. Neonates and young infants less than 56 weeks postmenstrual age with hematocrit < 0.30 and frequent and/or severe apnea/bradycardia, poor weight gain, sustained tachycardia and/or tachypnea or mild respiratory distress. d. Neonates and young infants less than 56 weeks postmenstrual age with hematocrit < 0.35 and moderate respiratory distress or with hematocrit < 0.40 and severe respiratory distress, cyanotic congenital heart disease or receiving extracorporeal membrane oxygenation. -
Laboratory Best Transfusion Practice for Neonates, Infants and Children
Laboratory Best Transfusion Practice for Neonates, Infants and Children This summary guidance should be used in conjunction with the appropriate 20161 and 20122 BSH Guidelines and laboratory SOPs Compatibility testing Neonates and infants < 4 months Obtain neonatal and maternal transfusion history (including any fetal transfusions) for all admissions. Obtain a maternal sample for initial testing where possible, in addition to the patient sample. Red cell selection: no maternal antibodies present Select appropriate group and correct neonatal specification red cells. Group O D-negative red cells may be issued electronically without serological crossmatch. If the laboratory does not universally select group O D-negative red cells for this age group, blood group selection should either be controlled by the LIMS or an IAT crossmatch should be performed using maternal or neonatal plasma to serologically confirm ABO compatibility with both mother and neonate. Red cell selection: where there is maternal antibody Select appropriate group red cells, compatible with maternal alloantibody/ies. An IAT crossmatch should be performed using the maternal plasma. If it is not possible to obtain a maternal sample it is acceptable to crossmatch antigen-negative units against the infant’s plasma. Where paedipacks are being issued from one donor unit it is only necessary to crossmatch the first split pack. Subsequent split packs from this multi-satellite unit can be automatically issued without further crossmatch until the unit expires or the infant is older than 4 months. If packs from a different donor are required, an IAT crossmatch should be performed. Infants and children ≥ 4 months For infants and children from 4 months of age, pre-transfusion testing and compatibility procedures should be performed as recommended for adults.