Occurrence and Function of the Na+-Translocating NADH:Quinone Oxidoreductase in Prevotella Spp
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

An Integrative Bayesian Dirichlet-Multinomial Regression Model for the Analysis of Taxonomic Abundances in Microbiome Data
Wadsworth et al. BMC Bioinformatics (2017) 18:94 DOI 10.1186/s12859-017-1516-0 METHODOLOGY ARTICLE Open Access An integrative Bayesian Dirichlet- multinomial regression model for the analysis of taxonomic abundances in microbiome data W. Duncan Wadsworth1, Raffaele Argiento2, Michele Guindani3, Jessica Galloway-Pena4, Samuel A. Shelburne5 and Marina Vannucci1* Abstract Background: The Human Microbiome has been variously associated with the immune-regulatory mechanisms involved in the prevention or development of many non-infectious human diseases such as autoimmunity, allergy and cancer. Integrative approaches which aim at associating the composition of the human microbiome with other available information, such as clinical covariates and environmental predictors, are paramount to develop a more complete understanding of the role of microbiome in disease development. Results: In this manuscript, we propose a Bayesian Dirichlet-Multinomial regression model which uses spike-and-slab priors for the selection of significant associations between a set of available covariates and taxa from a microbiome abundance table. The approach allows straightforward incorporation of the covariates through a log-linear regression parametrization of the parameters of the Dirichlet-Multinomial likelihood. Inference is conducted through a Markov Chain Monte Carlo algorithm, and selection of the significant covariates is based upon the assessment of posterior probabilities of inclusions and the thresholding of the Bayesian false discovery rate. We design a simulation study to evaluate the performance of the proposed method, and then apply our model on a publicly available dataset obtained from the Human Microbiome Project which associates taxa abundances with KEGG orthology pathways. The method is implemented in specifically developed R code, which has been made publicly available. -

Ninety-Nine De Novo Assembled Genomes from the Moose (Alces Alces) Rumen Microbiome Provide New Insights Into Microbial Plant Biomass Degradation
The ISME Journal (2017) 11, 2538–2551 © 2017 International Society for Microbial Ecology All rights reserved 1751-7362/17 www.nature.com/ismej ORIGINAL ARTICLE Ninety-nine de novo assembled genomes from the moose (Alces alces) rumen microbiome provide new insights into microbial plant biomass degradation Olov Svartström1, Johannes Alneberg2, Nicolas Terrapon3,4, Vincent Lombard3,4, Ino de Bruijn2, Jonas Malmsten5,6, Ann-Marie Dalin6, Emilie EL Muller7, Pranjul Shah7, Paul Wilmes7, Bernard Henrissat3,4,8, Henrik Aspeborg1 and Anders F Andersson2 1School of Biotechnology, Division of Industrial Biotechnology, KTH Royal Institute of Technology, Stockholm, Sweden; 2School of Biotechnology, Division of Gene Technology, KTH Royal Institute of Technology, Science for Life Laboratory, Stockholm, Sweden; 3CNRS UMR 7257, Aix-Marseille University, 13288 Marseille, France; 4INRA, USC 1408 AFMB, 13288 Marseille, France; 5Department of Pathology and Wildlife Diseases, National Veterinary Institute, Uppsala, Sweden; 6Division of Reproduction, Department of Clinical Sciences, Swedish University of Agricultural Sciences, Uppsala, Sweden; 7Luxembourg Centre for Systems Biomedicine, University of Luxembourg, Esch-sur-Alzette, Luxembourg and 8Department of Biological Sciences, King Abdulaziz University, Jeddah, Saudi Arabia The moose (Alces alces) is a ruminant that harvests energy from fiber-rich lignocellulose material through carbohydrate-active enzymes (CAZymes) produced by its rumen microbes. We applied shotgun metagenomics to rumen contents from six moose to obtain insights into this microbiome. Following binning, 99 metagenome-assembled genomes (MAGs) belonging to 11 prokaryotic phyla were reconstructed and characterized based on phylogeny and CAZyme profile. The taxonomy of these MAGs reflected the overall composition of the metagenome, with dominance of the phyla Bacteroidetes and Firmicutes. -

