Gases & Applications
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Nitrogen Trifluoride Hazard Summary Identification
Common Name: NITROGEN TRIFLUORIDE CAS Number: 7783-54-2 RTK Substance number: 1380 DOT Number: UN 2451 Date: March 2001 ------------------------------------------------------------------------- ------------------------------------------------------------------------- HAZARD SUMMARY * Nitrogen Trifluoride can affect you when breathed in. * Exposure to hazardous substances should be routinely * Contact may irritate the skin and eyes. evaluated. This may include collecting personal and area * High levels can interfere with the ability of the blood to air samples. You can obtain copies of sampling results carry Oxygen causing headache, fatigue, dizziness, and a from your employer. You have a legal right to this blue color to the skin and lips (methemoglobinemia). information under OSHA 1910.1020. Higher levels can cause trouble breathing, collapse and * If you think you are experiencing any work-related health even death. problems, see a doctor trained to recognize occupational * Repeated high exposure can cause weakness, muscle diseases. Take this Fact Sheet with you. twitching, seizures and convulsions. * Nitrogen Trifluoride may damage the liver and kidneys. WORKPLACE EXPOSURE LIMITS * Repeated high exposure can cause deposits of Fluorides in OSHA: The legal airborne permissible exposure limit the bones and teeth, a condition called "Fluorosis." This (PEL) is 10 ppm averaged over an 8-hour can cause pain, disability and mottling of the teeth. workshift. * The above health effects do NOT occur at the level of Fluoride used in water for preventing cavities in teeth. NIOSH: The recommended airborne exposure limit is 10 ppm averaged over a 10-hour workshift. IDENTIFICATION Nitrogen Trifluoride is a colorless gas with a moldy odor. It ACGIH: The recommended airborne exposure limit is is used as a Fluorine source in the electronics industry and in 10 ppm averaged over an 8-hour workshift. -

NF3, the Greenhouse Gas Missing from Kyoto Michael J
GEOPHYSICAL RESEARCH LETTERS, VOL. 35, L12810, doi:10.1029/2008GL034542, 2008 NF3, the greenhouse gas missing from Kyoto Michael J. Prather1 and Juno Hsu1 Received 5 May 2008; accepted 27 May 2008; published 26 June 2008. [1] Nitrogen trifluoride (NF3) can be called the missing chemical industry are following: Kanto Denka produces greenhouse gas: It is a synthetic chemical produced in 1,000 tons per yr; DuPont is setting up a manufacturing industrial quantities; it is not included in the Kyoto basket of facility in China; Formosa Plastics and Mitsui Chemicals greenhouse gases or in national reporting under the United are expanding production. Data on total production is not Nations Framework Convention on Climate Change freely available, but based on press releases and industry (UNFCCC); and there are no observations documenting its data, we estimate 2008 production to be about 4,000 tons, atmospheric abundance. Current publications report a long with a likely range of ±25%. Planned expansion could lifetime of 740 yr and a global warming potential (GWP), double this by 2010. which in the Kyoto basket is second only to SF6.Were- [3] The published global warming potentials (GWP) of examine the atmospheric chemistry of NF3 and calculate a NF3 are 12,300, 17,200 and 20,700 for time horizons of shorter lifetime of 550 yr, but still far beyond any societal 20, 100, and 500 years, respectively [Forster et al., 2007]. time frames. With 2008 production equivalent to 67 million The increasing GWP with time horizon reflects the longer metric tons of CO2,NF3 has a potential greenhouse impact atmospheric residence time of NF3 compared with that of larger than that of the industrialized nations’ emissions of CO2. -

