Reviewtext.Pdf
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Use of Inflated Foley Catheters to Prevent Early Empty Pelvis Complications Following Pelvic Exenteration
ANTICANCER RESEARCH 35: 5543-5546 (2015) Use of Inflated Foley Catheters to Prevent Early Empty Pelvis Complications Following Pelvic Exenteration NICOLAE BACALBASA1, DANA TOMESCU2 and IRINA BALESCU3 1Carol Davila University of Medicine and Pharmacy, Bucharest, Romania; 2Fundeni Clinical Institute, Department of Anaethesia and Critical Care III, Bucharest, Romania; 3Ponderas Hospital, Bucharest, Romania Abstract. For most patients with bulky pelvic tumors, neoadjuvant chemo-irradiation is performed in order to pelvic exenteration remains the only curative option. diminish the local invasion and to transform the patient into Although initially reported as a palliative procedure, a candidate for a less extended resection. However, there nowadays it is rather performed with curative intent. Once are cases in which local invasion persists after neoadjuvant the resectional phase is ended, a large defect will remain at treatment and in which pelvic exenteration is needed. Since the level of the pelvic diaphragm, predisposing to severe Brunschwig reported it for the first time in 1948, this complications which are generically included under the surgical procedure has become the golden-standard for name of empty pelvis syndrome. It has been widely patients with locally invasive pelvic malignancies (3). demonstrated that this type of complication is associated Although the resectional phase has remained practically with severe mortality, even if the patient is free of any pelvic unchanged, the reconstructive phase has undergone multiple recurrence. We present the case of a 56-year-old patient improvements in order to improve the patient quality of submitted to total pelvic exenteration for locally invasive life. However, there are still cases in which a reconstruction previously chemo-irradiated cervical cancer who presented is not possible at the time of resection; in all such cases, a six months after surgery with a severe enteroperineal fistula. -
Detail Report
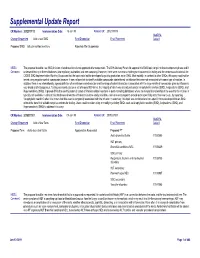
Supplemental Update Report CR Number: 2012319113 Implementation Date: 16-Jan-19 Related CR: 2012319113 MedDRA Change Requested Add a new SMQ Final Disposition Final Placement Code # Proposed SMQ Infusion related reactions Rejected After Suspension MSSO The proposal to add a new SMQ Infusion related reactions is not approved after suspension. The ICH Advisory Panel did approve this SMQ topic to go into the development phase and it Comment: underwent testing in three databases (two regulatory authorities and one company). However, there were numerous challenges encountered in testing and the consensus decision of the CIOMS SMQ Implementation Working Group was that the topic could not be developed to go into production as an SMQ. Most notably, in contrast to other SMQs, this query could not be tested using negative control compounds because it was not possible to identify suitable compounds administered via infusion that were not associated with some type of reaction. In addition, there is no internationally agreed definition of an infusion related reaction and the range of potential reactions associated with the large variety of compounds given by infusion is very broad and heterogenous. Testing was conducted on a set of around 500 terms, the majority of which was already included in Anaphylactic reaction (SMQ), Angioedema (SMQ), and Hypersensitivity (SMQ). It proved difficult to identify potential cases of infusion related reactions in post-marketing databases where the temporal relationship of the event to the infusion is typically not available. In clinical trial databases where this information is more easily available, users are encouraged to provide more specificity about the event, e.g., by reporting “Anaphylactic reaction” when it is known that this event is temporally associated with the infusion. -