Xiexin Tang Improves the Symptom of Type 2 Diabetic Rats by Modulation of the Gut Microbiota
www.nature.com/scientificreports OPEN Xiexin Tang improves the symptom of type 2 diabetic rats by modulation of the gut microbiota Received: 30 August 2017 Xiaoyan Wei, Jinhua Tao , Suwei Xiao, Shu Jiang, Erxin Shang, Zhenhua Zhu, Dawei Qian Accepted: 13 February 2018 & Jinao Duan Published: xx xx xxxx Type 2 diabetes mellitus (T2DM), a chronic metabolic disease which severely impairs peoples’ quality of life, currently attracted worldwide concerns. There are growing evidences that gut microbiota can exert a great impact on the development of T2DM. Xiexin Tang (XXT), a traditional Chinese medicine prescription, has been clinically used to treat diabetes for thousands of years. However, few researches are investigated on the modulation of gut microbiota community by XXT which will be very helpful to unravel how it works. In this study, bacterial communities were analyzed based on high-throughput 16S rRNA gene sequencing. Results indicated that XXT could notably shape the gut microbiota. T2DM rats treated with XXT exhibited obvious changes in the composition of the gut microbiota, especially for some short chain fatty acids producing and anti-infammatory bacteria such as Adlercreutzia, Alloprevotella, Barnesiella, [Eubacterium] Ventriosum group, Blautia, Lachnospiraceae UCG-001, Papillibacter and Prevotellaceae NK3B31 group. Additionally, XXT could also signifcantly ameliorate hyperglycemia, lipid metabolism dysfunction and infammation in T2DM rats. Moreover, the correlation analysis illustrated that the key microbiota had a close relationship with the T2DM related indexes. The results probably provided useful information for further investigation on its active mechanism and clinical application. T2DM, a chronic metabolic disease characterized by hyperglycemia as a result of insufcient insulin secretion, insulin action or both1, is estimated that its numbers in the adults will increase by 55% by 20352. -

1587714682 277 2.Pdf
Systematic and Applied Microbiology 42 (2019) 107–116 Contents lists available at ScienceDirect Systematic and Applied Microbiology jou rnal homepage: http://www.elsevier.com/locate/syapm The diverse and extensive plant polysaccharide degradative apparatuses of the rumen and hindgut Prevotella species: A factor in their ubiquity? ∗ Tomazˇ Accetto , Gorazd Avgustinˇ University of Ljubljana, Biotechnical faculty, Animal Science Department, Groblje 3, 1230 Domzale,ˇ Slovenia a r t i c l e i n f o a b s t r a c t Article history: Although the Prevotella are commonly observed in high shares in the mammalian hindgut and rumen Received 2 August 2018 studies using NGS approach, the knowledge on their actual role, though postulated to lie in soluble fibre Received in revised form 2 October 2018 degradation, is scarce. Here we analyse in total 23, more than threefold of hitherto known rumen and Accepted 3 October 2018 hindgut Prevotella species and show that rumen/hindgut Prevotella generally possess extensive reper- toires of polysaccharide utilization loci (PULs) and carbohydrate active enzymes targeting various plant Keywords: polysaccharides. These PUL repertoires separate analysed Prevotella into generalists and specialists yet a Prevotella finer diversity among generalists is evident too, both in range of substrates targeted and in PUL combi- Rumen Hindgut nations targeting the same broad substrate classes. Upon evaluation of the shares of species analysed in this study in rumen metagenomes we found firstly, that they contributed significantly to total Prevotella Polysaccharide utilization locus CAZYme abundance though much of rumen Prevotella diversity may still be unknown. Secondly, the hindgut Pre- Metagenome votella species originally isolated in pigs and humans occasionally dominated among the Prevotella with surprisingly high metagenome read shares and were consistently found in rumen metagenome samples from sites as apart as New Zealand and Scotland. -