Chm 303 Course Guide
COURSE GUIDE CHM 303 INORGANIC CHEMISTRY III Course Team Professor Phd Aliyu (Course Writer) - Department of Chemistry University of Abuja Prof. Sulaiman o. Idris (Course Reviewer) - Department of Chemistry Ahmadu Bello University Zaria Dr Emeka C. Ogoko - (Head of Department) - NOUN NATIONAL OPEN UNIVERSITY OF NIGERIA CHM 303 COURSE GUIDE © 2021 by NOUN Press National Open University of Nigeria Headquarters University Village Plot 91, Cadastral Zone NnamdiAzikiwe Expressway Jabi, Abuja Lagos Office 14/16 Ahmadu Bello Way Victoria Island, Lagos e-mail: [email protected] URL: www.nou.edu.ng All rights reserved. No part of this book may be reproduced, in any form or by any means, without permission in writing from the publisher. Printed 2021 ISBN: 978-978-058-092-6 ii CHM 303 COURSE GUIDE CONTENTS PAGE Introduction………………………………………………. iv What You Will Learn in This Course…………………….. iv Course Aim………………………………………………… iv Course Objectives…………………………………………. v Working through this Course……………………………… v The Course Materials……………………………………… v Study Sessions……………………………………………… vi Presentation Schedule……………………………………… vii Assessment………………………………………………… vii Tutor Marked Assignments ………………………………… vii Final Examination and Grading……………………………. vii Course Marking Scheme…………………………………… viii Facilitators/Tutors and Tutorials…………………………… viii iii CHM 303 COURSE GUIDE INTRODUCTION Inorganic Chemistry III course (CHM 303) is one of the core courses for the Bachelor of Science degree programme in Chemistry. It is a three- credit unit course at 300 level of the National Open University of Nigeria, designed for students with a fair background knowledge in inorganic Chemistry II course. This course gives an over view of the physical and chemical properties of the elements of the periodic table in addition to the extraction and purification of metals. -

Group 18 - the Elements
Group 18 - The Elements ! All found in minute quantities in air. • Argon is most abundant (and cheapest), comprising 0.934% of air by volume. ! Although rare on earth, helium is the second most abundant element in the universe (H, 76%; He, 23%), being a major component of stars. ! All radon isotopes are short-lived and are continuously being produced by natural decay processes. 222 • Longest lived isotope is Rn (á, t½ = 3.825 days). ! Condensed phases are held together by van der Waals forces, which increase smoothly down the group. Element b.p. (K) I (kJ/mol) He 4.18 2372 Ne 27.13 2080 Ar 87.29 1520 Kr 120.26 1351 Xe 166.06 1169 Rn 208.16 1037 Helium ! Helium is found in certain natural gas deposits (e.g., in Kansas), where it probably forms as a result of radioactive decay in the rocks. ! Because of its low boiling point, it is used widely in cryogenic applications. • Also used in airships (e.g., Goodyear blimp) and as a balloon filler. • Used as a fill gas for deep diving, because it is less soluble in blood than nitrogen, thus avoiding the "bends." ! The 4He isotope has the lowest know boiling point, 4.12 K, at which point it is called He-I. • On cooling to 2.178 K a phase transition to He-II occurs. • He-II has an expanded volume, almost no viscosity, and is superconducting. • He-II can readily flow uphill to equalize volumes in adjacent vessels. • It cannot be stored in glass Dewars, because it leaks through glass into the evacuated space, posing an explosion risk. -

Nitrogen Trifluoride
Nitrogen trifluoride (CAS No: 7783-54-2) Health-based Reassessment of Administrative Occupational Exposure Limits Committee on Updating of Occupational Exposure Limits, a committee of the Health Council of the Netherlands No. 2000/15OSH/125, The Hague, June 8, 2004 Preferred citation: Health Council of the Netherlands: Committee on Updating of Occupational Exposure Limits. Nitrogen trifluoride; Health-based Reassessment of Administrative Occupational Exposure Limits. The Hague: Health Council of the Netherlands, 2004; 2000/15OSH/125. all rights reserved 1 Introduction The present document contains the assessment of the health hazard of nitrogen trifluoride by the Committee on Updating of Occupational Exposure Limits, a committee of the Health Council of the Netherlands. The first draft of this document was prepared by MA Maclaine Pont, M.Sc. (Wageningen University and Research Centre, Wageningen, the Netherlands). In November 1999, literature was searched in the databases Toxline, Medline, and Chemical Abstracts, starting from 1981, 1966, and 1937, respectively, and using the following key words: nitrogen trifluoride, nitrogen fluoride (NF3), and 7783-54-2. In February 2001, the President of the Health Council released a draft of the document for public review. No comments were received. An additional search in Toxline and Medline in January 2004 did not result in information changing the committee’s conclusions. 2Identity name : nitrogen trifluoride synonyms : nitrogen fluoride; trifluoroamine; trifluoroammonia; perfluoroammonia molecular formula : NF3 CAS number : 7783-54-2 3 Physical and chemical properties molecular weight : 71.0 boiling point : -129oC melting point : -208.5oC flash point : - vapour pressure : at 20°C: >100 kPa solubility in water : very slightly soluble log Poctanol/water : -1.60 conversion factors : at 20°C, 101.3 kPa: 1 mg/m3 = 0.34 ppm 1 ppm = 2.96 mg/m3 Data from ACG91, NLM04, http://esc.syrres.com. -