Effectiveness of Distal Tibial Osteotomy
Nozaka et al. BMC Musculoskeletal Disorders (2020) 21:31 https://doi.org/10.1186/s12891-020-3061-7 RESEARCH ARTICLE Open Access Effectiveness of distal tibial osteotomy with distraction arthroplasty in varus ankle osteoarthritis Koji Nozaka* , Naohisa Miyakoshi, Takeshi Kashiwagura, Yuji Kasukawa, Hidetomo Saito, Hiroaki Kijima, Shuichi Chida, Hiroyuki Tsuchie and Yoichi Shimada Abstract Background: In highly active older individuals, end-stage ankle osteoarthritis has traditionally been treated using tibiotalar arthrodesis, which provides considerable pain relief. However, there is a loss of ankle joint movement and a risk of future arthrosis in the adjacent joints. Distraction arthroplasty is a simple method that allows joint cartilage repair; however, the results are currently mixed, with some reports showing improved pain scores and others showing no improvement. Distal tibial osteotomy (DTO) without fibular osteotomy is a type of joint preservation surgery that has garnered attention in recent years. However, to our knowledge, there are no reports on DTO with joint distraction using a circular external fixator. Therefore, the purpose of this study was to examine the effect of DTO with joint distraction using a circular external fixator for treating ankle osteoarthritis. Methods: A total of 21 patients with medial ankle arthritis were examined. Arthroscopic synovectomy and a microfracture procedure were performed, followed by angled osteotomy and correction of the distal tibia; the ankle joint was then stabilized after its condition improved. An external fixator was used in all patients, and joint distraction of approximately 5.8 mm was performed. All patients were allowed full weight-bearing walking immediately after surgery. Results: The anteroposterior and lateral mortise angle during weight-bearing, talar tilt angle, and anterior translation of the talus on ankle stress radiography were improved significantly (P < 0.05). -
Musculoskeletal Program CPT Codes and Descriptions
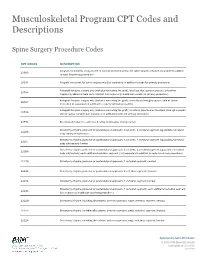
Musculoskeletal Program CPT Codes and Descriptions Spine Surgery Procedure Codes CPT CODES DESCRIPTION Allograft, morselized, or placement of osteopromotive material, for spine surgery only (List separately in addition 20930 to code for primary procedure) 20931 Allograft, structural, for spine surgery only (List separately in addition to code for primary procedure) Autograft for spine surgery only (includes harvesting the graft); local (eg, ribs, spinous process, or laminar 20936 fragments) obtained from same incision (List separately in addition to code for primary procedure) Autograft for spine surgery only (includes harvesting the graft); morselized (through separate skin or fascial 20937 incision) (List separately in addition to code for primary procedure) Autograft for spine surgery only (includes harvesting the graft); structural, bicortical or tricortical (through separate 20938 skin or fascial incision) (List separately in addition to code for primary procedure) 20974 Electrical stimulation to aid bone healing; noninvasive (nonoperative) Osteotomy of spine, posterior or posterolateral approach, 3 columns, 1 vertebral segment (eg, pedicle/vertebral 22206 body subtraction); thoracic Osteotomy of spine, posterior or posterolateral approach, 3 columns, 1 vertebral segment (eg, pedicle/vertebral 22207 body subtraction); lumbar Osteotomy of spine, posterior or posterolateral approach, 3 columns, 1 vertebral segment (eg, pedicle/vertebral 22208 body subtraction); each additional vertebral segment (List separately in addition to code for -

Treatment of Common Hip Fractures: Evidence Report/Technology
This report is based on research conducted by the Minnesota Evidence-based Practice Center (EPC) under contract to the Agency for Healthcare Research and Quality (AHRQ), Rockville, MD (Contract No. HHSA 290 2007 10064 1). The findings and conclusions in this document are those of the authors, who are responsible for its content, and do not necessarily represent the views of AHRQ. No statement in this report should be construed as an official position of AHRQ or of the U.S. Department of Health and Human Services. The information in this report is intended to help clinicians, employers, policymakers, and others make informed decisions about the provision of health care services. This report is intended as a reference and not as a substitute for clinical judgment. This report may be used, in whole or in part, as the basis for the development of clinical practice guidelines and other quality enhancement tools, or as a basis for reimbursement and coverage policies. AHRQ or U.S. Department of Health and Human Services endorsement of such derivative products may not be stated or implied. Evidence Report/Technology Assessment Number 184 Treatment of Common Hip Fractures Prepared for: Agency for Healthcare Research and Quality U.S. Department of Health and Human Services 540 Gaither Road Rockville, MD 20850 www.ahrq.gov Contract No. HHSA 290 2007 10064 1 Prepared by: Minnesota Evidence-based Practice Center, Minneapolis, Minnesota Investigators Mary Butler, Ph.D., M.B.A. Mary Forte, D.C. Robert L. Kane, M.D. Siddharth Joglekar, M.D. Susan J. Duval, Ph.D. Marc Swiontkowski, M.D. -