Gut Microbiome Composition Remains Stable in Individuals with Diabetes-Related Early to Late Stage Chronic Kidney Disease
biomedicines Article Gut Microbiome Composition Remains Stable in Individuals with Diabetes-Related Early to Late Stage Chronic Kidney Disease Ashani Lecamwasam 1,2,3,*, Tiffanie M. Nelson 4, Leni Rivera 3, Elif I. Ekinci 2,5, Richard Saffery 1,6 and Karen M. Dwyer 3 1 Epigenetics Research, Murdoch Children’s Research Institute, VIC 3052, Australia; [email protected] 2 Department of Endocrinology, Austin Health, VIC 3079, Australia; [email protected] 3 School of Medicine, Faculty of Health, Deakin University, VIC 3220, Australia; [email protected] (L.R.); [email protected] (K.M.D.) 4 Menzies Health Institute Queensland, Griffith University, QLD 4222, Australia; [email protected] 5 Department of Medicine, University of Melbourne, VIC 3010, Australia 6 Department of Paediatrics, University of Melbourne, VIC 3010, Australia * Correspondence: [email protected]; Tel.: +613-8341-6200; Fax: +613-9348-1391 Abstract: (1) Background: Individuals with diabetes and chronic kidney disease display gut dysbiosis when compared to healthy controls. However, it is unknown whether there is a change in dysbiosis across the stages of diabetic chronic kidney disease. We investigated a cross-sectional study of patients with early and late diabetes associated chronic kidney disease to identify possible microbial differences between these two groups and across each of the stages of diabetic chronic kidney disease. (2) Methods: This cross-sectional study recruited 95 adults. DNA extracted from collected stool samples were used for 16S rRNA sequencing to identify the bacterial community in the Citation: Lecamwasam, A.; Nelson, gut. (3) Results: The phylum Firmicutes was the most abundant and its mean relative abundance T.M.; Rivera, L.; Ekinci, E.I.; Saffery, was similar in the early and late chronic kidney disease group, 45.99 ± 0.58% and 49.39 ± 0.55%, R.; Dwyer, K.M. -
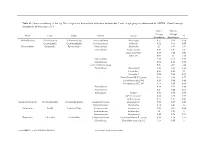
Genus Contributing to the Top 70% of Significant Dissimilarity of Bacteria Between Day 7 and 18 Age Groups As Determined by SIMPER
Table S1: Genus contributing to the top 70% of significant dissimilarity of bacteria between day 7 and 18 age groups as determined by SIMPER. Overall average dissimilarity between ages is 51%. Day 7 Day 18 Average Average Phyla Class Order Family Genus % Abundance Abundance Actinobacteria Actinobacteria Actinomycetales Actinomycetaceae Actinomyces 0.57 0.24 0.74 Coriobacteriia Coriobacteriales Coriobacteriaceae Collinsella 0.51 0.61 0.57 Bacteroidetes Bacteroidia Bacteroidales Bacteroidaceae Bacteroides 2.1 1.63 1.03 Marinifilaceae Butyricimonas 0.82 0.81 0.9 Sanguibacteroides 0.08 0.42 0.59 CAG-873 0.01 1.1 1.68 Marinifilaceae 0.38 0.78 0.91 Marinifilaceae 0.78 1.04 0.97 p-2534-18B5 gut group 0.06 0.7 1.04 Prevotellaceae Alloprevotella 0.46 0.83 0.89 Prevotella 2 0.95 1.11 1.3 Prevotella 7 0.22 0.42 0.56 Prevotellaceae NK3B31 group 0.62 0.68 0.77 Prevotellaceae UCG-003 0.26 0.64 0.89 Prevotellaceae UCG-004 0.11 0.48 0.68 Prevotellaceae 0.64 0.52 0.89 Prevotellaceae 0.27 0.42 0.55 Rikenellaceae Alistipes 0.39 0.62 0.77 dgA-11 gut group 0.04 0.38 0.56 RC9 gut group 0.79 1.17 0.95 Epsilonbacteraeota Campylobacteria Campylobacterales Campylobacteraceae Campylobacter 0.58 0.83 0.97 Helicobacteraceae Helicobacter 0.12 0.41 0.6 Firmicutes Bacilli Lactobacillales Enterococcaceae Enterococcus 0.36 0.31 0.64 Lactobacillaceae Lactobacillus 1.4 1.24 1 Streptococcaceae Streptococcus 0.82 0.58 0.53 Firmicutes Clostridia Clostridiales Christensenellaceae Christensenellaceae R-7 group 0.38 1.36 1.56 Clostridiaceae Clostridium sensu stricto 1 1.16 0.58 -