Cleaning Gas Mixture for an Apparatus and Cleaning Method
Europäisches Patentamt *EP001529854A1* (19) European Patent Office Office européen des brevets (11) EP 1 529 854 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: (51) Int Cl.7: C23C 16/44, H01L 21/00 11.05.2005 Bulletin 2005/19 (21) Application number: 04256591.1 (22) Date of filing: 26.10.2004 (84) Designated Contracting States: (72) Inventors: AT BE BG CH CY CZ DE DK EE ES FI FR GB GR • Isaki, Ryuichiro, HU IE IT LI LU MC NL PL PT RO SE SI SK TR Taiyo Nippon Sanso Corporation Designated Extension States: Shinagawa-ku, Tokyo (JP) AL HR LT LV MK • Shinriki, Manabu, Taiyo Nippon Sanso Corporation (30) Priority: 04.11.2003 JP 2003374160 Shinagawa-ku, Tokyo (JP) (71) Applicant: Taiyo Nippon Sanso Corporation (74) Representative: Smyth, Gyles Darren Shinagawa-ku, Tokyo (JP) Marks & Clerk 90 Long Acre London WC2E 9RA (GB) (54) Cleaning gas mixture for an apparatus and cleaning method (57) A cleaning gas improves the etching reaction and an integer from 1 to 3, y is an integer from 1 to 12, rate of cleaning gas including a fluorocarbon gas, and and z is selected from 0 and 1 and oxygen gas, to which increases the cleaning effect. And the cleaning method is added at least one selected from the group of nitrogen uses the cleaning gas. A mixed gas of a fluorocarbon trifluoride, fluorine, nitrous oxide, nitrogen, and rare gas- gas represented by the general formula of CvHxFyOz, es up to 10% by volume based on the total gas volume. wherein v is an integer from 1 to 5, x is selected from 0 EP 1 529 854 A1 Printed by Jouve, 75001 PARIS (FR) 1 EP 1 529 854 A1 2 Description ciency since these are raw materials for producing fluor- ocarbon type resins such as TEFLON (trademark), and BACKGROUND OF THE INVENTION are used in significantly large amounts in the chemical industry. -

1. Hydrogen Forms Compounds with Most Non-Metallic Elements and with Some Metals
PMT 1. Hydrogen forms compounds with most non-metallic elements and with some metals. (a) Calculate the empirical formula of the compound used in the manufacture of artificial rubber which has the following composition by mass. Hydrogen 11.1% Carbon 88.9% (3) (b) The boiling temperatures of hydrogen chloride and hydrogen iodide are: Hydrogen chloride ±85ºC Hydrogen iodide ±35ºC Explain why hydrogen iodide has a higher boiling temperature than hydrogen chloride. ............................................................................................................................. ... ............................................................................................................................. ... ............................................................................................................................. ... (2) (c) Draw and explain the shapes of: (i) the PH3 molecule; .......................................................... ............................................................ ...................................................................................................................... (2) 1 PMT ± (ii) the AlH4 ion. ...................................................................................................................... ...................................................................................................................... (2) 3 (d) Calculate the number of molecules in 8.0 cm of gaseous phosphine, PH3, at room temperature and pressure. (The molar volume of -