Pediatric MSK Protocols
UT Southwestern Department of Radiology Ankle and Foot Protocols - Last Update 5-18-2015 Protocol Indications Notes Axial Coronal Sagittal Ankle / Midfoot - Routine Ankle Pain Axial = In Relation to Leg "Footprint" (Long Axis to Foot) T1 FSE PD SPAIR T1 FSE Injury, Internal Derangement Coronal = In Relation to Leg (Short Axis Foot) PD SPAIR STIR Talar OCD, Coalition Protocol Indications Notes Axial Coronal Sagittal Ankle / Midfoot - Arthritis Arthritis Axial = In Relation to Leg "Footprint" (Long Axis to Foot) PD SPAIR PD SPAIR T1 FSE Coronal = In Relation to Leg (Short Axis Foot) STIR T1 SPIR POST T1 SPIR POST Protocol Indications Notes Axial Coronal Sagittal Foot - Routine Pain, AVN Axial = In Relation to Leg "Footprint" (Long Axis to Foot) T1 FSE PD FSE T1 FSE Coronal = In Relation to Leg (Short Axis Foot) PD SPAIR PD SPAIR STIR Protocol Indications Notes Axial Coronal Sagittal Foot - Arthritis Arthritis Axial = In Relation to Leg "Footprint" (Long Axis to Foot) T1 FSE PD SPAIR STIR Coronal = In Relation to Leg (Short Axis Foot) PD SPAIR T1 SPIR POST 3D WATS T1 SPIR POST Protocol Indications Notes Axial Coronal Sagittal Great Toe / MTP Joints Turf Toe Smallest Coil Possible (Microcoil if Available) PD FSE T1 FSE PD FSE Sesamoiditis FoV = Mid Metatarsal Through Distal Phalanges PD SPAIR PD SPAIR PD SPAIR Slice thickness = 2-3 mm, 10% gap Axial = In relation to the great toe (short axis foot) Coronal = In relation to the great toe (long axis foot / footprint) Appropriate Coronal Plane for Both Ankle and Foot Imaging UT Southwestern Department -

Priority Health Spine and Joint Code List
Priority Health Joint Services Code List Category CPT® Code CPT® Code Description Joint Services 23000 Removal of subdeltoid calcareous deposits, open Joint Services 23020 Capsular contracture release (eg, Sever type procedure) Joint Services 23120 Claviculectomy; partial Joint Services 23130 Acromioplasty or acromionectomy, partial, with or without coracoacromial ligament release Joint Services 23410 Repair of ruptured musculotendinous cuff (eg, rotator cuff) open; acute Joint Services 23412 Repair of ruptured musculotendinous cuff (eg, rotator cuff) open;chronic Joint Services 23415 Coracoacromial ligament release, with or without acromioplasty Joint Services 23420 Reconstruction of complete shoulder (rotator) cuff avulsion, chronic (includes acromioplasty) Joint Services 23430 Tenodesis of long tendon of biceps Joint Services 23440 Resection or transplantation of long tendon of biceps Joint Services 23450 Capsulorrhaphy, anterior; Putti-Platt procedure or Magnuson type operation Joint Services 23455 Capsulorrhaphy, anterior;with labral repair (eg, Bankart procedure) Joint Services 23460 Capsulorrhaphy, anterior, any type; with bone block Joint Services 23462 Capsulorrhaphy, anterior, any type;with coracoid process transfer Joint Services 23465 Capsulorrhaphy, glenohumeral joint, posterior, with or without bone block Joint Services 23466 Capsulorrhaphy, glenohumeral joint, any type multi-directional instability Joint Services 23470 ARTHROPLASTY, GLENOHUMERAL JOINT; HEMIARTHROPLASTY ARTHROPLASTY, GLENOHUMERAL JOINT; TOTAL SHOULDER [GLENOID -