Role of Actinobacteria and Coriobacteriia in the Antidepressant Effects of Ketamine in an Inflammation Model of Depression
Pharmacology, Biochemistry and Behavior 176 (2019) 93–100 Contents lists available at ScienceDirect Pharmacology, Biochemistry and Behavior journal homepage: www.elsevier.com/locate/pharmbiochembeh Role of Actinobacteria and Coriobacteriia in the antidepressant effects of ketamine in an inflammation model of depression T Niannian Huanga,1, Dongyu Huaa,1, Gaofeng Zhana, Shan Lia, Bin Zhub, Riyue Jiangb, Ling Yangb, ⁎ ⁎ Jiangjiang Bia, Hui Xua, Kenji Hashimotoc, Ailin Luoa, , Chun Yanga, a Department of Anesthesiology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, China b Department of Internal Medicine, The Third Affiliated Hospital of Soochow University, Changzhou 213003, China c Division of Clinical Neuroscience, Chiba University Center for Forensic Mental Health, Chiba 260-8670, Japan ARTICLE INFO ABSTRACT Keywords: Ketamine, an N-methyl-D-aspartic acid receptor (NMDAR) antagonist, elicits rapid-acting and sustained anti- Ketamine depressant effects in treatment-resistant depressed patients. Accumulating evidence suggests that gut microbiota Depression via the gut-brain axis play a role in the pathogenesis of depression, thereby contributing to the antidepressant Lipopolysaccharide actions of certain compounds. Here we investigated the role of gut microbiota in the antidepressant effects of Gut microbiota ketamine in lipopolysaccharide (LPS)-induced inflammation model of depression. Ketamine (10 mg/kg) sig- nificantly attenuated the increased immobility time in forced swimming test (FST), which was associated with the improvements in α-diversity, consisting of Shannon, Simpson and Chao 1 indices. In addition to α-diversity, β-diversity, such as principal coordinates analysis (PCoA), and linear discriminant analysis (LDA) coupled with effect size measurements (LEfSe), showed a differential profile after ketamine treatment. -

Gut Microbiota Differs in Composition and Functionality Between Children
Diabetes Care Volume 41, November 2018 2385 Gut Microbiota Differs in Isabel Leiva-Gea,1 Lidia Sanchez-Alcoholado,´ 2 Composition and Functionality Beatriz Mart´ın-Tejedor,1 Daniel Castellano-Castillo,2,3 Between Children With Type 1 Isabel Moreno-Indias,2,3 Antonio Urda-Cardona,1 Diabetes and MODY2 and Healthy Francisco J. Tinahones,2,3 Jose´ Carlos Fernandez-Garc´ ´ıa,2,3 and Control Subjects: A Case-Control Mar´ıa Isabel Queipo-Ortuno~ 2,3 Study Diabetes Care 2018;41:2385–2395 | https://doi.org/10.2337/dc18-0253 OBJECTIVE Type 1 diabetes is associated with compositional differences in gut microbiota. To date, no microbiome studies have been performed in maturity-onset diabetes of the young 2 (MODY2), a monogenic cause of diabetes. Gut microbiota of type 1 diabetes, MODY2, and healthy control subjects was compared. PATHOPHYSIOLOGY/COMPLICATIONS RESEARCH DESIGN AND METHODS This was a case-control study in 15 children with type 1 diabetes, 15 children with MODY2, and 13 healthy children. Metabolic control and potential factors mod- ifying gut microbiota were controlled. Microbiome composition was determined by 16S rRNA pyrosequencing. 1Pediatric Endocrinology, Hospital Materno- Infantil, Malaga,´ Spain RESULTS 2Clinical Management Unit of Endocrinology and Compared with healthy control subjects, type 1 diabetes was associated with a Nutrition, Laboratory of the Biomedical Research significantly lower microbiota diversity, a significantly higher relative abundance of Institute of Malaga,´ Virgen de la Victoria Uni- Bacteroides Ruminococcus Veillonella Blautia Streptococcus versityHospital,Universidad de Malaga,M´ alaga,´ , , , , and genera, and a Spain lower relative abundance of Bifidobacterium, Roseburia, Faecalibacterium, and 3Centro de Investigacion´ BiomedicaenRed(CIBER)´ Lachnospira. -