X-Panding Halogen Bonding Interactions: Hybrid Cocrystals Composed of Ionic Halides, Iodine and Organoiodines
Clemson University TigerPrints All Dissertations Dissertations 5-2020 X-Panding Halogen Bonding Interactions: Hybrid Cocrystals Composed of Ionic Halides, Iodine and Organoiodines Khadijatul Kobra Clemson University Follow this and additional works at: https://tigerprints.clemson.edu/all_dissertations Part of the Chemistry Commons Recommended Citation Kobra, Khadijatul, "X-Panding Halogen Bonding Interactions: Hybrid Cocrystals Composed of Ionic Halides, Iodine and Organoiodines" (2020). All Dissertations. 2572. https://tigerprints.clemson.edu/all_dissertations/2572 This Dissertation is brought to you for free and open access by the Dissertations at TigerPrints. It has been accepted for inclusion in All Dissertations by an authorized administrator of TigerPrints. For more information, please contact [email protected]. X-PANDING HALOGEN BONDING INTERACTIONS: HYBRID COCRYSTALS COMPOSED OF IONIC HALIDES, IODINE AND ORGANOIODINES A Dissertation Presented to the Graduate School of Clemson University In Partial Fulfillment of the Requirements for the Degree Doctor of Philosophy Chemistry by Khadijatul Kobra May 2020 Accepted by: William T. Pennington, Committee Chair Colin D. McMillen, Committee Co-chair Joseph S. Thrasher Stephen E. Creager Rakesh Sachdeva i ABSTRACT Halogen bonding referred to as an attractive, noncovalent interaction between an electrophilic region of a halogen atom X (acts as Lewis acid) and a nucleophilic region of a molecule Y (acts as Lewis base). Such interactions and the resulting polymeric networks play an important role in many fields related to crystal engineering, including for example, the fabrication of liquid crystals and novel drug design. The application of halogen bonding has particular promise in biological systems by increasing the lipophilicity of drugs to improve penetration through lipid membranes and tissues, enabling better intracellular delivery. -
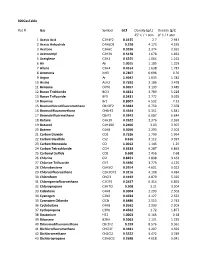
Gas Conversion Factor for 300 Series
300GasTable Rec # Gas Symbol GCF Density (g/L) Density (g/L) 25° C / 1 atm 0° C / 1 atm 1 Acetic Acid C2H4F2 0.4155 2.7 2.947 2 Acetic Anhydride C4H6O3 0.258 4.173 4.555 3 Acetone C3H6O 0.3556 2.374 2.591 4 Acetonitryl C2H3N 0.5178 1.678 1.832 5 Acetylene C2H2 0.6255 1.064 1.162 6 Air Air 1.0015 1.185 1.293 7 Allene C3H4 0.4514 1.638 1.787 8 Ammonia NH3 0.7807 0.696 0.76 9 Argon Ar 1.4047 1.633 1.782 10 Arsine AsH3 0.7592 3.186 3.478 11 Benzene C6H6 0.3057 3.193 3.485 12 Boron Trichloride BCl3 0.4421 4.789 5.228 13 Boron Triflouride BF3 0.5431 2.772 3.025 14 Bromine Br2 0.8007 6.532 7.13 15 Bromochlorodifluoromethane CBrClF2 0.3684 6.759 7.378 16 Bromodifluoromethane CHBrF2 0.4644 5.351 5.841 17 Bromotrifluormethane CBrF3 0.3943 6.087 6.644 18 Butane C4H10 0.2622 2.376 2.593 19 Butanol C4H10O 0.2406 3.03 3.307 20 Butene C4H8 0.3056 2.293 2.503 21 Carbon Dioxide CO2 0.7526 1.799 1.964 22 Carbon Disulfide CS2 0.616 3.112 3.397 23 Carbon Monoxide CO 1.0012 1.145 1.25 24 Carbon Tetrachloride CCl4 0.3333 6.287 6.863 25 Carbonyl Sulfide COS 0.668 2.456 2.68 26 Chlorine Cl2 0.8451 2.898 3.163 27 Chlorine Trifluoride ClF3 0.4496 3.779 4.125 28 Chlorobenzene C6H5Cl 0.2614 4.601 5.022 29 Chlorodifluoroethane C2H3ClF2 0.3216 4.108 4.484 30 Chloroform CHCl3 0.4192 4.879 5.326 31 Chloropentafluoroethane C2ClF5 0.2437 6.314 6.892 32 Chloropropane C3H7Cl 0.308 3.21 3.504 33 Cisbutene C4H8 0.3004 2.293 2.503 34 Cyanogen C2N2 0.4924 2.127 2.322 35 Cyanogen Chloride ClCN 0.6486 2.513 2.743 36 Cyclobutane C4H8 0.3562 2.293 2.503 37 Cyclopropane C3H6 0.4562 -

Chemistry of the Noble Gases*
CHEMISTRY OF THE NOBLE GASES* By Professor K. K. GREE~woon , :.\I.Sc., sc.D .. r".lU.C. University of N ewca.stle 1tpon Tyne The inert gases, or noble gases as they are elements were unsuccessful, and for over now more appropriately called, are a remark 60 years they epitomized chemical inertness. able group of elements. The lightest, helium, Indeed, their electron configuration, s2p6, was recognized in the gases of the sun before became known as 'the stable octet,' and this it was isolated on ea.rth as its name (i]A.tos) fotmed the basis of the fit·st electronic theory implies. The first inert gas was isolated in of valency in 1916. Despite this, many 1895 by Ramsay and Rayleigh; it was named people felt that it should be possible to induce argon (apy6s, inert) and occurs to the extent the inert gases to form compounds, and many of 0·93% in the earth's atmosphere. The of the early experiments directed to this end other gases were all isolated before the turn have recently been reviewed.l of the century and were named neon (v€ov, There were several reasons why chemists new), krypton (KpVn'TOV, hidden), xenon believed that the inert gases might form ~€vov, stmnger) and radon (radioactive chemical compounds under the correct con emanation). Though they occur much less ditions. For example, the ionization poten abundantly than argon they cannot strictly tial of xenon is actually lower than those of be called rare gases; this can be illustrated hydrogen, nitrogen, oxygen, fl uorine and by calculating the volumes occupied a.t s.t.p. -