Tension Band Wiring Is As Effective As a Compression Screw in a Neglected, Medial Maleolus Non-Union
Case Report Journal of Orthopaedic Case Reports 2017 Jul-Aug: 7(4):Page 72-75 Tension Band Wiring Is As Effective As A Compression Screw In A Neglected, Medial Maleolus Non-Union: A Case-Based Discussion & Literature Review Rakesh John¹, Mandeep Singh Dhillon¹, Ankit Khurana², Sameer Aggarwal¹, Prasoon Kumar¹ Learning Points for this Article: Compression screw fixation has been the workhorse implant for medial malleolar nonunions; however, tension band wiring may be a better technique for such nonunions, as seen in this rare case of isolated, medial malleolus gap nonunion. Abstract Introduction: Isolated, neglected medial malleolus nonunion cases are a rare entity in orthopedic literature. All studies (except one) have described the use of compression screws (with or without plates) for medial malleolar nonunion management. In acute fractures, tension band wiring (TBW) has shown excellent results both in biomechanical and in clinical studies. On the contrary, it has seldom been used in nonunion or in neglected cases. Case Report: We describe a 6-month-old neglected medial malleolus gap nonunion case who presented with progressive pain and limp. TBW with a monoblock, inlay, tricortical, and iliac crest bone graft for the defect was performed. The fracture united within 12 weeks and patient went back to his normal work routine; on the latest follow-up at 3 years, the patient was asymptomatic with no clinicoradiologic signs of secondary osteoarthritis of the ankle joint. Conclusion: TBW may be better than screw fixation in the management of medial malleolus nonunion as it is technically straightforward and cost-effective, can provide equal or more compression than a screw; it does not damage the sandwiched inlay bone graft, and the amount of compression is surgeon-controlled. -

Case Report Arthroscopic Removal of a Wire Fragment from the Posterior Septum of the Knee Following Tension Band Wiring of a Patellar Fracture
Hindawi Publishing Corporation Case Reports in Orthopedics Volume 2015, Article ID 827140, 5 pages http://dx.doi.org/10.1155/2015/827140 Case Report Arthroscopic Removal of a Wire Fragment from the Posterior Septum of the Knee following Tension Band Wiring of a Patellar Fracture Yasuaki Tamaki, Takashi Nakayama, Kenichiro Kita, Katsutosi Miyatake, Yoshiteru Kawasaki, Koji Fujii, and Yoshitsugu Takeda Department of Orthopedic Surgery, Tokushima Red Cross Hospital, 103 Irinokuchi, Komatsushima-cho, Komatsushima, Tokushima 773-8502, Japan Correspondence should be addressed to Yoshitsugu Takeda; [email protected] Received 25 November 2014; Accepted 22 January 2015 Academic Editor: Dimitrios S. Karataglis Copyright © 2015 Yasuaki Tamaki et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Tension band wiring with cerclage wiring is most widely used for treating displaced patellar fractures. Although wire breakage is not uncommon, migration of a fragment of the broken wire is rare, especially migration into the knee joint. We describe here a rare case of migration of a wire fragment into the posterior septum of the knee joint after fixation of a displaced patellar fracture with tension band wiring and cerclage wiring. Although it was difficult to determine whether the wire fragment was located within or outside the knee joint from the preoperative plain radiographs or three-dimensional computed tomography (3D CT), we found it arthroscopically through the posterior transseptal portal with assistance of intraoperative fluoroscopy. Surgeons who treat such cases should bear in mind the possibility that wire could be embedded in the posterior septum of the knee joint. -

Osteoarthritis Epidemiologicosteoarthritis and Genetic Aspects Epidemiologic and Genetic Aspects
From the Department of Orthopedics, Clinical Sciences From the DepartmentLund University, of Orthopedics, Lund, Sweden Clinical Sciences Lund University, Lund, Sweden Osteoarthritis EpidemiologicOsteoarthritis and genetic aspects Epidemiologic and genetic aspects Jonas Franklin Jonas Franklin Thesis 2010 Thesis 2010 Contact address Jonas Franklin Department of Orthopedics Akureyri University Hospital IS-600 Akureyri Iceland E-mail: [email protected] ISSN 1652-8220 ISBN 978-91-86443-87-0 Lund University, Faculty of Medicine Doctoral Dissertation Series 2010:71 Printed in Sweden Mediatryck, Lund 2010 To Hlíf Atli Egill and Jóhann Jonas Franklin 1 Contents List of papers, 2 Radiographic techniques, 17 Radiographic classification, 17 Definitions and abbreviations, 3 Statistical methods, 17 Thesis at a glance, 4 Ethics, 18 Description of contributions, 6 Data encryption and protection of the individual, 18 Introduction, 7 Symptoms and signs of osteoarthritis, 7 Summary of results of papers I-V, 19 Natural history of osteoarthritis, 8 Discussion, 24 Radiographic features of osteoarthritis, 8 Research methodology, 24 Definition of osteoarthritis, 9 Abnormal mechanical loading is a risk factor for Definition of hip fractures, 9 OA, 25 Study methodology, 9 Natural history of OA, 27 Epidemiology of osteoarthritis, 11 OA and hip fracture, 28 Epidemiology of hip fractures, 11 Conclusions, 30 Risk factors for osteoarthritis ,12 Summary, 31 Risk factors for hip fracture, 13 Populärvetenskaplig sammanfattning på Aims, 14 svenska, 33 Patients and methods, 15 Ágrip á íslensku, 35 Overview of patient/subject allocation, 15 Acknowledgements, 37 Patient identification, 15 References, 38 Populations examined, 16 2 Osteoarthritis - Epidemiologic and genetic aspects List of papers This thesis is based on the following papers: I. -