Impact Du Régime Alimentaire Sur La Dynamique Structurale Et Fonctionnelle Du Microbiote Intestinal Humain Julien Tap
Impact du régime alimentaire sur la dynamique structurale et fonctionnelle du microbiote intestinal humain Julien Tap To cite this version: Julien Tap. Impact du régime alimentaire sur la dynamique structurale et fonctionnelle du microbiote intestinal humain. Microbiologie et Parasitologie. Université Pierre et Marie Curie - Paris 6, 2009. Français. tel-02824828 HAL Id: tel-02824828 https://hal.inrae.fr/tel-02824828 Submitted on 6 Jun 2020 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. THESE DE DOCTORAT DE L’UNIVERSITE PIERRE ET MARIE CURIE Spécialité Physiologie et physiopathologie Présentée par M. Julien Tap Pour obtenir le grade de DOCTEUR de l’UNIVERSITÉ PIERRE ET MARIE CURIE Sujet de la thèse : Impact du régime alimentaire sur la dynamique structurale et fonctionnelle du microbiote intestinal humain soutenue le 16 décembre 2009 devant le jury composé de : M. Philippe LEBARON, Président du jury Mme Karine CLEMENT, Examinateur Mme Annick BERNALIER, Rapporteur Mme Gabrielle POTOCKI-VERONESE, Examinateur M. Jean FIORAMONTI, Rapporteur M. Eric PELLETIER, Examinateur Mme Marion LECLERC, Examinateur Université Pierre & Marie Curie - Paris 6 Tél. Secrétariat : 01 42 34 68 35 Bureau d’accueil, inscription des doctorants et base de Fax : 01 42 34 68 40 données Tél. -

Prevotella Multisaccharivorax Type Strain (PPPA20T)
Lawrence Berkeley National Laboratory Recent Work Title Non-contiguous finished genome sequence of the opportunistic oral pathogen Prevotella multisaccharivorax type strain (PPPA20). Permalink https://escholarship.org/uc/item/0p79h5ds Journal Standards in genomic sciences, 5(1) ISSN 1944-3277 Authors Pati, Amrita Gronow, Sabine Lu, Megan et al. Publication Date 2011-10-01 DOI 10.4056/sigs.2164949 Peer reviewed eScholarship.org Powered by the California Digital Library University of California Standards in Genomic Sciences (2011) 5:41-49 DOI:10.4056/sigs.2164949 Non-contiguous finished genome sequence of the opportunistic oral pathogen Prevotella multisaccharivorax type strain (PPPA20T) Amrita Pati1, Sabine Gronow2, Megan Lu1,3, Alla Lapidus1, Matt Nolan1, Susan Lucas1, Nancy Hammon1, Shweta Deshpande1, Jan-Fang Cheng1, Roxanne Tapia1,3, Cliff Han1,3, Lynne Goodwin1,3 Sam Pitluck1, Konstantinos Liolios1, Ioanna Pagani1, Konstantinos Mavromatis1, Natalia Mikhailova1, Marcel Huntemann1, Amy Chen4, Krishna Palaniappan4, Miriam Land1,5, Loren Hauser1,5, John C. Detter1,3, Evelyne-Marie Brambilla2, Manfred Rohde6, Markus Göker2, Tanja Woyke1, James Bristow1, Jonathan A. Eisen1,7, Victor Markowitz4, Philip Hugenholtz1,8, Nikos C. Kyrpides1, Hans-Peter Klenk2*, and Natalia Ivanova1 1 DOE Joint Genome Institute, Walnut Creek, California, USA 2 DSMZ - German Collection of Microorganisms and Cell Cultures GmbH, Braunschweig, Germany 3 Los Alamos National Laboratory, Bioscience Division, Los Alamos, New Mexico, USA 4 Biological Data Management and -