And Perfluoroalkyl Substances (PFAS): Sources, Pathways and Environmental Data
Poly- and perfluoroalkyl substances (PFAS): sources, pathways and environmental data Chief Scientist’s Group report August 2021 We are the Environment Agency. We protect and improve the environment. We help people and wildlife adapt to climate change and reduce its impacts, including flooding, drought, sea level rise and coastal erosion. We improve the quality of our water, land and air by tackling pollution. We work with businesses to help them comply with environmental regulations. A healthy and diverse environment enhances people's lives and contributes to economic growth. We can’t do this alone. We work as part of the Defra group (Department for Environment, Food & Rural Affairs), with the rest of government, local councils, businesses, civil society groups and local communities to create a better place for people and wildlife. Published by: Author: Emma Pemberton Environment Agency Horizon House, Deanery Road, Environment Agency’s Project Manager: Bristol BS1 5AH Mark Sinton www.gov.uk/environment-agency Citation: Environment Agency (2021) Poly- and © Environment Agency 2021 perfluoroalkyl substances (PFAS): sources, pathways and environmental All rights reserved. This document may data. Environment Agency, Bri be reproduced with prior permission of the Environment Agency. Further copies of this report are available from our publications catalogue: www.gov.uk/government/publications or our National Customer Contact Centre: 03708 506 506 Email: research@environment- agency.gov.uk 2 of 110 Research at the Environment Agency Scientific research and analysis underpins everything the Environment Agency does. It helps us to understand and manage the environment effectively. Our own experts work with leading scientific organisations, universities and other parts of the Defra group to bring the best knowledge to bear on the environmental problems that we face now and in the future. -

Octafluoropropane
Revised edition no : 4 SAFETY DATA SHEET ACCORDING TO REGULATION 1907/2006 Date : 08.10.2019 OCTAFLUOROPROPANE 1. IDENTIFICATION OF THE SUBSTANCE/COMPOUND AND COMPANY 1.1. Identification of the product Name: Octafluoropropane Chemical nomenclature: IUPAC name: Octafluoropropane Synonyms: Perfluoropropane, Khladon 218, R218 Molecular formula: С3F8 Structural formula Molar mass: 188,02 EC number: 200-941-9 (EINECS) REACH Pre-Registration Reference number 05-2114096818-30-0000 30/06/2008 C&L bulk notification Reference number 02-2119708805-36-0000 CAS number: 76-19-7 1.2. Use of substance/ Is used as reaction mass for plasmochemical etching of semiconducting compound materials, and as a refrigerant. Recommended use For industrial or professional use only, not for use in everyday life. 1.3. Company identification Manufacturer Joint Stock Company «HaloPolymer Perm» 614042, Russia, Perm, ul. Lasvinskaya 98 Phone № +7(342) 250-61-50 Web site: www.halopolymer.ru Only REACH representative in JSC «HaloPolymer Perm» (Submitting legal entity URALCHEM Assist EU: GmbH) Johannssenstrasse 10 30159, Hannover, Germany Tel: +49 511 45 99 444 1.4 Emergency telephone number: +7-342-282-85-45 (24 hours) Great Britain +44 (0) 203 394 9870 (24/7) USA 1-877 271 7077 2. HAZARDS IDENTIFICATION 2.1. Substance classification 2.1.1. Classification in accordance with the Regulation (EC) No 1272/2008 [CLP/GHS] Liquefied gas, H280 2.1.2. Classification in Not classified as a dangerous product. accordance with the Directive 67/548/EEC 2.2 Label elements Hazard pictograms 2.2.1 Labeling according to the Regulation (EC) No 1272/2008 [CLP/GHS] GHS04 Signal word: Warning Hazard statements: H280 (Contains gas under pressure; may explode if heated).