Desarrollo De La Podología En España
Desarrollo de la podología en España Virginia Novel Martí ADVERTIMENT. La consulta d’aquesta tesi queda condicionada a l’acceptació de les següents condicions d'ús: La difusió d’aquesta tesi per mitjà del servei TDX (www.tdx.cat) i a través del Dipòsit Digital de la UB (diposit.ub.edu) ha estat autoritzada pels titulars dels drets de propietat intelꞏlectual únicament per a usos privats emmarcats en activitats d’investigació i docència. No s’autoritza la seva reproducció amb finalitats de lucre ni la seva difusió i posada a disposició des d’un lloc aliè al servei TDX ni al Dipòsit Digital de la UB. No s’autoritza la presentació del seu contingut en una finestra o marc aliè a TDX o al Dipòsit Digital de la UB (framing). Aquesta reserva de drets afecta tant al resum de presentació de la tesi com als seus continguts. En la utilització o cita de parts de la tesi és obligat indicar el nom de la persona autora. ADVERTENCIA. La consulta de esta tesis queda condicionada a la aceptación de las siguientes condiciones de uso: La difusión de esta tesis por medio del servicio TDR (www.tdx.cat) y a través del Repositorio Digital de la UB (diposit.ub.edu) ha sido autorizada por los titulares de los derechos de propiedad intelectual únicamente para usos privados enmarcados en actividades de investigación y docencia. No se autoriza su reproducción con finalidades de lucro ni su difusión y puesta a disposición desde un sitio ajeno al servicio TDR o al Repositorio Digital de la UB. -

Overuse Injuries in Elite Athletes
ATHLETES: OVERUSE MRI of Overuse INJURIES VERY COMMON Injury in the Elite Athlete • Muscle William B. Morrison, M.D. Associate Professor of • Joints Radiology Thomas Jefferson • Tendon University Hospital Philadelphia, PA USA • Ligament • Bone [email protected] How is this Relevant to My Practice? • High performance athletes get similar injuries as ‘regular’ people… but: – More of them, & at a younger age MUSCLE / SOFT TISSUE – More commonly imaged – Secondary gain involved INJURY • All the cases here are high performance athletes – but most injuries are conventional • Exceptions – Some weird sport-specific patterns of stress and other injuries – Acute injuries as opposed to overuse •Overuse –DOMS Muscle Injury Delayed Onset Muscle • Acute injury Soreness (DOMS) – Tendon – Myotendinous junction • All athletes are susceptible if they – Muscle belly change training regimen • Athletes tend to work out intensely – leads to muscle injury • Rarely imaged (“no pain no gain”) – Weightlifting / aerobic exercise – 24hr later – soreness – Can be severe, even look like a tear 1 Acute Muscle Injury • Direct → muscle belly, esp quadriceps T2 – esp rugby, football • Indirect → myotendinous junction – eccentric contraction – sudden acceleration / DOMS: Lateral gastrocnemius deceleration Finding can be subtle, even with T2 and fat sat Professional football player Acute Injury – T1 Quadriceps hematoma Muscle belly injury Myotendinous Unit Hit with helmet • Myotendinous Junction – ‘weak link’ of normal myotendinous complex T2 – Common place for injury – Most common mechanism: eccentric contraction (muscle lengthens and contracts at the same High time) T1 = blood Gastrocnemius tear: Grade 1 Muscle Strain “V” sign • Ill-defined edema T2 T1 STIR Weishaupt D, JCAT 2001; 25:677 2 Gd Gd can help identify Grade 2 Muscle Strain subtle muscle injury AKA “Partial Tear” STIR Professional baseball player Grade 1 strain Sartorius m.