Characterization of Antibiotic Resistance Genes in the Species of the Rumen Microbiota
ARTICLE https://doi.org/10.1038/s41467-019-13118-0 OPEN Characterization of antibiotic resistance genes in the species of the rumen microbiota Yasmin Neves Vieira Sabino1, Mateus Ferreira Santana1, Linda Boniface Oyama2, Fernanda Godoy Santos2, Ana Júlia Silva Moreira1, Sharon Ann Huws2* & Hilário Cuquetto Mantovani 1* Infections caused by multidrug resistant bacteria represent a therapeutic challenge both in clinical settings and in livestock production, but the prevalence of antibiotic resistance genes 1234567890():,; among the species of bacteria that colonize the gastrointestinal tract of ruminants is not well characterized. Here, we investigate the resistome of 435 ruminal microbial genomes in silico and confirm representative phenotypes in vitro. We find a high abundance of genes encoding tetracycline resistance and evidence that the tet(W) gene is under positive selective pres- sure. Our findings reveal that tet(W) is located in a novel integrative and conjugative element in several ruminal bacterial genomes. Analyses of rumen microbial metatranscriptomes confirm the expression of the most abundant antibiotic resistance genes. Our data provide insight into antibiotic resistange gene profiles of the main species of ruminal bacteria and reveal the potential role of mobile genetic elements in shaping the resistome of the rumen microbiome, with implications for human and animal health. 1 Departamento de Microbiologia, Universidade Federal de Viçosa, Viçosa, Minas Gerais, Brazil. 2 Institute for Global Food Security, School of Biological -

Redalyc.Bacterial Diversity in Bovine Rumen by Metagenomic 16S Rdna
Acta Scientiarum. Animal Sciences ISSN: 1806-2636 [email protected] Universidade Estadual de Maringá Brasil Barbetta de Jesus, Raphael; Pine Omori, Wellington; de Macedo Lemos, Eliana Gertrudes; Marcondes de Souza, Jackson Antônio Bacterial diversity in bovine rumen by metagenomic 16S rDNA sequencing and scanning electron microscopy Acta Scientiarum. Animal Sciences, vol. 37, núm. 3, julio-septiembre, 2015, pp. 251-257 Universidade Estadual de Maringá Maringá, Brasil Available in: http://www.redalyc.org/articulo.oa?id=303141017006 How to cite Complete issue Scientific Information System More information about this article Network of Scientific Journals from Latin America, the Caribbean, Spain and Portugal Journal's homepage in redalyc.org Non-profit academic project, developed under the open access initiative Acta Scientiarum http://www.uem.br/acta ISSN printed: 1806-2636 ISSN on-line: 1807-8672 Doi: 10.4025/actascianimsci.v37i3.26535 Bacterial diversity in bovine rumen by metagenomic 16S rDNA sequencing and scanning electron microscopy Raphael Barbetta de Jesus1,2, Wellington Pine Omori1,2, Eliana Gertrudes de Macedo Lemos1,3 and Jackson Antônio Marcondes de Souza1,2* 1Faculdade de Ciências Agrárias e Veterinárias, Universidade Estadual Paulista, Via de Acesso Professor Paulo Donato Castellane, s/n, 14884- 900, Jaboticabal, São Paulo, Brazil. 2Departamento de Biologia Aplicada à Agropecuária, Faculdade de Ciências Agrárias e Veterinárias, Jaboticabal, São Paulo, Brazil. 3Departamento de Tecnologia, Faculdade de Ciências Agrárias e Veterinárias, Jaboticabal, São Paulo, Brazil. *Author for correspondence. E-mail: [email protected] ABSTRACT. The bacterial diversity by 16S rDNA partial sequencing and scanning electron microscope (SEM) of the rumen microbiome was characterized. Three Nellore bovines, cannulated at the rumen, were utilized.